
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
Canada

0
Australia

DRUG PRODUCT COMPOSITIONS
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
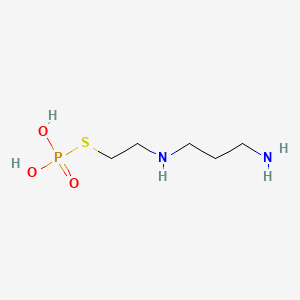

1. Amifostine Anhydrous
2. Amifostine Disodium Salt
3. Amifostine Monohydrate
4. Amifostine Monohydrochloride
5. Amifostine Trihydrate
6. Aminopropyl Aminoethylthiophosphate
7. Aminopropylaminoethylthiophosphate
8. Aminopropylaminoethylthiophosphoric Acid
9. Apaetp
10. Ethanethiol, 2-((3-aminopropyl)amino)-, Dihydrogen Phosphate (ester), Trihydrate
11. Ethiofos
12. Ethiofos Anhydrous
13. Ethyol
14. Gammaphos
15. Nsc 296961
16. Nsc-296961
17. Nsc296961
18. S-(n-(3-aminopropyl)-2-aminoethyl)thiophosphoric Acid
19. Wr 2721
20. Wr-2721
21. Wr2721
22. Ym 08310
23. Ym-08310
24. Ym08310
1. 20537-88-6
2. Ethiofos
3. Gammaphos
4. Ethyol
5. Sapep
6. Apaetp
7. Aminopropylaminoethyl Thiophosphate
8. Ethiofos Anhydrous
9. Amifostine Anhydrous
10. Ym-08310
11. Nsc-296961
12. Wr 2721
13. 2-(3-aminopropylamino)ethyl Thiophosphate
14. Au-95722
15. Nsc296961
16. Wr2721
17. Wr-2721
18. Amifostine (hydrate)
19. Nsc 296961
20. 2-(3-aminopropylamino)ethylsulfanylphosphonic Acid
21. S-(2-(3-aminopropylamino)ethyl) Phosphorothioate
22. Nsc-758236
23. Chebi:2636
24. Ila426l95o
25. S-(2-((3-aminopropyl)amino)ethyl) Dihydrogen Phosphorothioate
26. Ethanethiol, 2-((3-aminopropyl)amino)-, Dihydrogen Phosphate (ester)
27. Wr 2721c
28. Amifostina
29. Ncgc00015073-02
30. Ethanethiol, 2-[(3-aminopropyl)amino]-, 1-(dihydrogen Phosphate)
31. Dsstox_cid_2585
32. Dsstox_rid_76644
33. Dsstox_gsid_22585
34. ({2-[(3-aminopropyl)amino]ethyl}sulfanyl)phosphonic Acid
35. S-{2-[(3-aminopropyl)amino]ethyl} Dihydrogen Thiophosphate
36. Wr-2721 Trihydrate
37. 2-((3-aminopropyl)amino)-ethanethiol, Dihydrogen Phosphate Ester (9ci)
38. Amifostine Ethiofos
39. Amifostine [usan:inn:ban]
40. Cas-20537-88-6
41. S-[2-(3-aminopropylamino)ethyl] Phosphorothioate
42. Sr-01000075680
43. Brn 2088122
44. Unii-ila426l95o
45. Ym 08310
46. S-[2-[(3-aminopropyl)amino]ethyl] Dihydrogen Phosphorothioate
47. Ccris 9316
48. Hsdb 7560
49. Amifostine,(s)
50. S,2-(3-aminopropylamino)ethyl-phosphorothioic Acid
51. S-2-(3-aminopropylamino)ethyl Phosphorothioic Acid
52. Ethyol;wr2721
53. Spectrum_000332
54. S-omega-(3-aminopropylamino)ethyl Dihydrogen Phosphorothioate
55. Amifostine [mi]
56. Specplus_000647
57. 2-(3-aminopropyl)aminoethylphosphorothioate
58. Amifostine [inn]
59. Amifostine [jan]
60. 2-((3-aminopropyl)amino)-ethanethiol, Dihydrogen Phosphate Ester
61. Acide ((amino-3 Propylamino)-2 Ethyl)-s-phosphorothioique [french]
62. Spectrum5_001920
63. Amifostine [hsdb]
64. Lopac-a-5922
65. Ncimech_000622
66. S 8744
67. Amifostine [who-dd]
68. Chembl1006
69. Lopac0_000029
70. Schembl18464
71. Kbioss_000812
72. Mls000028473
73. Bidd:gt0059
74. Divk1c_006743
75. Dtxsid8022585
76. Kbio1_001687
77. Kbio2_000812
78. Kbio2_003380
79. Kbio2_005948
80. Acide ((amino-3 Propylamino)-2 Ethyl)-s-phosphorothioique
81. Hms2090i03
82. Hms3260e19
83. Hms3713c06
84. Pharmakon1600-01503081
85. Phosphorothioic Acid, S-(2-(3-aminopropylamino)ethyl) Ester
86. Bcp08957
87. Hy-b0639
88. Phosphorothioic Acid, S-[2-[(3-aminopropyl)amino]ethyl] Ester
89. Tox21_110077
90. Tox21_500029
91. Ccg-35734
92. Mfcd00233058
93. Nsc758236
94. Zinc21992285
95. Akos015895196
96. Tox21_110077_1
97. Ac-1126
98. Db01143
99. Lp00029
100. Sdccgsbi-0050018.p005
101. Smp2_000335
102. Ncgc00015073-01
103. Ncgc00015073-03
104. Ncgc00015073-04
105. Ncgc00015073-05
106. Ncgc00015073-15
107. Ncgc00093549-01
108. Ncgc00093549-02
109. Ncgc00260714-01
110. As-13020
111. Ethanethiol, Dihydrogen Phosphate (ester)
