
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA
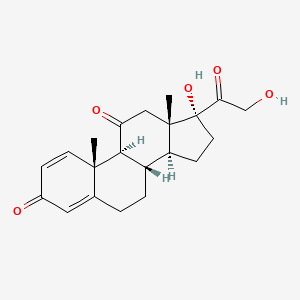

1. Acsis, Prednison
2. Apo-prednisone
3. Cortan
4. Cortancyl
5. Cutason
6. Dacortin
7. Decortin
8. Decortisyl
9. Dehydrocortisone
10. Delta-cortisone
11. Deltasone
12. Encorton
13. Encortone
14. Enkortolon
15. Kortancyl
16. Liquid Pred
17. Meticorten
18. Orasone
19. Panafcort
20. Panasol
21. Predni Tablinen
22. Prednidib
23. Predniment
24. Prednison Acsis
25. Prednison Galen
26. Prednison Hexal
27. Pronisone
28. Rectodelt
29. Sone
30. Sterapred
31. Ultracorten
32. Winpred
1. 53-03-2
2. Dehydrocortisone
3. Decortin
4. Deltasone
5. Meticorten
6. Orasone
7. Supercortil
8. Metacortandracin
9. Decortisyl
10. Rectodelt
11. Sterapred
12. Ultracorten
13. Dacortin
14. Encorton
15. Paracort
16. Cortan
17. 1,2-dehydrocortisone
18. Liquid Pred
19. Deltacortisone
20. Deltacortone
21. Ancortone
22. Colisone
23. Decortancyl
24. Deltison
25. Encortone
26. Prednilonga
27. Prednison
28. Servisone
29. Lodotra
30. Prednicen-m
31. Delta-dome
32. Di-adreson
33. Bicortone
34. Cortidelt
35. Dekortin
36. Diadreson
37. Enkorton
38. Hostacortin
39. Lisacort
40. Panafcort
41. Prednisonum
42. Prednizon
43. Pronison
44. Ultracortene
45. Zenadrid
46. Adasone
47. Cotone
48. Deltra
49. Juvason
50. Nurison
51. Winpred
52. Wojtab
53. Prednisone Intensol
54. Delta-cortelan
55. 17,21-dihydroxypregna-1,4-diene-3,11,20-trione
56. Delta E
57. In-sone
58. Cartancyl
59. Deltisona
60. Sk-prednisone
61. Prednisona
62. Prednisonum [inn-latin]
63. Prednisona [inn-spanish]
64. 1,4-pregnadiene-17alpha,21-diol-3,11,20-trione
65. Pregna-1,4-diene-3,11,20-trione, 17,21-dihydroxy-
66. Prednisone Anhydrous
67. Deltisone
68. Rayos
69. Zenadrid (veterinary)
70. Fernisone
71. Prednisone Tablets
72. 1-dehydrocortisone
73. 3en3hg4wsw
74. Nsc-10023
75. Nci-c04897
76. Nsc 10023
77. U 6020
78. Prednidib
79. Chebi:8382
80. Panasol
81. Delta-cortisone
82. Sone
83. Zenadrid [veterinary]
84. Apo-prednisone
85. Novoprednisone
86. Dellacort
87. Deltacortene
88. Econosone
89. Incocortyl
90. Parmenison
91. Predeltin
92. Prednicorm
93. Prednicort
94. Prednicot
95. Prednitone
96. Prednovister
97. Retrocortine
98. Dacorten
99. Fiasone
100. Pehacort
101. Presone
102. Delta Cortelan
103. Nisona
104. Nizon
105. Dellacort A
106. Vb0r961hzt
107. Me-korti
108. Origen Prednisone
109. (8s,9s,10r,13s,14s,17r)-17-hydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-6,7,8,9,12,14,15,16-octahydrocyclopenta[a]phenanthrene-3,11-dione
110. .delta.-cortisone
111. 1-cortisone
112. .delta.1-cortisone
113. Delta-1-cortisone
114. Ncgc00090766-01
115. Precort
116. .delta.1-dehydrocortisone
117. Dsstox_cid_1185
118. Meticorten (veterinary)
119. Delta(sup 1)-cortisone
120. Delta-1-dehydrocortisone
121. Dsstox_rid_75997
122. Dsstox_gsid_21185
123. (8s,9s,10r,13s,14s,17r)-17-hydroxy-17-(2-hydroxyacetyl)-10,13-dimethyl-7,8,9,10,12,13,14,15,16,17-decahydro-3h-cyclopenta[a]phenanthrene-3,11(6h)-dione
