
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Canada

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
FDF
0
Data Compilation #PharmaFlow
0
Weekly News Recap #Phispers
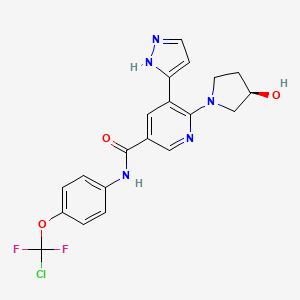

1. Abl001
2. Asciminib Hydrochloride
1. Abl-001
2. 1492952-76-7
3. Abl001
4. Asciminib Free Base
5. Abl001-nx
6. Nvp-abl001
7. Asciminib [usan]
8. Scemblix
9. Example 9
10. L1f3r18w77
11. 1492952-76-7 (free Base)
12. (r)-n-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-1-yl)-5-(1h-pyrazol-5-yl)nicotinamide
13. 3-pyridinecarboxamide, N-(4-(chlorodifluoromethoxy)phenyl)-6-((3r)-3-hydroxy-1-pyrrolidinyl)-5-(1h-pyrazol-3-yl)-
14. N-[4-[chloro(difluoro)methoxy]phenyl]-6-[(3r)-3-hydroxypyrrolidin-1-yl]-5-(1h-pyrazol-5-yl)pyridine-3-carboxamide
15. 3-pyridinecarboxamide, N-[4-(chlorodifluoromethoxy)phenyl]-6-[(3r)-3-hydroxy-1-pyrrolidinyl]-5-(1h-pyrazol-3-yl)-
16. Asciminib [inn]
17. Asciminib (abl001)
18. Asciminib (usan/inn)
19. Asciminib [who-dd]
20. Unii-l1f3r18w77
21. Gtpl8962
22. Chembl4208229
23. Schembl15388306
24. Tqp0925
25. Ex-a3030
26. Bdbm50459091
27. Nsc789925
28. S8555
29. Zinc150275965
30. At30330
31. Ccg-269232
32. Compound 1 [pmid: 30137981]
33. Cs-7655
34. Db12597
35. Nsc-789925
36. (r)-n- (4-(chlorodifluoromethoxy)phenyl)- 6-(3- Hydroxypyrrolidin-1- Yl)-5- (1h-pyrazol- 5-yl)nicotinamide
37. Ba166957
38. Bs-15538
39. Hy-104010
40. D11403
41. A910986
42. Q27074535
43. (r)-n-(4-(chloro Difluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-1-yl)-5-(1h-pyrazol-5-yl)nicotinamide
44. (r)-n-(4-(chlorodifluoromethoxy)phenyl)-6-(3-hydroxypyrrolidin-1-yl)-5-(1h-pyrazol-3-yl)nicotinamide
45. Ay7
46. N-(4-(chlorodifluoromethoxy)phenyl)-6-((3r)-3- Hydroxypyrrolidin-1-yl)-5-(1h-pyrazol-3-yl)pyridine- 3-carboxamide
| Molecular Weight | 449.8 g/mol |
|---|---|
| Molecular Formula | C20H18ClF2N5O3 |
| XLogP3 | 3 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 6 |
| Exact Mass | 449.1066235 g/mol |
| Monoisotopic Mass | 449.1066235 g/mol |
| Topological Polar Surface Area | 103 Ų |
| Heavy Atom Count | 31 |
| Formal Charge | 0 |
| Complexity | 626 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Asciminib is indicated for the treatment of adult patients with Philadelphia chromosome-positive chronic myeloid leukemia (Ph+ CML) in chronic phase who have been previously treated with 2 tyrosine kinase inhibitors. It is also indicated in the treatment of Ph+ CML in adult patients with the T315I mutation.
Scemblix is indicated for the treatment of adult patients with Philadelphia chromosome positive chronic myeloid leukaemia in chronic phase (Ph+ CML CP) previously treated with two or more tyrosine kinase inhibitors (see section 5. 1).
Asciminib exerts its therapeutic activity by inhibiting an oncogenic protein responsible for the proliferation of CML. It may be administered orally once or twice a day depending on the condition being treated. By increasing the total daily dose 5-fold as compared to standard therapy (80mg daily vs. 400mg daily), it can be used to treat Ph+ CML with the T315I mutation, a typically treatment-resistant variant of the disease. As with many other chemotherapeutic agents, asciminib treatment can result in various forms of myelosuppression, including thrombocytopenia and neutropenia. Patients should receive frequent laboratory monitoring throughout therapy and dose adjustments may be required based on the severity of observed effects. Patients may also experience pancreatic and/or cardiovascular toxicity, both of which require frequent monitoring and may require dose adjustments as per prescribing information.
L01EA06
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01EA - Bcr-abl tyrosine kinase inhibitors
L01EA06 - Asciminib
Absorption
The median Tmax of asciminib following oral administration is 2.5 hours. At a dose of 80mg once daily, the steady-state Cmax and AUCtau were 1781 ng/mL and 15112 ng.h/mL, respectively. At a dose of 40mg twice daily, the steady-state Cmax and AUCtau were 793 ng/mL and 5262 ng.h/mL, respectively. At a dose of 200mg twice daily (for treatment of T315I mutants), the steady-state Cmax and AUCtau were 5642 ng/mL and 37547 ng.h/mL, respectively. As compared to the fasted state, the co-administration of asciminib with a high-fat meal decreased the AUC and Cmax by 62% and 68%, respectively, and its co-administration with a low-fat meal decreased the AUC and Cmax by 30% and 35%, respectively.
Route of Elimination
Asciminib is eliminated via biliary secretion facilitated by breast cancer-resistant protein (BCRP) transporters. Following oral administration, approximately 80% and 11% of an asciminib dose was recovered in the feces and urine, respectively. Unchanged parent drug accounted for 57% of drug material recovered in the feces and 2.5% in the urine.
Volume of Distribution
At steady-state, the apparent volume of distribution of asciminib is 151 L.
Clearance
The total apparent clearance of asciminib is 6.7 L/h at a total daily dose of 80mg and 4.1 L/h at a dose of 200mg twice daily.
Asciminib is negligibly metabolized, with unchanged parent drug comprising the main drug component in plasma (~93%) and following excretion (~57% in feces). The main circulating metabolites are M30.5, M44, and M29.5, accounting for approximately 5%, 2%, and 0.4% of the total administered dose, respectively. The oxidative metabolism of asciminib is mediated by CYP3A4, and the glucuronidation of asciminib is mediated by UGT2B7 and UGT2B17.
The terminal elimination half-life asciminib is 5.5 hours when administered at 40mg twice daily and 9.0 hours when administered at 200mg twice daily.
In most patients with chronic myeloid leukemia (CML), progression of the disease is driven primarily by a translocation of the Philadelphia chromosome that creates an oncogenic fusion gene, _BCR-ABL1_, between the _BCR_ and _ABL1_ genes. This fusion gene produces a resultant fusion protein, BCR-ABL1, which exhibits elevated tyrosine kinase and transforming activities that contribute to CML proliferation. Asciminib is an allosteric inhibitor of the BCR-ABL1 tyrosine kinase. It binds to the myristoyl pocket of the ABL1 portion of the fusion protein and locks it into an inactive conformation, preventing its oncogenic activity.

