
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
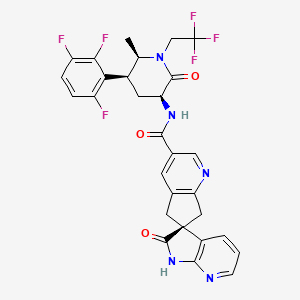

1. (3s)-n-((3s,5s,6r)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl)-2-oxospiro(1h-pyrrolo(2,3-b)pyridine-3,6'-5,7-dihydrocyclopenta(b)pyridine)-3'-carboxamide
1. Qulipta
2. Mk-8031
3. 1374248-81-3
4. Agn-241689
5. Atogepant [inn]
6. Atogepant [who-dd]
7. 7crv8rr151
8. Atogepant (usan)
9. Atogepant [usan]
10. (3's)-1',2',5,7-tetrahydro-n-((3s,5s,6r)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)-3-piperidinyl)-2'-oxospiro(6h-cyclopenta(b)pyridine-6,3'-(3h)pyrrolo(2,3-b)pyridine)-3-carboxamide
11. (3's)-n-[(3s,5s,6r)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl]-2'-oxo-1',2',5,7-tetrahydrospiro[cyclopenta[b]pyridine-6,3'-pyrrolo[2,3-b]pyridine]-3-carboxamide
12. (3s)-n-[(3s,5s,6r)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)piperidin-3-yl]-2-oxospiro[1h-pyrrolo[2,3-b]pyridine-3,6'-5,7-dihydrocyclopenta[b]pyridine]-3'-carboxamide
13. Spiro(6h-cyclopenta(b)pyridine-6,3'-(3h)pyrrolo(2,3-b)pyridine)-3-carboxamide, 1',2',5,7-tetrahydro-n-((3s,5s,6r)-6-methyl-2-oxo-1-(2,2,2-trifluoroethyl)-5-(2,3,6-trifluorophenyl)-3-piperidinyl)-2'-oxo-, (3's)-
14. Atogepant [usan:inn]
15. Unii-7crv8rr151
16. Gtpl9730
17. Schembl4570348
18. Atogepant [orange Book]
19. Chembl3991065
20. Dtxsid901337079
21. Bcp29018
22. Ex-a6847
23. Mk 8031; Mk8031; Atogepant
24. Example 4 [us20120122911]
25. Hy-109022
26. Cs-0030524
27. D11313
| Molecular Weight | 603.5 g/mol |
|---|---|
| Molecular Formula | C29H23F6N5O3 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 4 |
| Exact Mass | 603.17050859 g/mol |
| Monoisotopic Mass | 603.17050859 g/mol |
| Topological Polar Surface Area | 104 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 1110 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Atogepant is indicated for the prevention of episodic migraine in adults.
Prevention of migraine headaches
Atogepant helps to prevent migraine headaches by antagonizing the activity of a pronociceptive molecule (CGRP) which has been implicated in migraine pathophysiology. Intended for preventative use, rather than abortive migraine therapy, atogepant is administered once daily. While no dose adjustments are required for patients with mild or moderate hepatic impairment, atogepant should be avoided in patients with severe hepatic impairment. Similarly, no dose adjustments are required for patients with mild or moderate renal impairment, but patients with severe renal impairment should be limited to a maximum daily dose of 10mg.
N - Nervous system
N02 - Analgesics
N02C - Antimigraine preparations
N02CD - Calcitonin gene-related peptide (cgrp) antagonists
N02CD07 - Atogepant
Absorption
The time to peak plasma concentration following oral administration is approximately 2-3 hours. Atogepant displays dose-proportional pharmacokinetics up to approximately 3-fold its recommended maximum dosage, and its pharmacokinetics are not significantly influenced by co-administration with food.
Route of Elimination
The elimination of atogepant occurs primarily via metabolism by CYP3A4. Following a single oral dose of radiolabeled atogepant to healthy male subjects, 42% of the administered dose was recovered as unchanged parent drug in the feces and 5% as unchanged parent drug in the urine. In total, approximately 81% of the radioactivity was recovered in the feces, with only 8% recovered in the urine.
