
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. Adipate
2. Adipic Acid, Calcium Salt
3. Adipic Acid, Cu Salt
4. Adipic Acid, Cu(+2) Salt
5. Adipic Acid, Cu(+2) Salt (1:1)
6. Adipic Acid, Diammonium Salt
7. Adipic Acid, Disodium Salt
8. Adipic Acid, Mg Salt (1:1)
9. Adipic Acid, Monoammonium Salt
10. Adipic Acid, Nickel Salt
11. Adipic Acid, Potassium Salt
12. Adipic Acid, Sodium Salt
13. Ammonium Adipate
14. Diammonium Adipate
15. Hexanedioic Acid
16. Magnesium Adipate
17. Sodium Adipate
1. Hexanedioic Acid
2. 124-04-9
3. Adipinic Acid
4. 1,4-butanedicarboxylic Acid
5. Adilactetten
6. Acifloctin
7. Acinetten
8. 1,6-hexanedioic Acid
9. Molten Adipic Acid
10. Kyselina Adipova
11. Adipinsaure [german]
12. Acide Adipique [french]
13. Fema No. 2011
14. Kyselina Adipova [czech]
15. Hexanedioate
16. Adipinsaeure
17. Adipic-acid
18. Adipidic Acid
19. Adi-pure
20. Nsc 7622
21. Adipic Acid [nf]
22. Nsc-7622
23. Hexan-1,6-dicarboxylate
24. 76a0je0fkj
25. Ins No.355
26. 1,6-hexane-dioic Acid
27. E-355
28. Chebi:30832
29. Ins-355
30. Nsc7622
31. Adipic Acid (nf)
32. Ncgc00091345-01
33. E355
34. Hexane-1,6-dioic Acid
35. Dsstox_cid_1605
36. Dsstox_rid_76232
37. Dsstox_gsid_21605
38. Adipinsaure
39. Acide Adipique
40. Fema Number 2011
41. Cas-124-04-9
42. Ccris 812
43. Hsdb 188
44. Einecs 204-673-3
45. Mfcd00004420
46. Unii-76a0je0fkj
47. Brn 1209788
48. Adipinate
49. Molten Adipate
50. Ai3-03700
51. Hexane Dioic Acid
52. 1,6-hexanedioate
53. 0l1
54. Adipic Acid, 99%
55. Neopentyl Glycol Flake
56. Adipic Acid-[13c6]
57. C6-140-polymorph-i
58. C6-180-polymorph-i
59. C6-220-polymorph-i
60. C6-260-polymorph-i
61. C6-298-polymorph-i
62. 1, 6-hexanedioic Acid
63. Adipic Acid, >=99%
64. Adipic Acid [ii]
65. Adipic Acid [mi]
66. Wln: Qv4vq
67. Adipic Acid [fcc]
68. Bmse000424
69. Ec 204-673-3
70. Adipic Acid [fhfi]
71. Adipic Acid [hsdb]
72. Adipic Acid [inci]
73. Schembl4930
74. Chembl1157
75. Nciopen2_001004
76. Nciopen2_001222
77. Hooc-(ch2)4-cooh
78. Adipic Acid [mart.]
79. Adipic Acid, >=99.5%
80. 4-02-00-01956 (beilstein Handbook Reference)
81. Adipic Acid [usp-rs]
82. Adipic Acid [who-dd]
83. Bidd:er0342
84. Ins No. 355
85. Dtxsid7021605
86. Adipic Acid, Puriss., 99.8%
87. Pharmakon1600-01301012
88. Adipic Acid [ep Monograph]
89. Zinc1530348
90. Tox21_111118
91. Tox21_202161
92. Tox21_300344
93. Bbl011615
94. Lmfa01170048
95. Nsc760121
96. S3594
97. Stl163338
98. Akos000119031
99. Tox21_111118_1
100. Ccg-230896
101. Cs-w018238
102. Hy-w017522
103. Nsc-760121
104. Adipic Acid 100 Microg/ml In Methanol
105. Ncgc00091345-02
106. Ncgc00091345-03
107. Ncgc00091345-04
108. Ncgc00091345-05
109. Ncgc00254389-01
110. Ncgc00259710-01
111. Ac-10343
112. Bp-21150
113. Bp-30248
114. Hexanedioic Acid, Butanedicarboxylic Acid
115. A0161
116. Adipic Acid, Bioxtra, >=99.5% (hplc)
117. Adipic Acid, Saj Special Grade, >=99.5%
118. E 355
119. Ft-0606810
120. Adipic Acid, Vetec(tm) Reagent Grade, >=99%
121. C06104
122. D08839
123. D70505
124. Ab00988898-01
125. Ab00988898-03
126. Q357415
127. Sr-01000944270
128. J-005034
129. J-519542
130. Sr-01000944270-2
131. Z57127533
132. Adipic Acid, Certified Reference Material, Tracecert(r)
133. F0001-0377
134. Adipic Acid, European Pharmacopoeia (ep) Reference Standard
135. 1f1316f2-7a32-4339-8c2a-8caa84696c95
136. Adipic Acid, United States Pharmacopeia (usp) Reference Standard
137. Adipic Acid, Meets Analytical Specification Of Ph.eur., Bp, E 355, 99.6-101.0% (calc. To The Dried Substance)
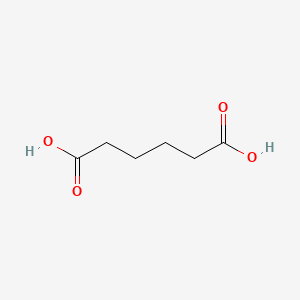
| Molecular Weight | 146.14 g/mol |
|---|---|
| Molecular Formula | C6H10O4 |