112. Nci60_002485
113. Smr000058413
114. Ethanethiol, Dihydrogen Phosphate- (ester)
115. Sbi-0050018.p003
116. 2-(3-aminopropyl)aminoethyl Phosphorothioate
117. Eu-0100029
118. Ft-0622272
119. 37a886
120. A 5922
121. C06819
122. Ab00053311-04
123. Ab00053311-05
124. Ab00053311_06
125. A814662
126. Q251698
127. {s-[2-(3-aminopropylamino)ethyl]} Phosphorothioate
128. J-013390
129. J-519582
130. Sr-01000075680-1
131. Sr-01000075680-3
132. Sr-01000075680-5
133. 2-(3-aminopropyl)aminoethyl Phosphorothioate; Wr2721
134. Brd-k73947551-001-01-0
135. 2-[(3-aminopropyl)amino]ethanethiol Dihydrogen Phosphate
136. 2-(3-aminopropylamino)ethylsulfanylphosphonic Acid,trihydrate
137. 2-(3-aminopropylamino)ethylsulfanylphosphonic Acid;amifostine
138. Phosphorothioic Acid S-(2-((3-aminopropyl)amino)ethyl) Ester
139. Phosphorothioic Acid, S-[2-[(3-aminopropyl)amino]ethyl]ester
140. S-2-(3-aminopropylamino)ethyl O,o-dihydrogen Phosphorothioate
141. {s-[2-[(3-aminopropyl)amino]ethyl]} Dihydrogen Phosphorothioate
142. 2-((3-aminopropyl)amino)ethanethiol 1-(dihydrogen Phosphate)
143. Ethanethiol, {s-[(3-aminopropyl)amino]-,} Dihydrogen Phosphate- (ester)
| Molecular Weight | 214.23 g/mol |
|---|---|
| Molecular Formula | C5H15N2O3PS |
| XLogP3 | -4.5 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 214.05410052 g/mol |
| Monoisotopic Mass | 214.05410052 g/mol |
| Topological Polar Surface Area | 121 Ų |
| Heavy Atom Count | 12 |
| Formal Charge | 0 |
| Complexity | 152 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Amifostine |
| PubMed Health | Amifostine (Injection) |
| Drug Classes | Cytoprotective Agent |
| Drug Label | Amifostine for Injection is an organic thiophosphate cytoprotective agent known chemically as 2-[(3-aminopropyl)amino]ethanethiol dihydrogen phosphate (ester) and has the following structural formula: H2N(CH2)3NH(CH2)2S-PO3H2Amifostine is a white cry... |
| Active Ingredient | Amifostine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 500mg/vial |
| Market Status | Prescription |
| Company | Sun Pharma Global |
| 2 of 4 | |
|---|---|
| Drug Name | Ethyol |
| PubMed Health | Amifostine (Injection) |
| Drug Classes | Cytoprotective Agent |
| Active Ingredient | Amifostine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 500mg/vial |
| Market Status | Prescription |
| Company | Medimmune |
| 3 of 4 | |
|---|---|
| Drug Name | Amifostine |
| PubMed Health | Amifostine (Injection) |
| Drug Classes | Cytoprotective Agent |
| Drug Label | Amifostine for Injection is an organic thiophosphate cytoprotective agent known chemically as 2-[(3-aminopropyl)amino]ethanethiol dihydrogen phosphate (ester) and has the following structural formula: H2N(CH2)3NH(CH2)2S-PO3H2Amifostine is a white cry... |
| Active Ingredient | Amifostine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 500mg/vial |
| Market Status | Prescription |
| Company | Sun Pharma Global |
| 4 of 4 | |
|---|---|
| Drug Name | Ethyol |
| PubMed Health | Amifostine (Injection) |
| Drug Classes | Cytoprotective Agent |
| Active Ingredient | Amifostine |
| Dosage Form | Injectable |
| Route | Injection |
| Strength | 500mg/vial |
| Market Status | Prescription |
| Company | Medimmune |
Radiation-protective agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Amifostine is indicated to reduce the cumulative renal toxicity associated with repeated administration of cisplatin in patients with advanced ovarian cancer. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 89
