
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
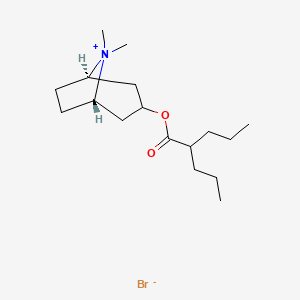

1. Anisotropine Methylbromide, (endo)-isomer
2. Octatropine
3. Octotropine Methylbromide
4. Valpin
1. Octatropine Methylbromide
2. Valpin
3. Valpin 50
4. 80-50-2
5. Methyloctatropine Bromide
6. Endovalpin
7. Octotropine Methylbromide
8. Anisotropine Methyl Bromide
9. Mls000028664
10. Mls001146935
11. 62m960dhil
12. Octatropini Methylbromidum
13. Methyloctatropine Bromide (jan)
14. Octatropine Methylbromide (inn)
15. Octatropine Methylbromide [inn]
16. Lytispasm
17. Methylbromure D'octatropine
18. Metilbromuro De Octatropina
19. Anisotropine Methylbromide (usan)
20. 8-methyltropinium Bromide 2-propylpentanoate
21. Smr000058851
22. Dsstox_cid_2612
23. Anisotropine (methobromide)
24. 8-azoniabicyclo(3.2.1)octane, 8,8-dimethyl-3-((1-oxo-2-propylpentyl)oxy)-, Bromide, Endo-
25. Dsstox_rid_76659
26. Dsstox_gsid_22612
27. Methyloctatropine Bromide [jan]
28. Cas-80-50-2
29. Anisotropine Methylbromide [usan]
30. Octatropone Bromide
31. Valpin (tn)
32. Valpin 50 (tn)
33. Ncgc00021394-03
34. Ncgc00021394-04
35. 2-propylpentanoyltropinium Methylbromide
36. Unii-62m960dhil
37. Methylbromure D'octatropine [inn-french]
38. Metilbromuro De Octatropina [inn-spanish]
39. Hsdb 3010
40. Einecs 201-285-6
41. Octatropini Methylbromidum [inn-latin]
42. Anisotropine Methylbromide [usan:inn:jan]
43. Opera_id_1463
44. 1alphah,5alphah-tropanium, 3alpha-hydroxy-8-methyl-, Bromide, 2-propylvalerate
45. 3-alpha-hydroxy-8-methyl-1-alpha-h,5-alpha-h-tropanium Bromide 2-propylvalerate
46. 3alpha-hydroxy-8-methyl-1alphah,5alphah-tropanium Bromide 2-propylvalerate
47. Chembl1578
48. Mls006010788
49. Schembl250226
50. Schembl1650394
51. Dtxsid8022612
52. Regid_for_cid_657201
53. Hms2230n16
54. Hy-b1475
55. Tox21_110871
56. Tox21_113475
57. Akos015916559
58. Anisotropine Methylbromide [mi]
59. Ccg-268189
60. (3-endo)-8,8-dimethyl-3-[(2-propylpentanoyl)oxy]-8-azoniabicyclo[3.2.1]octane Bromide
61. Anisotropine Methylbromide [hsdb]
62. Octatropine Methylbromide [mart.]
63. Anisotropine Methylbromide [vandf]
64. Octatropine Methylbromide [who-dd]
65. Smr004701473
66. Mls-0002939.p022
67. Cs-0013173
68. Anisotropine Methylbromide [orange Book]
69. C06830
70. D00232
71. Anisotropine Methylbromide(octatropine Methylbromide)
72. Q7076760
73. (1r,3r,5s)-8,8-dimethyl-3-(2-propylpentanoyloxy)-8-azoniabicyclo[3.2.1]octane Bromide
74. (3s)-8,8-dimethyl-3-[(2-propylpentanoyl)oxy]-8-azabicyclo[3.2.1]octan-8-ium Bromide
75. [(1s,5r)-8,8-dimethyl-8-azoniabicyclo[3.2.1]octan-3-yl] 2-propylpentanoate;bromide
76. 1-alpha-h,5-alpha-h-tropanium, 3-alpha-hydroxy-8-methyl-, Bromide, 2-propylvalerate
77. 3.alpha.-hydroxy-8-methyl-1.alpha.h,5.alpha.h-tropanium Bromide 2-propylvalerate
| Molecular Weight | 362.3 g/mol |
|---|---|
| Molecular Formula | C17H32BrNO2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 7 |
| Exact Mass | 361.16164 g/mol |
| Monoisotopic Mass | 361.16164 g/mol |
| Topological Polar Surface Area | 26.3 Ų |
| Heavy Atom Count | 21 |
| Formal Charge | 0 |
| Complexity | 318 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Parasympatholytics
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
...ANTIMUSCARINIC DRUG. IT APPEARS TO BE RELATIVELY SELECTIVE FOR GI TRACT... IT IS USED IN TREATMENT OF MUCOUS COLITIS & IRRITABLE COLON, SPASTIC COLITIS, SPLENIC FLEXURE SYNDROME, BILIARY DYSKINESIA, CHOLELITHIASIS, PYLOROSPASM, GASTRITIS, DUODENITIS, ENTEROCOLITIS, & PEPTIC ULCER.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 849
VALPIN 50-PB INDICATED FOR USE AS ADJUNCTIVE THERAPY IN PEPTIC ULCER; IRRITABLE BOWEL SYNDROME (IRRITABLE COLON, SPASTIC COLON, MUCOUS COLITIS, ACUTE ENTEROCOLITIS, & FUNCTIONAL GI DISORDERS).