124. Delta(sup 1)-dehydrocortisone
125. Lodtra
126. Smr000718760
127. Smr001227202
128. Delta E.
129. Ccris 2646
130. Hsdb 3168
131. Mls002638114
132. Einecs 200-160-3
133. Mfcd00003608
134. Unii-vb0r961hzt
135. Prednisone [usp:inn:ban]
136. 1,4-pregnadiene-17-alpha,21-diol-3,11,20-trione
137. Ai3-52939
138. .delta.sone
139. .delta.-cortone
140. .delta.-cortelan
141. Cas-53-03-2
142. Deltadehydrocortisone
143. Prednisone(adasone)
144. Prestwick_405
145. .delta. E
146. .delta.-e
147. Prednisone (adasone)
148. Prednisone, >=98%
149. Prednisone [mi]
150. Prednisone [inn]
151. Prestwick0_000077
152. Prestwick1_000077
153. Prestwick2_000077
154. Prestwick3_000077
155. Prednisone [hsdb]
156. Prednisone [iarc]
157. .delta.(sup1)-cortisone
158. P1276
159. Modified-release Prednisone
160. Prednisone [vandf]
161. Chembl635
162. Prednisone [mart.]
163. Schembl3288
164. Prednisone [usp-rs]
165. Prednisone [who-dd]
166. Bspbio_000293
167. Mls001061265
168. Mls001304073
169. Mls001335907
170. Mls001335908
171. Mls002154191
172. Mls002207083
173. Mls002548880
174. Spbio_002214
175. Bpbio1_000323
176. Gtpl7096
177. Megxm0_000443
178. Prednisone [green Book]
179. Dtxsid4021185
180. Prednisone [orange Book]
181. Acon0_000082
182. Acon1_000297
183. Prednisone [ep Monograph]
184. Hms1568o15
185. Hms2090j13
186. Hms2095o15
187. Hms2231i24
188. Hms3039k07
189. Hms3259i09
190. Hms3712o15
191. Hms3884c04
192. Prednisone [usp Monograph]
193. 1,21-diol-3,11,20-trione
194. Bcp09049
195. Hy-b0214
196. Nsc10023
197. Prednisone Tablets [usp-rs]
198. Pregna-1,4-diene-3,11,20-trione Monohydrate, 17,21-dihydroxy-
199. Zinc3875357
200. Tox21_111014
201. Tox21_201564
202. Tox21_300196
203. Bdbm50550126
204. Lmst02030180
205. S1622
206. Akos005267096
207. Akos007930684
208. Prednisone 100 Microg/ml In Methanol
209. Tox21_111014_1
210. Ccg-220077
211. Db00635
212. Nc00475
213. Ncgc00090766-02
214. Ncgc00090766-03
215. Ncgc00090766-04
216. Ncgc00090766-05
217. Ncgc00090766-07
218. Ncgc00254096-01
219. Ncgc00259113-01
220. Prednisone, Tested According To Ph.eur.
221. Ac-11112
222. As-11685
223. Nci60_000008
224. Prednisone 100 Microg/ml In Acetonitrile
225. Prednisolone Impurity B [ep Impurity]
226. Pregna-1,11,20-trione, 17,21-hydroxy-
227. En300-52605
228. Pregna-1,11,20-trione, 17,21-dihydroxy-
229. C07370
230. 003p608
231. Q424972
232. Sr-01000837536
233. Sr-01000837536-3
234. Brd-k85883481-001-04-2
235. Brd-k85883481-001-08-3
236. Brd-k85883481-001-25-7
237. 17alpha,21-dihydroxypregna-1,4-diene-3,11,20-trione
238. Wln: L E5 B666 Cv Ov Ahttt&j A1 E1 Fv1q Fq
239. Prednisone, British Pharmacopoeia (bp) Reference Standard
240. Prednisone, European Pharmacopoeia (ep) Reference Standard
241. Prednisone, United States Pharmacopeia (usp) Reference Standard
242. Prednisone, Pharmaceutical Secondary Standard; Certified Reference Material
243. Prednisone For Peak Identification, European Pharmacopoeia (ep) Reference Standard
244. Prednisone, Pharmaceutical Secondary Standard; Traceable To Usp, Pheur And Bp
245. (8s,10r,13s,17r)-17-hydroxy-17-(2-hydroxy-acetyl)-10,13-dimethyl-7,8,9,10,12,13,14,15,16,17-decahydro-6h-cyclopenta[a]phenanthrene-3,11-dione
246. Prednisone Solution, 100 Mug/ml In Acetonitrile, Ampule Of 1 Ml, Certified Reference Material
| Molecular Weight | 358.4 g/mol |
|---|---|
| Molecular Formula | C21H26O5 |
| XLogP3 | 1.5 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 358.17802393 g/mol |
| Monoisotopic Mass | 358.17802393 g/mol |
| Topological Polar Surface Area | 91.7 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 764 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 6 | |
|---|---|
| Drug Name | Prednisone |
| PubMed Health | Prednisone (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Immune Suppreant |
