
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
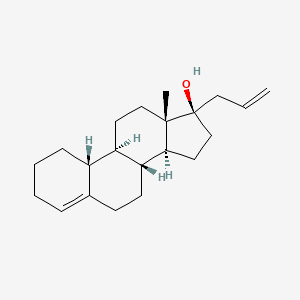

1. Allyloestrenol
2. Gestanin
3. Gestanon
4. Turinal
1. 432-60-0
2. Gestanin
3. Gestanon
4. Allyloestrenol
5. Gestanol
6. Orageston
7. Turinal
8. Organon
9. Gestanyn
10. Estrenol, Allyl-
11. 17-allylestr-4-en-17-beta-ol
12. 17alpha-allyl-4-estren-17beta-ol
13. 17alpha-allylestr-4-en-17beta-ol
14. Perselin
15. I47vb5dz8o
16. 17.alpha.-allylestr-4-en-17.beta.-ol
17. (8r,9s,10r,13s,14s,17r)-13-methyl-17-prop-2-enyl-2,3,6,7,8,9,10,11,12,14,15,16-dodecahydro-1h-cyclopenta[a]phenanthren-17-ol
18. Chebi:31189
19. Estr-4-en-17-ol, 17-(2-propenyl)-, (17b)-
20. Nsc-37723
21. 19-norpregn-4-ene-20-yn-17beta-ol
22. Allyloestrenolum
23. 17-alpha-allylestr-4-en-17-beta-ol
24. 17alpha-allylestrenol
25. Allilestrenolo [dcit]
26. Gestanon-r
27. Alilestrenol [inn-spanish]
28. Allylestrenolum [inn-latin]
29. Perselin (tn)
30. 17.alpha.-allylestrenol
31. 17alpha-allyl-4-destrene-17beta-ol
32. 17alpha-allyl-4-oestrene-17beta-ol
33. Allylestrenol (jan/inn)
34. 17-hydroxy-17-alpha-allyl-4-estrene
35. 17-alpha-allyl-4-oestrene-17-beta-ol
36. 17alpha-allyl-17beta-hydroxy-4-estrene
37. Alilestrenol
38. Allilestrenolo
39. Allylestrenolum
40. 17alpha-allyl-3-deoxy-19-nortestosterone
41. Allyl Estrenol
42. 17-alpha-allyl-17-beta-hydroxy-4-estrene
43. Ccris 9068
44. 17-alpha-allyl-3-deoxy-19-nortestosterone
45. 17-alpha-allylhydroxy-19-nor-4-androstene
46. 3-deoxy-17-alpha-allyl-19-nortestosterone
47. Ncgc00159497-02
48. 21-methylene-19-nor-17-alpha-preg-4-en-17-ol
49. Einecs 207-082-9
50. Estr-4-en-17beta-ol, 17-allyl-
51. 17-alpha-allyl-17-beta-hydroxy-delta(sup 4)-estren
52. 17alpha-allyl-19-nor-delta-4-androstene-17beta-ol
53. Nsc 37723
54. Estr-4-en-17-ol, 17-(2-propenyl)-, (17beta)-
55. Brn 3148038
56. 17.alpha.-allyl-4-estren-17.beta.-ol
57. 17.alpha.-allyl-4-destrene-17.beta.-ol
58. 17.alpha.-allyl-4-oestrene-17.beta.-ol
59. Estr-4-en-17-beta-ol, 17-allyl-
60. 17.alpha.-allyl-3-deoxy-19-nortestosterone
61. Dsstox_cid_2574
62. 17.alpha.-allyl-17.beta.-hydroxy-4-estrene
63. Allylestrenol [mi]
64. Unii-i47vb5dz8o
65. Allylestrenol [inn]
66. Allylestrenol [jan]
67. Dsstox_rid_76637
68. (17beta)-17-prop-2-en-1-ylestr-4-en-17-ol
69. Dsstox_gsid_22574
70. Allylestrenol [mart.]
71. Schembl329623
72. 19-nor-17-alpha-preg-4-en-17-ol, 21-methylene-
73. Allylestrenol [who-dd]
74. Allylestrenol [inn:ban:jan]
75. Chembl3185133
76. Dtxsid9022574
77. 17.alpha.-allyl-17-.beta.-hydroxy-.delta.(sup 4)-estren
78. Bcp10809
79. Nsc37723
80. Zinc4214767
81. Tox21_111718
82. Lmst02030125
83. Mfcd00198957
84. S5219
85. Akos015964938
86. Db01431
87. Ds-8255
88. Estr-4-en-17-ol, (17.beta.)-
89. Estr-4-en-17.beta.-ol, 17-allyl-
90. Cas-432-60-0
91. Hy-17375
92. 3-deketo-17alpha-allyl-19-nortestosterone
93. Cs-0006769
94. Wln: L E5 B666 Mutj E1 Fq F2u1
95. D01374
96. 4-06-00-04143 (beilstein Handbook Reference)
97. 432a600
98. Q531965
99. Sr-01000883956
100. Sr-01000883956-1
| Molecular Weight | 300.5 g/mol |
|---|---|
| Molecular Formula | C21H32O |
| XLogP3 | 5.3 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Exact Mass | 300.245315640 g/mol |
| Monoisotopic Mass | 300.245315640 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 492 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Allylestrenol was designed to be used for miscarriage prevention, prevention of premature labour and has been investigated for possible use in men for treatment for benign prostatic hyperplasia.
Allylestrenol is a progestogen structurally related to progesterone that has been given in threatened and recurrent miscarriage, and to prevent premature labour. However, with the exception of proven progesterone deficiency, such use is no longer recommended. In threatened miscarriage in progesterone-deficient women a suggested dose is 5 mg three times daily by mouth for 5 to 7 days.
Progestins
Compounds that interact with PROGESTERONE RECEPTORS in target tissues to bring about the effects similar to those of PROGESTERONE. Primary actions of progestins, including natural and synthetic steroids, are on the UTERUS and the MAMMARY GLAND in preparation for and in maintenance of PREGNANCY. (See all compounds classified as Progestins.)