| XLogP3 | 0.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 5 |
| Exact Mass | 146.05790880 g/mol |
| Monoisotopic Mass | 146.05790880 g/mol |
| Topological Polar Surface Area | 74.6 Ų |
| Heavy Atom Count | 10 |
| Formal Charge | 0 |
| Complexity | 114 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Absorbed adipic acid is primarily excreted in the urine unchanged or in the breath of carbon dioxide.
Kennedy GL Jr; Drug Chem Toxicol 25 (2): 191-202 (2002)
In man following ingestion, adipic acid is only partially metabolized; the balance is eliminated unchanged in the urine.
Kennedy GL Jr; Drug Chem Toxicol 25 (2): 191-202 (2002)
Radioactive adipic acid was fed to fasted experimental rats and the metabolic products identified in the urine were urea, glutamic acid, lactic acid, beta-ketoadipic acid, and citric acid. The presence of beta-ketoadipic acid provided some evidence that adipic acid is metabolized by beta-oxidation in much the same fashion as fatty acids. Further evidence was provided by the appearance of succinate in the urine of rats fed radioactive (14)C-adipic acid and injected with malonic acid. The presence of radioactive acetyl-gamma-phenyl-alphaaminobutyric acid after feeding gamma-phenyl-alpha-aminobutyric acid and (14)C-labeled adipic acid provided very strong evidence that acetate is a metabolite of adipic acid. Radioactive glycogen was isolated following feeding of glucose and radioactive adipic acid.
PMID:14440268 Rusoff II et al; Toxicol Appl Pharmacol 2 (3): 316-30 (1960)
Adipic acid is metabolized via beta-oxidation to succinic and acetic acids, and subsequently to other normal intermediary metabolites.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V5 765

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
30
PharmaCompass offers a list of Adipic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Adipic Acid manufacturer or Adipic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Adipic Acid manufacturer or Adipic Acid supplier.
PharmaCompass also assists you with knowing the Adipic Acid API Price utilized in the formulation of products. Adipic Acid API Price is not always fixed or binding as the Adipic Acid Price is obtained through a variety of data sources. The Adipic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Adipic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Adipic Acid, including repackagers and relabelers. The FDA regulates Adipic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Adipic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Adipic Acid manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Adipic Acid supplier is an individual or a company that provides Adipic Acid active pharmaceutical ingredient (API) or Adipic Acid finished formulations upon request. The Adipic Acid suppliers may include Adipic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Adipic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Adipic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Adipic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Adipic Acid GMP manufacturer or Adipic Acid GMP API supplier for your needs.
A Adipic Acid CoA (Certificate of Analysis) is a formal document that attests to Adipic Acid's compliance with Adipic Acid specifications and serves as a tool for batch-level quality control.
Adipic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Adipic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Adipic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Adipic Acid EP), Adipic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Adipic Acid USP).