PDR; 33RD EDITION, 1979, MEDICAL ECONOMICS CO, LITTON INDUST, ORADELL, NJ 07649, 871-872
VALPIN 50-PB EFFECTIVELY PRODUCES VISCERAL SMOOTH MUSCLE RELAXATION. CONTROLS SMOOTH MUSCLE SPASM SEEN IN MOTILITY DISORDERS OF GI TRACT, SUCH AS SPASTIC COLON & FUNCTIONAL GI DISORDERS.
PDR; 33RD EDITION, 1979. MEDICAL ECONOMICS CO, LITTON INDUST, ORADELL, NJ 07649, 871-872
For more Therapeutic Uses (Complete) data for ANISOTROPINE METHYLBROMIDE (11 total), please visit the HSDB record page.
ALTHOUGH INCIDENCE & INTENSITY OF SIDE EFFECTS IS LESS THAN THOSE OF METHANTHELINE BROMIDE, THEY ARE QUALITATIVELY SAME.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 849
...NO DRUG OF THIS CLASS HAS YET BEEN SHOWN TO PRODUCE ADEQUATE CONTROL OF GASTRIC SECRETION OR GI MOTILITY @ DOSES THAT ARE DEVOID OF SIGNIFICANT SIDE EFFECTS DUE TO MUSCARINIC BLOCKADE @ OTHER SITES. /QUATERNARY AMMONIUM CMPD/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 158
...USED WITH GREAT CAUTION, & IN SOME CASES MAY BE CONTRAINDICATED, IN PATIENTS WITH REFLUX ESOPHAGITIS, ACHALASIA, PARTIAL GASTRIC OUTLET OBSTRUCTION, PARALYTIC ILEUS, INTESTINAL OBSTRUCTION, NARROW ANGLE GLAUCOMA, & PROSTATISM. /ANTISPASMODICS/
Miller, R. R., and D. J. Greenblatt. Handbook of Drug Therapy. New York: Elsevier North Holland, 1979., p. 1041
ANTICHOLINERGIC ANTISPASMODICS SHOULD BE USED WITH CAUTION IN PATIENTS WITH PROSTATIC HYPERTROPHY, PYLORIC OBSTRUCTION, OBSTRUCTION OF BLADDER NECK, CONGESTIVE HEART FAILURE WITH TACHYCARDIA, & ACHALASIA (CARDIOSPASM). /ANTICHOLINERGIC AGENTS/
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 935
For more Drug Warnings (Complete) data for ANISOTROPINE METHYLBROMIDE (11 total), please visit the HSDB record page.
Muscarinic Antagonists
Drugs that bind to but do not activate MUSCARINIC RECEPTORS, thereby blocking the actions of endogenous ACETYLCHOLINE or exogenous agonists. Muscarinic antagonists have widespread effects including actions on the iris and ciliary muscle of the eye, the heart and blood vessels, secretions of the respiratory tract, GI system, and salivary glands, GI motility, urinary bladder tone, and the central nervous system. (See all compounds classified as Muscarinic Antagonists.)
...POORLY & UNRELIABLY ABSORBED AFTER ORAL ADMIN... PENETRATION OF CONJUNCTIVA IS ALSO POOR, SO THAT...CMPD ARE OF LITTLE VALUE IN OPHTHALMOLOGY. CENTRAL EFFECTS ARE GENERALLY LACKING, BECAUSE THESE AGENTS DO NOT READILY PASS BLOOD-BRAIN BARRIER. ...LITTLE...KNOWN OF FATE & EXCRETION... /QUATERNARY AMMONIUM CMPD/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 158
WHOLE-BODY AUTORADIOGRAPHY OF MICE GIVEN SC, ORAL, OR IP DOSES OF ANTISPASMODIC, [(14)C]ANISOTROPINE METHBROMIDE SHOWED THAT PARENTERAL DOSES WERE DISTRIBUTED MAINLY IN EXCRETORY ORGANS (KIDNEYS, LIVER, SALIVARY GLANDS, & INTESTINES).