| Drug Label | Each tablet for oral administration contains:Prednisone.............................................................1 mg, 2.5 mg, 5 mg, 10 mg, 20 mg, and 50 mgEach 5 mL of oral solution for oral administration contains:Prednisone........................ |
| Active Ingredient | Prednisone |
| Dosage Form | Tablet; Solution |
| Route | Oral |
| Strength | 2.5mg; 1mg; 5mg; 50mg; 10mg; 5mg/5ml; 20mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Jubilant Cadista; Hikma Pharms; Roxane; Watson Labs; Mutual Pharm; Contract Pharmacal |
| 2 of 6 | |
|---|---|
| Drug Name | Prednisone intensol |
| PubMed Health | Prednisone (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Immune Suppreant |
| Drug Label | Each tablet for oral administration contains: Prednisone.....................................................1 mg, 2.5 mg, 5 mg, 10 mg, 20 mg, and 50 mgEach 5 mL of oral solution for oral administration contains: Prednisone.............................. |
| Active Ingredient | Prednisone |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 5mg/ml |
| Market Status | Prescription |
| Company | Roxane |
| 3 of 6 | |
|---|---|
| Drug Name | Rayos |
| Drug Label | The active ingredient in RAYOS is prednisone (a corticosteroid). Corticosteroids are adrenocortical steroids, both naturally occurring and synthetic. The molecular formula for prednisone is C21H26O5. The chemical name for prednisone is 17,21-dihydrox... |
| Active Ingredient | Prednisone |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 1mg; 5mg; 2mg |
| Market Status | Prescription |
| Company | Horizon Pharma |
| 4 of 6 | |
|---|---|
| Drug Name | Prednisone |
| PubMed Health | Prednisone (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Immune Suppreant |
| Drug Label | Each tablet for oral administration contains:Prednisone.............................................................1 mg, 2.5 mg, 5 mg, 10 mg, 20 mg, and 50 mgEach 5 mL of oral solution for oral administration contains:Prednisone........................ |
| Active Ingredient | Prednisone |
| Dosage Form | Tablet; Solution |
| Route | Oral |
| Strength | 2.5mg; 1mg; 5mg; 50mg; 10mg; 5mg/5ml; 20mg |
| Market Status | Prescription |
| Company | Vintage Pharms; Jubilant Cadista; Hikma Pharms; Roxane; Watson Labs; Mutual Pharm; Contract Pharmacal |
| 5 of 6 | |
|---|---|
| Drug Name | Prednisone intensol |
| PubMed Health | Prednisone (By mouth) |
| Drug Classes | Endocrine-Metabolic Agent, Immune Suppreant |
| Drug Label | Each tablet for oral administration contains: Prednisone.....................................................1 mg, 2.5 mg, 5 mg, 10 mg, 20 mg, and 50 mgEach 5 mL of oral solution for oral administration contains: Prednisone.............................. |
| Active Ingredient | Prednisone |
| Dosage Form | Solution |
| Route | Oral |
| Strength | 5mg/ml |
| Market Status | Prescription |
| Company | Roxane |
| 6 of 6 | |
|---|---|
| Drug Name | Rayos |
| Drug Label | The active ingredient in RAYOS is prednisone (a corticosteroid). Corticosteroids are adrenocortical steroids, both naturally occurring and synthetic. The molecular formula for prednisone is C21H26O5. The chemical name for prednisone is 17,21-dihydrox... |
| Active Ingredient | Prednisone |
| Dosage Form | Tablet, delayed release |
| Route | Oral |
| Strength | 1mg; 5mg; 2mg |
| Market Status | Prescription |
| Company | Horizon Pharma |
Anti-Inflammatory Agents; Antineoplastic Agents, Hormonal; Glucocorticoids
National Library of Medicine's Medical Subject Headings. Prednisone. Online file (MeSH, 2016). Available from, as of October 28, 2016: https://www.nlm.nih.gov/mesh/2016/mesh_browser/MBrowser.html
Prednisone is usually considered the oral glucocorticoid of choice for anti-inflammatory or immunosuppressant effects. Because it has only minimal mineralocorticoid properties, the drug is inadequate alone for the management of adrenocortical insufficiency. If prednisone is used in the treatment of this condition, concomitant therapy with a mineralocorticoid is also required.