Amifostine is indicated to reduce the cumulative renal toxicity associated with repeated administration of cisplatin in patients with advanced solid tumor of non-germ cell origin. /NOT included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 89
Amifostine is indicated to reduce the incidence of moderate to severe xerostomia in patients undergoing post-operative radiation treatment for head and neck cancer, where radiation port includes a substantial portion of the parotid gland. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 89
For more Therapeutic Uses (Complete) data for AMIFOSTINE (8 total), please visit the HSDB record page.
Although hypocalcemia occurs only rarely during cytoprotective therapy with amifostine, serum calcium concentrations should be monitored in patients at risk of hypocalcemia, such as those with nephrotic syndrome or those receiving multiple doses of amifostine, and calcium supplementation initiated as necessary.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
Because nausea and/or vomiting occur frequently during amifostine therapy and may be severe, effective antiemetic therapy (e.g., an IV corticosteroid such as dexamethasone and a serotonin 5-HT3 receptor antagonist) should be administered prior to and in conjunction with amifostine therapy. Additional antiemetics may be required depending on the emetogenic potential of concomitantly administered chemotherapy or radiation therapy. In addition, the patient's fluid balance should be monitored carefully in those receiving highly emetogenic chemotherapy.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
Patients also should be closely monitored during and after IV administration of amifostine for possible allergic manifestations, including anaphylactic reactions that have rarely been associated with cardiac arrest. If acute hypersensitivity reactions occur, the infusion should be immediately and permanently discontinued. The manufacturer states that epinephrine and other appropriate measures should be available for immediate treatment of such events.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
During infusions of less than 5 minutes' duration, blood pressure should be monitored at least before and immediately after completion of the infusion, and thereafter if needed. If hypotension requiring interruption of the amifostine infusion occurs, the patient should be placed in the Trendelenburg position and an iv infusion of 0.9% sodium chloride initiated in a separate line.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
For more Drug Warnings (Complete) data for AMIFOSTINE (9 total), please visit the HSDB record page.
For reduction in the cumulative renal toxicity in patients with ovarian cancer (using cisplatin) and moderate to severe xerostomia in patients undergoing post-operative radiation treatment for head and neck cancer.
FDA Label
Amifostine is an organic thiophosphate cytoprotective agent indicated to reduce the cumulative renal toxicity associated with repeated administration of cisplatin in patients with advanced ovarian cancer or non-small cell lung cancer and also to reduce the incidence of moderate to severe xerostomia in patients undergoing post-operative radiation treatment for head and neck cancer. Amifostine is a prodrug that is dephosphorylated by alkaline phosphatase in tissues to a pharmacologically active free thiol metabolite, believed to be responsible for the reduction of the cumulative renal toxicity of cisplatin and for the reduction of the toxic effects of radiation on normal oral tissues. Healthy cells are preferentially protected because amifostine and metabolites are present in healthy cells at 100-fold greater concentrations than in tumour cells.
