
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
API
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
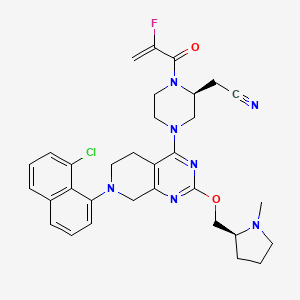

1. 2-((s)-4-(7-(8-chloronaphthalen-1-yl)-2-(((s)-1-methylpyrrolidin-2-yl)methoxy)-5,6,7,8-tetrahydropyrido(3,4-d)pyrimidin-4-yl)-1-(2-fluoroacryloyl)piperazin-2-yl)acetonitrile
2. 2-piperazineacetonitrile, 4-(7-(8-chloro-1-naphthalenyl)-5,6,7,8-tetrahydro-2-(((2s)-1-methyl-2-pyrrolidinyl)methoxy)pyrido(3,4-d)pyrimidin-4-yl)-1-(2-fluoro-1-oxo-2-propen-1-yl)-, (2s)-
3. Mrtx-849
4. Mrtx849
1. Mrtx849
2. 2326521-71-3
3. Mrtx-849
4. Adagrasib [usan]
5. Kras G12c Inhibitor Mrtx849
6. 8eoo6hqf8y
7. 2-((s)-4-(7-(8-chloronaphthalen-1-yl)-2-(((s)-1-methylpyrrolidin-2-yl)methoxy)-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl)-1-(2-fluoroacryloyl)piperazin-2-yl)acetonitrile
8. 2-[(2s)-4-[7-(8-chloronaphthalen-1-yl)-2-[[(2s)-1-methylpyrrolidin-2-yl]methoxy]-6,8-dihydro-5h-pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-yl]acetonitrile
9. 2-piperazineacetonitrile, 4-[7-(8-chloro-1-naphthalenyl)-5,6,7,8-tetrahydro-2-[[(2s)-1-methyl-2-pyrrolidinyl]methoxy]pyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoro-1-oxo-2-propen-1-yl)-, (2s)-
10. 2-piperazineacetonitrile, 4-(7-(8-chloro-1-naphthalenyl)-5,6,7,8-tetrahydro-2-(((2s)-1-methyl-2-pyrrolidinyl)methoxy)pyrido(3,4-d)pyrimidin-4-yl)-1-(2-fluoro-1-oxo-2-propen-1-yl)-, (2s)-
11. Adagrasib [inn]
12. Unii-8eoo6hqf8y
13. Adagrasib [who-dd]
14. Chembl4594350
15. Schembl20974691
16. Gtpl10888
17. Dtxsid801336759
18. Bcp31538
19. Ex-a3258
20. Mrtx-849; Mrtx 849
21. Bdbm50539763
22. Mfcd32263433
23. Nsc831453
24. S8884
25. Who 11519
26. Akos037648997
27. At23561
28. Nsc-831453
29. Compound 20 [pmid: 32250617]
30. Ac-35659
31. Bm177692
32. Bs-16211
33. Hy-130149
34. Cs-0105265
35. A936721
36. ((2s)-4-(7-(8-chloronaphthalen-1-yl)-2-(((2s)-1- Methylpyrrolidin-2-yl)methoxy)-5,6,7,8- Tetrahydropyrido(3,4-d)pyrimidin-4-yl)-1-(2-fluoroprop2-enoyl)piperazin-2-yl)acetonitrile
37. [(2s)-4-[7-(8-chloro-1-naphthyl)-2-{[(2s)-1-methyl-2-pyrrolidinyl]methoxy}-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroacryloyl)-2-piperazinyl]acetonitrile
| Molecular Weight | 604.1 g/mol |
|---|---|
| Molecular Formula | C32H35ClFN7O2 |
| XLogP3 | 5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 7 |
| Exact Mass | 603.2524792 g/mol |
| Monoisotopic Mass | 603.2524792 g/mol |
| Topological Polar Surface Area | 88.8 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 1060 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
MRTX849 is an experimental KRAS inhibitor being investigated for the treatment of KRAS G12C mutant lung and colon adenocarcinomas.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Normally GTP binds to KRAS, activating the protein and promoting effectors to the MAP kinase pathway. GTP is hydrolyzed to GDP, and KRAS is inactivated. KRAS G12C mutations impair hydrolysis of GTP, leaving it in the active form. MRTX849 inhibits KRAS in these types of cancers. This mutation is present in 13% of non small cell lung cancer, 3% of colorectal and appendix cancer, and 1-3% of solid tumors.
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
KRAZATI (adagrasib), a potent oral small-molecule inhibitor of KRASG12C, is indicated in combination with cetuximab for patients with locally advanced or metastatic colorectal cancer.
Lead Product(s): Adagrasib,Cetuximab
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 21, 2024

