
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
API
0
FDF
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
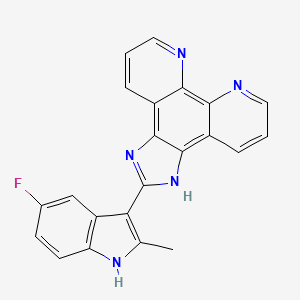

1. Apto-253
1. Apto-253
2. 916151-99-0
3. Lor-253
4. Lt-253
5. Lt-253(former Code)
6. Lor-253(former Code)
7. Apto253 Free Base
8. Wb59mrw00u
9. 916151-99-0 (free Base)
10. 1h-imidazo(4,5-f)(1,10)phenanthroline, 2-(5-fluoro-2-methyl-1h-indol-3-yl)-
11. 2-(5-fluoro-2-methyl-1h-indol-3-yl)-1h-imidazo(4,5-f)(1,10)phenanthroline
12. Apto253
13. Apto 253
14. Lor253
15. Unii-wb59mrw00u
16. Chembl4297261
17. Schembl10067266
18. Lt-253lor-253
19. Ex-a3460
20. Lor-253;lt-253
21. Nsc789042
22. S6963
23. Zinc137222115
24. Cs-5226
25. Nsc-789042
26. Ncgc00484061-01
27. Ac-36395
28. Ba166663
29. Hy-16291
30. A13838
31. E76865
32. A915416
| Molecular Weight | 367.4 g/mol |
|---|---|
| Molecular Formula | C22H14FN5 |
| XLogP3 | 4.3 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Exact Mass | 367.12332363 g/mol |
| Monoisotopic Mass | 367.12332363 g/mol |
| Topological Polar Surface Area | 70.2 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 589 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?