Radiation-Protective Agents
Drugs used to protect against ionizing radiation. They are usually of interest for use in radiation therapy but have been considered for other purposes, e.g. military. (See all compounds classified as Radiation-Protective Agents.)
V - Various
V03 - All other therapeutic products
V03A - All other therapeutic products
V03AF - Detoxifying agents for antineoplastic treatment
V03AF05 - Amifostine
Route of Elimination
After a 10-second bolus dose of 150 mg/m2 of ETHYOL, renal excretion of the parent drug and its two metabolites was low during the hour following drug administration, averaging 0.69%, 2.64% and 2.22% of the administered dose for the parent, thiol and disulfide, respectively.
Measurable concentrations of the active free thiol metabolite have been found in bone marrow cells 5 to 8 minutes after intravenous administration.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
It is not known whether amifostine or its metabolites are distributed into breast milk.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
Elimination /is/ primarily via rapid metabolism and uptake into tissues.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
... Studies in rats using a single dose of amifostine showed that /with/ SC administration, there is no evidence of drug accumulation in either normal or tumor tissue, with tumor WR-1065 levels peaking just above the limits of quantitation during treatment. ...
PMID:12577236 Cassatt D, Fazenbaker C et al; Semin Oncol 29 (6 Suppl 19): 2-8 (2002)
The pharmacokinetics of the cytoprotective agent amifostine (EthyolR; WR 2721) and its main metabolites (WR 1065 and the disulphides) were studied in patients participating in two phase I trials concerning carboplatin or cisplatin in combination with amifostine. Patients were treated with a single dose or three doses of amifostine (740 or 910 mg/sq m). The single or first dose was given as a 15 min iv infusion just before administration of the chemotherapeutic agent. The additional two infusions were administered 2 and 4 hr thereafter. Amifostine was rapidly cleared from the plasma, due to, at least in part, the fast conversion into WR 1065. A biphasic decrease with a final half-life of 0.8 hr was observed. The active metabolite WR 1065 was cleared from the plasma with a final half-life of 7.3 +/- 3.6 hr. The short initial half-life of WR 1065 can be explained by its fast uptake in tissues and the formation of disulphides. The disulphides were cleared with a final half-life of 8.4-13.4 hr and were detectable for at least 24 hr after treatment. They may serve as an exchangeable pool of WR 1065. The amifostine peak values at the end of each 15 min infusion did not accumulate in the multiple dosing schedule. For WR 1065 a trend towards an increase in the peak levels was observed [C1,max: 47.5 +/- 11.9 uM, C2,max: 79.0 +/- 13.2 uM, C3,max: 84.8 +/- 15.1 uM, (n = 6)], whereas a trend towards a small decrease was observed for the peak levels of the disulphides [C1,max: 184.2 +/- 12.6 uM, C2,max: 175.0 +/- 23.7 uM, C3,max: 166.0 +/- 17.2 uM, (n = 6)]. This latter finding might suggest a saturation of the disulphide formation or a change in the uptake or elimination of WR 1065, which would result in higher WR 1065 levels in plasma and tissues, after multiple doses of amifostine.
PMID:9337685 Korst A, Eeltink C et al; Eur J Cancer 33 (9): 1425-9 (1997)
Amifostine is rapidly dephosphorylated by alkaline phosphatase in tissues primarily to the active free thiol metabolite and, subsequently, to a less active disulfide metabolite.