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3122-3
Prednisone tablets and solutions are indicated in the following conditions: Endocrine Disorders: Primary or secondary adrenocortical insufficiency (hydrocortisone or cortisone is the first choice: synthetic analogs may be used in conjunction with mineralocorticoids where applicable; in infancy mineralocorticoid supplementation is of particular importance); congenital adrenal hyperplasia; hypercalcemia associated with cancer; nonsuppurative thyroiditis. /Included in US product label/
NIH; DailyMed. Current Medication Information for Prednisone (Pednisone Tablet) Prednisone (Prednisone Solution) Prednisone Intensol (Prednisone Intensol Solution, Concentrate) (Updated: July 2016). Available from, as of November 22, 2016: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=3115aef0-fd50-4ec8-a064-3effb695f3f2
Prednisone tablets and solutions are indicated in the following conditions: Rheumatic Disorders: As adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in: psoriatic arthritis, rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low-dose maintenance therapy), ankylosing spondylitis, acute and subacute bursitis, acute nonspecific tenosynovitis, acute gouty arthritis, post-traumatic osteoarthritis, synovitis of osteoarthritis, epicondylitis. /Included in US product label/
NIH; DailyMed. Current Medication Information for Prednisone (Pednisone Tablet) Prednisone (Prednisone Solution) Prednisone Intensol (Prednisone Intensol Solution, Concentrate) (Updated: July 2016). Available from, as of November 22, 2016: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=3115aef0-fd50-4ec8-a064-3effb695f3f2
For more Therapeutic Uses (Complete) data for PREDNISONE (19 total), please visit the HSDB record page.
The profound effects of prednisone on the immune system place patients at increased risk of developing infections of various types. Prednisone may mask some of the signs of infection, and may decrease host resistance and interfere with the ability to localize infections. During prednisone therapy, a polymorphonuclear leukocytosis may develop and may give rise to confusion in the diagnosis of infection. This elevation is dose-related.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 302 (1981)
Psychiatric reactions have been reported in 4-36% of patients. These disturbances may take various forms, for example, insomnia, changes in mood or psyche, and psychopathies of the manic-depressive or schizophrenic type.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 302 (1981)
Ophthalmic complications include the development of posterior subcapsular cataracts, and increased intraocular pressure which may lead to glaucoma. In patients with ocular herpes simplex, it may cause corneal perforation.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 302 (1981)
There are numerous endocrine side effects. Most frequent is development of the Cushingoid state. Fatty deposits in the mediastinum causing mediastinal widening may simulate mediastinal lymphadenopathy. Menstrual irregularities, including amenorrhoea, may occur. There may be secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress, as in trauma, surgery or illness. The processes of recovery of normal pituitary and adrenal function require about 1 year in some patients. There may be stunted growth and delayed skeletal maturation in children. Prednisone causes decreased carbohydrate tolerance and may unmask the features of latent diabetes.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 301-2 (1981)
For more Drug Warnings (Complete) data for PREDNISONE (38 total), please visit the HSDB record page.