The Chemical Society. Foreign Compound Metabolism in Mammals. Volume 2: A Review of the Literature Published Between 1970 and 1971. London: The Chemical Society, 1972., p. 123
SOME (14)C /ANISOTROPINE METHBROMIDE/ WAS...PRESENT IN STOMACH MUCOSA, PANCREAS, OVIDUCTS, UTERUS, & SPERMATIC DUCTS /AFTER ADMIN TO MICE/. CATION WOULD NOT BE EXPECTED TO DIFFUSE EASILY ACROSS BIOLOGICAL MEMBRANES...DID NOT APPEAR READILY TO CROSS BLOOD-BRAIN OR PLACENTAL BARRIERS, OR GASTROINTESTINAL EPITHELIUM.
The Chemical Society. Foreign Compound Metabolism in Mammals. Volume 2: A Review of the Literature Published Between 1970 and 1971. London: The Chemical Society, 1972., p. 123
THERE IS CLINICAL IMPRESSION THAT QUATERNARY AMMONIUM CMPD HAVE RELATIVELY GREATER EFFECT ON GASTROINTESTINAL ACTIVITY & THAT DOSES NECESSARY TO TREAT GASTROINTESTINAL DISORDERS ARE, CONSEQUENTLY...MORE READILY TOLERATED; THIS HAS BEEN ATTRIBUTED TO ADDITIONAL ELEMENT OF GANGLIONIC BLOCK. /QUATERNARY AMMONIUM CMPD/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 158-9
RATIO OF GANGLIONIC BLOCKING TO ANTIMUSCARINIC ACTIVITY IS GREATER FOR CMPD WITH QUATERNARY AMMONIUM STRUCTURE BECAUSE OF THEIR GREATER POTENCY AT NICOTINIC RECEPTORS; SOME OF SIDE EFFECTS SEEN AFTER HIGH DOSES ARE DUE TO GANGLIONIC BLOCKADE. /QUATERNARY AMMONIUM CMPD/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 159
ANTISPASMODICS ARE COMPETITIVE ANTAGONISTS OF ACETYLCHOLINE, WHICH IS RELEASED @ ENDINGS OF PARASYMPATHETIC NERVES SUPPLYING GI TRACT. ...THESE DRUGS CAUSE MEAN REDUCTION OF BASAL ACID OUTPUT OF ABOUT 50%. THEY ALSO REDUCE MAXIMAL ACID OUTPUT BY 15 TO 50%. /ANTISPASMODICS/
Miller, R. R., and D. J. Greenblatt. Handbook of Drug Therapy. New York: Elsevier North Holland, 1979., p. 1039
ABOUT THIS PAGE
16
PharmaCompass offers a list of Anisotropine methobromide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Anisotropine methobromide manufacturer or Anisotropine methobromide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Anisotropine methobromide manufacturer or Anisotropine methobromide supplier.
PharmaCompass also assists you with knowing the Anisotropine methobromide API Price utilized in the formulation of products. Anisotropine methobromide API Price is not always fixed or binding as the Anisotropine methobromide Price is obtained through a variety of data sources. The Anisotropine methobromide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Anisotropine methobromide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Anisotropine methobromide, including repackagers and relabelers. The FDA regulates Anisotropine methobromide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Anisotropine methobromide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Anisotropine methobromide supplier is an individual or a company that provides Anisotropine methobromide active pharmaceutical ingredient (API) or Anisotropine methobromide finished formulations upon request. The Anisotropine methobromide suppliers may include Anisotropine methobromide API manufacturers, exporters, distributors and traders.
click here to find a list of Anisotropine methobromide suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Anisotropine methobromide DMF (Drug Master File) is a document detailing the whole manufacturing process of Anisotropine methobromide active pharmaceutical ingredient (API) in detail. Different forms of Anisotropine methobromide DMFs exist exist since differing nations have different regulations, such as Anisotropine methobromide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Anisotropine methobromide DMF submitted to regulatory agencies in the US is known as a USDMF. Anisotropine methobromide USDMF includes data on Anisotropine methobromide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Anisotropine methobromide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Anisotropine methobromide suppliers with USDMF on PharmaCompass.
Anisotropine methobromide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Anisotropine methobromide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Anisotropine methobromide GMP manufacturer or Anisotropine methobromide GMP API supplier for your needs.
A Anisotropine methobromide CoA (Certificate of Analysis) is a formal document that attests to Anisotropine methobromide's compliance with Anisotropine methobromide specifications and serves as a tool for batch-level quality control.
Anisotropine methobromide CoA mostly includes findings from lab analyses of a specific batch. For each Anisotropine methobromide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Anisotropine methobromide may be tested according to a variety of international standards, such as European Pharmacopoeia (Anisotropine methobromide EP), Anisotropine methobromide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Anisotropine methobromide USP).