Amifostine is dephosphorylated by alkaline phosphatase in tissues primarily to the active free thiol metabolite and, subsequently, to a less active disulfide metabolite.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
Within 1 hour after infusion of 740 to 910 mg per square meter of body surface area over 15 minutes or rapid intravenous injection of 150 mg per square meter body surface area over 10 seconds, urinary recovery of unchanged amifostine, of the disulfide metabolite, and the thiol metabolite accounts for only 0.69%, 2.22%, and 2.64%, respectively, of the dose.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
This study investigated the metabolism of the radio- and chemoprotector compound, WR-2721 [amifostine; s-2-(3- aminopropylamino)ethylphosphorothioate], in the Balb/c mouse. ... It is known that /radiation/ protection requires conversion of the parent drug to its free thiol metabolite, WR-1065, in cultured cells. Because it is possible that metabolites of WR-1065 could be involved in protection and because thiols are metabolically very reactive molecules, we investigated the metabolism of WR-2721 using electrochemical detection-HPLC methods. The following are the major findings in this study: 1) WR-2721 drug was rapidly cleared from the bloodstream. Blood concentration of the parent drug decreased 10-fold 30 min after administration from the maximal observed value at 5 min 2) WR-1065 rapidly appeared in the perchloric acid (PCA)-soluble fraction of normal solid tissues. The highest WR-1065 concentrations in liver and kidney were 965 and 2195 mumol/kg, respectively, 10 min after parent drug administration, whereas for heart and small intestine the highest values were 739 and 410 mumol/kg at 30 min. 3) WR-1065 accumulated in the PCA-soluble fraction of two experimental tumors at a lower rate than for the other tissues.
PMID:7895607 Shaw L, Bonner H et al; Drug Metab Dispos 22 (6): 895-902 (1994)
8 minutes
Approximately 8 minutes; less than 10% of amifostine remains in the plasma 6 minutes after drug administration.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 90
The pharmacokinetics of the cytoprotective agent amifostine (EthyolR; WR 2721) and its main metabolites (WR 1065 and the disulphides) were studied in patients participating in two phase I trials concerning carboplatin or cisplatin in combination with amifostine. Patients were treated with a single dose or three doses of amifostine (740 or 910 mg/sq m). The single or first dose was given as a 15 min iv infusion just before administration of the chemotherapeutic agent. The additional two infusions were administered 2 and 4 hr thereafter. Amifostine was rapidly cleared from the plasma, due to, at least in part, the fast conversion into WR 1065. A biphasic decrease with a final half-life of 0.8 hr was observed. The active metabolite WR 1065 was cleared from the plasma with a final half-life of 7.3 +/- 3.6 hr. The short initial half-life of WR 1065 can be explained by its fast uptake in tissues and the formation of disulphides. The disulphides were cleared with a final half-life of 8.4-13.4 hr and were detectable for at least 24 hr after treatment. They may serve as an exchangeable pool of WR 1065. ...
PMID:9337685 Korst A, Eeltink C et al; Eur J Cancer 33 (9): 1425-9 (1997)
The thiol metabolite is responsible for most of the cytoprotective and radioprotective properties of amifostine. It is readily taken up by cells where it binds to and detoxifies reactive metabolites of platinum and alkylating agents as well as scavenges free radicals. Other possible effects include inhibition of apoptosis, alteration of gene expression and modification of enzyme activity.
Amifostine is a prodrug that is dephosphorylated by alkaline phosphatase to an active free sulfhydryl (thiol) metabolite (WR-1065). The pharmacologically active free sulfhydryl is believed to bind to and detoxify cytotoxic platinum-containing metabolites of cisplatin and scavenge free radicals induced by the drug. Cytoprotection against cisplatin-induced toxicity appears to result from prevention and/or, to a lesser extent, reversal of DNA platination by the drug (cisplatin-DNA adducts). Healthy cells appear to be protected preferentially because of the increased cellular uptake of amifostine and more rapid generation of the active free sulfhydryl metabolite in these cells compared with malignant cells. Because healthy cells have better perfusion, higher capillary alkaline phosphatase activity, and higher pH than malignant cells, and actively concentrate amifostine while malignant cells absorb the drug passively, there is increased cellular uptake of amifostine and more rapid generation of the free active sulfhydryl; therefore, compared with malignant cells, healthy cells appear to be selectively protected by amifostine against cisplatin-induced cytotoxicity.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
Similar to the drug's chemoprotection, amifostine's radioprotective effect may occur preferentially in healthy rather than malignant cells secondary to the drug's increased cellular uptake and conversion to the active sulfhydryl. Amifostine's radioprotectant effect appears to be mediated at least in part by removal of oxygen from tissues. Sulfhydryl compounds such as amifostine also appear to protect cells from the cytotoxic effects of radiation by scavenging hydroxyl radicals and repairing radiation-induced DNA radicals through donation of hydrogen.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