Prednisone is indicated as an anti-inflammatory or immunosuppressive drug for allergic, dermatologic, gastrointestinal, hematologic, ophthalmologic, nervous system, renal, respiratory, rheumatologic, infectious, endocrine, or neoplastic conditions as well as in organ transplant.
Corticosteroids bind to the glucocorticoid receptor, inhibiting pro-inflammatory signals, and promoting anti-inflammatory signals. Prednisone has a short duration of action as the half life is 2-3 hours. Corticosteroids have a wide therapeutic window as patients make require doses that are multiples of what the body naturally produces. Patients taking corticosteroids should be counselled regarding the risk of hypothalamic-pituitary-adrenal axis suppression and increased susceptibility to infections.
Antineoplastic Agents, Hormonal
Antineoplastic agents that are used to treat hormone-sensitive tumors. Hormone-sensitive tumors may be hormone-dependent, hormone-responsive, or both. A hormone-dependent tumor regresses on removal of the hormonal stimulus, by surgery or pharmacological block. Hormone-responsive tumors may regress when pharmacologic amounts of hormones are administered regardless of whether previous signs of hormone sensitivity were observed. The major hormone-responsive cancers include carcinomas of the breast, prostate, and endometrium; lymphomas; and certain leukemias. (From AMA Drug Evaluations Annual 1994, p2079) (See all compounds classified as Antineoplastic Agents, Hormonal.)
Glucocorticoids
A group of CORTICOSTEROIDS that affect carbohydrate metabolism (GLUCONEOGENESIS, liver glycogen deposition, elevation of BLOOD SUGAR), inhibit ADRENOCORTICOTROPIC HORMONE secretion, and possess pronounced anti-inflammatory activity. They also play a role in fat and protein metabolism, maintenance of arterial blood pressure, alteration of the connective tissue response to injury, reduction in the number of circulating lymphocytes, and functioning of the central nervous system. (See all compounds classified as Glucocorticoids.)
Anti-Inflammatory Agents
Substances that reduce or suppress INFLAMMATION. (See all compounds classified as Anti-Inflammatory Agents.)
A - Alimentary tract and metabolism
A07 - Antidiarrheals, intestinal antiinflammatory/antiinfective agents
A07E - Intestinal antiinflammatory agents
A07EA - Corticosteroids acting locally
A07EA03 - Prednisone
H - Systemic hormonal preparations, excl. sex hormones and insulins
H02 - Corticosteroids for systemic use
H02A - Corticosteroids for systemic use, plain
H02AB - Glucocorticoids
H02AB07 - Prednisone
Absorption
Oral prednisone has a Tmax of 2 hours, while the delayed-release formulation has a Tmax of 6-6.5 hours. A 5mg dose of prednisone has an AUC of 572mL/min/1.73m2, a 20mg dose of prednisone has an AUC of 1034mL/min/1.73m2, and a 50mg dose of prednisone has an AUC of 2271mL/min/1.73m2. Data regarding the Cmax of prednisone is not readily available.
Route of Elimination
Prednisone is excreted mainly in the urine as sulfate and glucuronide conjugates.
Volume of Distribution
Data regarding the volume of distribution for prednisone is not readily available. However, a 0.15mg/kg dose of prednisolone has a volume of distribution of 29.3L, while a 0.30mg/kg dose has a volume of distribution of 44.2L.
Clearance
Data regarding the clearance of prednisone is not readily available. A 5.5g/h/kg infusion of prednisolone has an average clearance of 0.0660.12L/h/kg, while a 0.150.03L/h/kg infusion has an average clearance of 0.15L/h/kg.