Volume of Distribution
The mean apparent volume of distribution of atogepant is 292 L.
Clearance
The mean apparent oral clearance of atogepant is approximately 19 L/h.
The metabolism of atogepant is mediated primarily via CYP3A4. The most prevalent circulating compounds in plasma are atogepant itself and a glucuronide conjugate metabolite (M23), comprising approximately 75% and 15% of the administered dose, respectively, with at least 10 other metabolites detected in feces representing <10% of the administered dose.
The elimination half-life of atogepant following oral administration is approximately 11 hours.
The currently accepted theory of migraine pathophysiology considers dysfunction of the central nervous system, in particular the trigeminal ganglion, to be the root cause behind the condition. Activation of the trigeminal ganglion triggers the stimulation of trigeminal afferents that project to the spinal cord and synapse on various pain-sensing intra- and extracranial structures, such as the dura mater. Pain signals are then further transmitted via second-order ascending neurons to the brainstem, hypothalamus, and thalamic nuclei, and from there to several cortical regions (e.g. auditory, visual, motor cortices). The trigeminal ganglion appears to amplify and perpetuate the migraine headache pain through the activation of perivascular fibers and the release of molecules involved in pain generation, such as calcitonin gene-related peptide (CGRP). The -isoform of CGRP, expressed in primary sensory neurons, is a potent vasodilator and has been implicated in migraine pathogenesis - CGRP levels are acutely elevated during migraine attacks, return to normal following treatment with triptan medications, and intravenous infusions of CGRP have been shown to trigger migraine-like headaches in migraine patients. In addition to its vasodilatory properties, CGRP appears to be a pronociceptive factor that modulates neuronal excitability to facilitate pain responses. Atogepant is an antagonist of the calcitonin gene-related peptide receptor - it competes with CGRP for occupancy at these receptors, preventing the actions of CGRP and its ability to induce and perpetuate migraine headache pain.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
25
PharmaCompass offers a list of Atogepant API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Atogepant manufacturer or Atogepant supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Atogepant manufacturer or Atogepant supplier.
A Atogepant manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Atogepant, including repackagers and relabelers. The FDA regulates Atogepant manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Atogepant API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Atogepant manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Atogepant supplier is an individual or a company that provides Atogepant active pharmaceutical ingredient (API) or Atogepant finished formulations upon request. The Atogepant suppliers may include Atogepant API manufacturers, exporters, distributors and traders.
click here to find a list of Atogepant suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Atogepant DMF (Drug Master File) is a document detailing the whole manufacturing process of Atogepant active pharmaceutical ingredient (API) in detail. Different forms of Atogepant DMFs exist exist since differing nations have different regulations, such as Atogepant USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Atogepant DMF submitted to regulatory agencies in the US is known as a USDMF. Atogepant USDMF includes data on Atogepant's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Atogepant USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Atogepant suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Atogepant Drug Master File in Korea (Atogepant KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Atogepant. The MFDS reviews the Atogepant KDMF as part of the drug registration process and uses the information provided in the Atogepant KDMF to evaluate the safety and efficacy of the drug.
After submitting a Atogepant KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Atogepant API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Atogepant suppliers with KDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Atogepant as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Atogepant API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Atogepant as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Atogepant and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Atogepant NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Atogepant suppliers with NDC on PharmaCompass.
Atogepant Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Atogepant GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Atogepant GMP manufacturer or Atogepant GMP API supplier for your needs.
A Atogepant CoA (Certificate of Analysis) is a formal document that attests to Atogepant's compliance with Atogepant specifications and serves as a tool for batch-level quality control.
Atogepant CoA mostly includes findings from lab analyses of a specific batch. For each Atogepant CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Atogepant may be tested according to a variety of international standards, such as European Pharmacopoeia (Atogepant EP), Atogepant JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Atogepant USP).