Bristol Myers Squibb Announces FDA Approval of KRAZATI with Cetuximab for CRC
Details : KRAZATI (adagrasib), a potent oral small-molecule inhibitor of KRASG12C, is indicated in combination with cetuximab for patients with locally advanced or metastatic colorectal cancer.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 21, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
KRAZATI (adagrasib), a selective oral inhibitor of KRASG12C, is being evaluated with cetuximab for KRASG12C-mutated locally advanced or metastatic colorectal cancer.
Lead Product(s): Adagrasib,Cetuximab
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: Phase I/ Phase IIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable April 08, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Adagrasib,Cetuximab
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
KRAZATI-Cetuximab Shows Activity for KRAS G12C-Mutated Metastatic Colorectal Cancer
Details : KRAZATI (adagrasib), a selective oral inhibitor of KRASG12C, is being evaluated with cetuximab for KRASG12C-mutated locally advanced or metastatic colorectal cancer.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
April 08, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
KRAZATI (adagrasib) is a selective oral KRASG12C inhibitor, currently in Phase III trials for treating locally advanced or metastatic non-small cell lung cancer.
Lead Product(s): Adagrasib
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Bristol Myers Squibb
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable April 01, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Adagrasib
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Bristol Myers Squibb
Deal Size : Not Applicable
Deal Type : Not Applicable
Zai Lab and Bristol Myers Squibb Announce KRYSTAL-12 Trial Meets Endpoint
Details : KRAZATI (adagrasib) is a selective oral KRASG12C inhibitor, currently in Phase III trials for treating locally advanced or metastatic non-small cell lung cancer.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
April 01, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Krazati (adagrasib) is an oral small-molecule KRAS G12C inhibitor under evaluation for treating KRASG12C-mutated advanced/metastatic non-small cell lung cancer.
Lead Product(s): Adagrasib
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 28, 2024