Thirty minutes after iv administration of (3)H-prednisone to a monkey, the concentration of prednisone was highest in the kidney. The drug was also found in the liver, spleen, lung, small intestine, serum and bile. The concentration of prednisolone was highest in the liver. It was also found in the kidney, pancreas, spleen, lung, small intestine, serum and bile.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 300 (1981)
Prednisone is readily absorbed from the gut. Serum concentrations of prednisone and prednisolone, its active metabolite, have been found to be maximal 1 hour after oral administration of a 5-mg tablet of prednisone to beagle dogs. Following both ip and oral administration of prednisone to mice, serum levels of prednisone, prednisolone and other metabolites were maximal at 15 min. These levels were higher in mice given ip injections of prednisone than in those receiving the same doses by the oral route. Oral administration of prednisone to dogs and monkeys led to serum levels comparable with those following iv injections, but individual variations were relatively large.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 299 (1981)
Prednisone is readily absorbed from the gut. In a series of 22 normal subjects, the mean peak serum concentration was 930 ug/L (range, 508-1579) following oral administration of a 50 mg tablet.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 303 (1981)
The protein binding characteristics of prednisone and prednisolone, alone and together, in human and rabbit plasma and human serum albumin, are reported. The kinetics of prednisolone binding were nonlinear and those of prednisone were linear in both human and rabbit plasma; prednisone binding was linear with human serum albumin, although to a lesser degree. It is suggested that prednisone binds to proteins other than albumin in plasma. Binding of prednisone was not influenced by prednisolone. The results support the hypothesis that the protein binding characteristics of prednisone and prednisolone do not explain the reported nonlinear pharmacokinetics of prednisone.
PMID:3593903 Ferry JJ, Wagner JG; Biopharm Drug Dispos 8 (3): 261-72 (1987)
Administration of physiologic doses unlikely to adversely affect infant. FDA Category: C (C = Studies in laboratory animals have revealed adverse effects on the fetus (teratogenic, embryocidal, etc.) but there are no controlled studies in pregnant women. The benefits from use of the drug in pregnant women may be acceptable despite its potential risks, or there are no laboratory animal studies or adequate studies in pregnant women.) /Adrenocorticosteroids/ /from table II/
Stockton, D.L. and A.S. Paller. J Am Acad Dermatol 23 (1):87-103 (1990)
Prednisoneis metabolized to 17,21-dihydroxy-pregnan-1,4,6-trien-3,11,30-trione (M-XVII), 20-dihydro-prednisone (M-V), 6hydroxy-prednisone (M-XII), 6-hydroxy-prednisone (M-XIII), or 20-dihydro-prednisone (M-IV).20-dihydro-prednisone is metabolized to 17,20,21-trihydroxy-5-pregn-1-en-3,11-dione(M-XVIII). Prednison is reversibly metabolized to [prednisolone].Prednisolone is metabolized to 6-prednisolone (M-XI), 20-dihydro-prednisolone (M-III), 20-dihydro-prednisolone (M-II), 6hydroxy-prednisolone (M-VII), or 6hydroxy-prednisolone(M-VI).6hydroxy-prednisolone is metabolized to 6,11,17,20,21-pentahydroxypregnan-1,4-diene-3-one (M-X).6hydroxy-prednisolone is metabolized to 6,11,17,20,21-pentahydroxypregnan-1,4-diene-3-one (M-VIII), 6,11,17,20,21-pentahydroxypregnan-1,4-diene-3-one (M-IX), and 6,11,17,21-tetrahydroxy-5-pregn-1-en-3,20-dione (M-XIV).MVIII is metabolized to 6,11,17,20,21-pentahydroxy-5-pregn-1-en-3-one (M-XV) and then to MXIV, while MIX is metabolized to 6,11,17,20,21-pentahydroxy-5-pregn-1-en-3-one (M-XVI) and then to MXIV.These metabolites and their glucuronide conjugates are excreted predominantly in the urine.