14.2k
<10
2.0
28,346.5
API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|---|---|---|
| INDIA | 2.00 | 14,173.3 | <10 |
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
35
PharmaCompass offers a list of Asciminib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Asciminib manufacturer or Asciminib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Asciminib manufacturer or Asciminib supplier.
PharmaCompass also assists you with knowing the Asciminib API Price utilized in the formulation of products. Asciminib API Price is not always fixed or binding as the Asciminib Price is obtained through a variety of data sources. The Asciminib Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Asciminib manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Asciminib, including repackagers and relabelers. The FDA regulates Asciminib manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Asciminib API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Asciminib manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Asciminib supplier is an individual or a company that provides Asciminib active pharmaceutical ingredient (API) or Asciminib finished formulations upon request. The Asciminib suppliers may include Asciminib API manufacturers, exporters, distributors and traders.
click here to find a list of Asciminib suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Asciminib as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Asciminib API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Asciminib as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Asciminib and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Asciminib NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Asciminib suppliers with NDC on PharmaCompass.
Asciminib Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Asciminib GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Asciminib GMP manufacturer or Asciminib GMP API supplier for your needs.
A Asciminib CoA (Certificate of Analysis) is a formal document that attests to Asciminib's compliance with Asciminib specifications and serves as a tool for batch-level quality control.
Asciminib CoA mostly includes findings from lab analyses of a specific batch. For each Asciminib CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Asciminib may be tested according to a variety of international standards, such as European Pharmacopoeia (Asciminib EP), Asciminib JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Asciminib USP).