Bristol Myers Squibb’s KRYSTAL-12 Trial for KRAZATI Meets Primary Endpoint
Details : Krazati (adagrasib) is an oral small-molecule KRAS G12C inhibitor under evaluation for treating KRASG12C-mutated advanced/metastatic non-small cell lung cancer.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 28, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Krazati (adagrasib) is a selective KRAS G12C inhibitor under evaluation for KRAS G12C-mutated metastatic colorectal cancer.
Lead Product(s): Adagrasib,Cetuximab,BMS-986466
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: Phase I/ Phase IIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 20, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Adagrasib,Cetuximab,BMS-986466
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
FDA Accepts KRAZATI® Supplemental NDA for KRAS G12C-Mutated Cancer Treatment
Details : Krazati (adagrasib) is a selective KRAS G12C inhibitor under evaluation for KRAS G12C-mutated metastatic colorectal cancer.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 20, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
BMS strengthens and diversifies oncology portfolio by gaining Krazati (adagrasib), a best-in-class KRASG12C inhibitor for advanced non-small cell lung cancer harboring a KRASG12C mutation.
Lead Product(s): Adagrasib
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Bristol Myers Squibb
Deal Size: $5,800.0 million Upfront Cash: $5,800.0 million
Deal Type: Acquisition January 23, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Adagrasib
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Bristol Myers Squibb
Deal Size : $5,800.0 million
Deal Type : Acquisition
Bristol Myers Squibb Completes Acquisition Of Mirati Therapeutics To Strengthen Oncology
Details : BMS strengthens and diversifies oncology portfolio by gaining Krazati (adagrasib), a best-in-class KRASG12C inhibitor for advanced non-small cell lung cancer harboring a KRASG12C mutation.
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : $5,800.0 million
January 23, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The collaboration aims to evaluate the combination of KO-2806, a next-generation farnesyl transferase inhibitor (FTI), and adagrasib, a highly selective KRASG12C inhibitor, in patients with KRASG12C-mutated non-small cell lung cancer (NSCLC).
Lead Product(s): KO-2806,Adagrasib
Therapeutic Area: Oncology Brand Name: KO-2806
Study Phase: PreclinicalProduct Type: Small molecule
Sponsor: Mirati Therapeutics
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Collaboration November 02, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : KO-2806,Adagrasib
Therapeutic Area : Oncology
Highest Development Status : Preclinical
Partner/Sponsor/Collaborator : Mirati Therapeutics
Deal Size : Undisclosed
Deal Type : Collaboration
Details : The collaboration aims to evaluate the combination of KO-2806, a next-generation farnesyl transferase inhibitor (FTI), and adagrasib, a highly selective KRASG12C inhibitor, in patients with KRASG12C-mutated non-small cell lung cancer (NSCLC).
Brand Name : KO-2806
Molecule Type : Small molecule
Upfront Cash : Undisclosed
November 02, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Through the acquisition, BMS strengthens and diversifies oncology portfolio by gaining Krazati (adagrasib), a best-in-class KRASG12C inhibitor approved by the U.S. FDA for the treatment of patients with advanced non-small cell lung cancer harboring a KRASG12C mutation.
Lead Product(s): Adagrasib
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Bristol Myers Squibb
Deal Size: $5,800.0 million Upfront Cash: $5,800.0 million
Deal Type: Acquisition October 08, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Adagrasib
Therapeutic Area : Oncology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Bristol Myers Squibb
Deal Size : $5,800.0 million
Deal Type : Acquisition
Details : Through the acquisition, BMS strengthens and diversifies oncology portfolio by gaining Krazati (adagrasib), a best-in-class KRASG12C inhibitor approved by the U.S. FDA for the treatment of patients with advanced non-small cell lung cancer harboring a KRA...
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : $5,800.0 million
October 08, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Krazati (adagrasib) is an investigational, highly selective and potent oral small molecule inhibitor of KRASG12C that is optimized to sustain target inhibition. It is indicated for the treatment KRASG12C-mutated NSCLC and currently is being investigated for colorectal cancer.
Lead Product(s): Adagrasib,Cetuximab
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable July 21, 2023

Details : Krazati (adagrasib) is an investigational, highly selective and potent oral small molecule inhibitor of KRASG12C that is optimized to sustain target inhibition. It is indicated for the treatment KRASG12C-mutated NSCLC and currently is being investigated ...
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
July 21, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Krazati (adagrasib) is an investigational, highly selective and potent oral small molecule inhibitor of KRASG12C that is optimized to sustain target inhibition. It is indicated for the treatment KRASG12C-mutated NSCLC and currently is being investigated for colorectal cancer.
Lead Product(s): Adagrasib
Therapeutic Area: Oncology Brand Name: Krazati
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable December 21, 2022

Mirati announces Adagrasib (Krazati™) Receives Breakthrough Therapy Designation from FDA for Pat...
Details : Krazati (adagrasib) is an investigational, highly selective and potent oral small molecule inhibitor of KRASG12C that is optimized to sustain target inhibition. It is indicated for the treatment KRASG12C-mutated NSCLC and currently is being investigated ...
Brand Name : Krazati
Molecule Type : Small molecule
Upfront Cash : Not Applicable
December 21, 2022

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?