In one study after an oral dose of prednisone, the plasma prednisolone concentration peaked between 60 and 120 min and then declined exponentially. After rapid iv injection of steroid, the plasma prednisolone concentration peaked within 10 to 20 min. An initial rapid distribution phase succeeded by a slower decay phase was expressed by a biphasic exponential disappearance curve of the plasma prednisolone concentration versus time. Plasma prednisolone concentrations achieved with an oral dose of prednisone were in the same range as those obtained during the second phase after iv administration.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 303 (1981)
Reduction of the 11-oxo to the 11alpha-hydroxyl group by the enzyme 11beta-hydroxydehydrogenase converts prednisone to prednisolone, its biologically active form. This reaction takes place mainly in the liver, and may proceed satisfactorily even in the presence of liver disease
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 303 (1981)
In vitro, prednisone is converted to prednisolone by liver, lung and renal tissue. Conversely, prednisolone is converted to prednisone by renal tissue.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 300 (1981)
... The aim of this work was to evaluate the effects of these corticosteroids on the expression of several forms of cytochromes p450, including p450 1A2, 2D6, 2E1, and 3A, and on cyclosporin A oxidase activity in human liver. For this purpose, human hepatocytes prepared from lobectomies were maintained in culture in a serum-free medium, in collagen-coated dishes, for 96-144 hr, in the absence or presence of 50-100 uM corticosteroids, rifampicin, or dexamethasone. To mimic more closely the current clinical protocol, hepatocyte cultures were also co-treated with corticosteroids and cyclosporin A or ketoconazole (a selective inhibitor of cytochromes p450 3A). Cyclosporin A oxidase activity, intracellular retention of cyclosporin A oxidized metabolites within hepatocytes, accumulation of cytochromes p450 proteins and corresponding messages, and de novo synthesis and half-lives of these cytochromes p450 were measured in parallel in these cultures. Our results, obtained from seven different hepatocyte cultures, showed that 1) dexamethasone and prednisone, but not prednisolone or methylprednisolone, were inducers of cytochrome p450 3A, at the level of protein and mRNA accumulation, as well as of cyclosporin A oxidase activity, known to be predominantly catalyzed by these cytochromes p450; 2) although corticosteroids are known to be metabolized in human liver, notably by cytochrome p450 3A, partial or total inhibition of this cytochromes p450 by cyclosporin or ketoconazole, respectively, did not affect the inducing efficiency of these molecules; 3) corticosteroids did not affect the half-life of cytochrome p450 3A or the accumulation of other forms of cytochromes p450, including 1A2, 2D6, and 2E1; 4) chronic treatment of cells with cyclosporin did not affect cytochrome p450 3A accumulation; 5) corticosteroids were all competitive inhibitors of cyclosporin A oxidase in human liver microsomes, with Ki values of 61 + or - 12, 125 + or - 25, 190 + or - 38, and 210 + or - 42 uM for dexamethasone, prednisolone, prednisone, and methylprednisolone, respectively; and 6) chronic treatment of cells with corticosteroids did not influence the excretion of oxidized metabolites of cyclosporin from the cells.
PMID:1614409 Pichard L et al; Mol Pharmacol 41 (6): 1047-55 (1992)
Prednisone and its active metabolite [prednisolone] have half lives of 2-3 hours from both immediate and delayed release preparations.
In a series of 22 normal subjects, the mean peak serum concentration was 930 ug/L (range, 508-1579) following oral administration of a 50 mg tablet. The overall mean serum half-life was 2.95 hours
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V26 303 (1981)
Prednisone is first metabolized in the liver to its active form, prednisolone, a glucocorticoid agonist corticosteroid. The short term effects of corticosteroids are decreased vasodilation and permeability of capillaries, as well as decreased leukocyte migration to sites of inflammation. Corticosteroids binding to the glucocorticoid receptor mediates changes in gene expression that lead to multiple downstream effects over hours to days. Glucocorticoids inhibit neutrophil apoptosis and demargination; they inhibit phospholipase A2, which decreases the formation of arachidonic acid derivatives; they inhibit NF-Kappa B and other inflammatory transcription factors; they promote anti-inflammatory genes like interleukin-10. Lower doses of corticosteroids provide an anti-inflammatory effect, while higher doses are immunosuppressive. High doses of glucocorticoids for an extended period bind to the mineralocorticoid receptor, raising sodium levels and decreasing potassium levels.