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03D - Progestogens
G03DC - Estren derivatives
G03DC01 - Allylestrenol
Route of Elimination
The glucuronide and sulfate conjugates of pregnanediol and pregnanolone are excreted in the urine and bile. Progesterone metabolites which are excreted in the bile may undergo enterohepatic recycling or may be excreted in the feces. Progesterone metabolites are excreted mainly by the kidneys.
Allylestrenol is similar in structure and function to progesterone. Progesterone shares the pharmacological actions of the progestins. Progesterone binds to the progesterone and estrogen receptors. Target cells include the female reproductive tract, the mammary gland, the hypothalamus, and the pituitary. Once bound to the receptor, progestins like Progesterone will slow the frequency of release of gonadotropin releasing hormone (GnRH) from the hypothalamus and blunt the pre-ovulatory LH (luteinizing hormone) surge. In women who have adequate endogenous estrogen, progesterone transforms a proliferative endometrium into a secretory one. Progesterone is essential for the development of decidual tissue and is necessary to increase endometrial receptivity for implantation of an embryo. Once an embryo has been implanted, progesterone acts to maintain the pregnancy. Progesterone also stimulates the growth of mammary alveolar tissue and relaxes uterine smooth muscle. It has little estrogenic and androgenic activity.
Market Place

ABOUT THIS PAGE
80
PharmaCompass offers a list of Allylestrenol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Allylestrenol manufacturer or Allylestrenol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Allylestrenol manufacturer or Allylestrenol supplier.
A Allylestrenol manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Allylestrenol, including repackagers and relabelers. The FDA regulates Allylestrenol manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Allylestrenol API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Allylestrenol manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Allylestrenol supplier is an individual or a company that provides Allylestrenol active pharmaceutical ingredient (API) or Allylestrenol finished formulations upon request. The Allylestrenol suppliers may include Allylestrenol API manufacturers, exporters, distributors and traders.
click here to find a list of Allylestrenol suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Allylestrenol Drug Master File in Japan (Allylestrenol JDMF) empowers Allylestrenol API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Allylestrenol JDMF during the approval evaluation for pharmaceutical products. At the time of Allylestrenol JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Allylestrenol suppliers with JDMF on PharmaCompass.
A Allylestrenol written confirmation (Allylestrenol WC) is an official document issued by a regulatory agency to a Allylestrenol manufacturer, verifying that the manufacturing facility of a Allylestrenol active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Allylestrenol APIs or Allylestrenol finished pharmaceutical products to another nation, regulatory agencies frequently require a Allylestrenol WC (written confirmation) as part of the regulatory process.
click here to find a list of Allylestrenol suppliers with Written Confirmation (WC) on PharmaCompass.
Allylestrenol Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Allylestrenol GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Allylestrenol GMP manufacturer or Allylestrenol GMP API supplier for your needs.
A Allylestrenol CoA (Certificate of Analysis) is a formal document that attests to Allylestrenol's compliance with Allylestrenol specifications and serves as a tool for batch-level quality control.
Allylestrenol CoA mostly includes findings from lab analyses of a specific batch. For each Allylestrenol CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Allylestrenol may be tested according to a variety of international standards, such as European Pharmacopoeia (Allylestrenol EP), Allylestrenol JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Allylestrenol USP).