Amifostine can lower serum calcium concentrations, including total, ionized, and ultrafilterable calcium, but clinically important hypocalcemia occurs rarely during cytoprotective therapy with the drug. The hypocalcemic effect of amifostine appears to result from inhibition of parathyroid hormone secretion and bone resorption and from facilitation of urinary calcium excretion.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 3782
Amifostine (WR-2721) is an inorganic thiophosphate-cytoprotective agent developed to selectively protect normal tissues against the toxicity of chemotherapy and radiation. We have previously shown that amifostine protects both chicken embryo chorioallantoic membrane (CAM) vessels and cells from the effects of X-rays. In the present work, we studied the effect of amifostine on angiogenesis in vivo, using the CAM model. Amifostine decreased the number of CAM vessels in a dose-dependent manner, without being toxic for the tissue. It also decreased the mRNA levels of both vascular endothelial growth factor (VEGF) isoforms VEGF(165) and VEGF(190), 6 and up to 48 hr after its application onto the CAM. Similarly, it decreased the mRNA levels of inducible nitric-oxide synthase, 24 and 48 hr after drug application. Furthermore, amifostine decreased the deposited amounts of laminin and collagen I 24 hr after its application, without affecting the expression of the corresponding genes. The protein amounts and activity of matrix metalloproteinase-2 were not affected, whereas the expression of the corresponding gene was decreased up to 48 hr after drug application. Finally, the activity of plasmin was increased 6 hr after amifostine application and remained increased at later time points. These findings suggest that amifostine alters the expression of several molecules implicated in the angiogenesis process and affects the composition of the extracellular matrix in a way that leads to inhibition of angiogenesis. Such an antiangiogenic action of amifostine, together with its radioprotective effects, further supports its use in combination with radiotherapy for increased therapeutic efficacy.
PMID:12538828 Giannopoulou E, Katsoris P et al; J Pharmacol Exp Ther 304 (2): 729-37 (2003)
For more Mechanism of Action (Complete) data for AMIFOSTINE (7 total), please visit the HSDB record page.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
38
PharmaCompass offers a list of Amifostine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Amifostine manufacturer or Amifostine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Amifostine manufacturer or Amifostine supplier.
PharmaCompass also assists you with knowing the Amifostine API Price utilized in the formulation of products. Amifostine API Price is not always fixed or binding as the Amifostine Price is obtained through a variety of data sources. The Amifostine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Amifostine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Amifostine, including repackagers and relabelers. The FDA regulates Amifostine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Amifostine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Amifostine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Amifostine supplier is an individual or a company that provides Amifostine active pharmaceutical ingredient (API) or Amifostine finished formulations upon request. The Amifostine suppliers may include Amifostine API manufacturers, exporters, distributors and traders.
click here to find a list of Amifostine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Amifostine DMF (Drug Master File) is a document detailing the whole manufacturing process of Amifostine active pharmaceutical ingredient (API) in detail. Different forms of Amifostine DMFs exist exist since differing nations have different regulations, such as Amifostine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Amifostine DMF submitted to regulatory agencies in the US is known as a USDMF. Amifostine USDMF includes data on Amifostine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Amifostine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Amifostine suppliers with USDMF on PharmaCompass.
A Amifostine written confirmation (Amifostine WC) is an official document issued by a regulatory agency to a Amifostine manufacturer, verifying that the manufacturing facility of a Amifostine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Amifostine APIs or Amifostine finished pharmaceutical products to another nation, regulatory agencies frequently require a Amifostine WC (written confirmation) as part of the regulatory process.
click here to find a list of Amifostine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Amifostine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Amifostine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Amifostine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Amifostine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Amifostine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Amifostine suppliers with NDC on PharmaCompass.
Amifostine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Amifostine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Amifostine GMP manufacturer or Amifostine GMP API supplier for your needs.
A Amifostine CoA (Certificate of Analysis) is a formal document that attests to Amifostine's compliance with Amifostine specifications and serves as a tool for batch-level quality control.
Amifostine CoA mostly includes findings from lab analyses of a specific batch. For each Amifostine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Amifostine may be tested according to a variety of international standards, such as European Pharmacopoeia (Amifostine EP), Amifostine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Amifostine USP).