In physiologic doses, corticosteroids are administered to replace deficient endogenous hormones. In larger (pharmacologic) doses, glucocorticoids decrease inflammation by stabilizing leukocyte lysosomal membranes, preventing release of destructive acid hydrolases from leukocytes; inhibiting macrophage accumulation in inflamed areas; reducing leukocyte adhesion to capillary endothelium; reducing capillary wall permeability and edema formation; decreasing complement components; antagonizing histamine activity and release of kinin from substrates; reducing fibroblast proliferation, collagen deposition, and subsequent scar tissue formation; and possibly by other mechanisms as yet unknown. The drugs suppress the immune response by reducing activity and volume of the lymphatic system, producing lymphocytopenia, decreasing immunoglobulin and complement concentrations, decreasing passage of immune complexes through basement membranes, and possibly by depressing reactivity of tissue to antigen-antibody interactions. Glucocorticoids stimulate erythroid cells of bone marrow, prolong survival time of erythrocytes and platelets, and produce neutrophilia and eosinopenia. Glucocorticoids promote gluconeogenesis, redistribution of fat from peripheral to central areas of the body, and protein catabolism, which results in negative nitrogen balance. They reduce intestinal absorption and increase renal excretion of calcium. /Corticosteroids/
American Society of Health-System Pharmacists 2016; Drug Information 2016. Bethesda, MD. 2016, p. 3097
Glucocorticoids are capable of suppressing the inflammatory process through numerous pathways. They interact with specific intracellular receptor proteins in target tissues to alter the expression of corticosteroid-responsive genes. Glucocorticoid-specific receptors in the cell cytoplasm bind with steroid ligands to form hormone-receptor complexes that eventually translocate to the cell nucleus. There these complexes bind to specific DNA sequences and alter their expression. The complexes may induce the transcription of mRNA leading to synthesis of new proteins. Such proteins include lipocortin, a protein known to inhibit PLA2a and thereby block the synthesis of prostaglandins, leukotrienes, and PAF. Glucocorticoids also inhibit the production of other mediators including AA metabolites such as COX, cytokines, the interleukins, adhesion molecules, and enzymes such as collagenase. /Glucocorticoids/
Kahn, C.M. (Ed.); The Merck Veterinary Manual 9th ed. Merck & Co. Whitehouse Station, NJ. 2005, p. 2128
About the Company : EUROAPI is the market leader in small molecule APIs with projected sales of about €1 billion in 2022. With around 200 APIs, it has one of the largest portfolios in the market. Th...
About the Company : Transo-Pharm, a fully licensed and certified distributor, specializes in pharmaceutical components for the health and veterinary industries. It offers support to clients throughout...
About the Company : DKSH, founded with the goal of improving people's lives, assists businesses with market expansion and business growth in both existing and emerging markets. It has been fostering g...
About the Company : Curia is a global contract research, development and manufacturing organization (CDMO), offering products and services across the drug development spectrum to help our partners tur...

About the Company : Founded more than 40 years ago, our purpose has always been to provide high-quality, affordable medicines to the people who need them. Based on the solid foundation we have built, ...

About the Company : With the high technology and excellent services, Xianju pharma earned a top worldwide reputation for quality in pharmaceutical industry. Xianju pharma has dedicated to scientific r...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Prednisone
Dosage Form : Prednisone 5Mg 20 Joined' Oral Use
Dosage Strength : 20 CPR 5 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : PREDNISONE DOC GENERIC
Dosage Form : Tablets
Dosage Strength : 5 mg
Packaging : 20 UNITS 5 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Prednisone Galepharm
Dosage Form : Tabl
Dosage Strength : 5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Prednisone Galepharm
Dosage Form : Tabl
Dosage Strength : 5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Lodotra
Dosage Form : MODIFIED-RELEASE TABLET
Dosage Strength : 2 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Lodotra
Dosage Form : Ret Tabl
Dosage Strength : 2mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Prednisone
Dosage Form : Prednisone 25Mg 10 Units Oral Use
Dosage Strength : 10 CPR 25 mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Prednisone Streuli
Dosage Form : Tabl
Dosage Strength : 5mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

Regulatory Info :
Registration Country : Spain
Brand Name : Prednisone Cinfa 2.5Mg 30 Pills Efg
Dosage Form : Tablet
Dosage Strength : 2.5 Mg/Tablet
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Spain
Brand Name : Dacortin 5Mg 30 Tablets
Dosage Form : Tablet
Dosage Strength : 5 Mg/Tablet
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?