
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Aziridine, Conjugate Acid
2. Ethyleneimine
3. Ethylenimine
1. Ethyleneimine
2. Ethylenimine
3. 151-56-4
4. Azacyclopropane
5. 9002-98-6
6. Dimethyleneimine
7. Ethylene Imine
8. Polyethyleneimine
9. Dihydroazirene
10. Aziran
11. Aziridine, Homopolymer
12. Dihydro-1h-azirine
13. Aethylenimin
14. Ethyleenimine
15. Etilenimina
16. Aziridin
17. Everamine
18. Polymin
19. 1h-azirine, Dihydro-
20. Polymine P
21. Polymin P
22. Polymin Fl
23. Montrek 6
24. Ethylenimine Resins
25. Everamine 50t
26. Poly(ethylenimine)
27. Everamine 210t
28. Rcra Waste Number P054
29. Azirane
30. Montrek Pei 6
31. Aziridine Homopolymer
32. Epamine 150t
33. Epomine 150t
34. Montrek Pei 18
35. Tydex 12
36. Epomine 1000
37. Ethylenimine, Polymers
38. Montrek 1000
39. Epomine P 1000
40. Cf 218 (polymer)
41. P 1000 (polyamine)
42. Pei 1
43. Pei 2
44. Pei 6
45. Ent-50324
46. Tl 337
47. Dimethylenimine
48. Pei 12
49. Pei 18
50. Pei 100
51. Pei 400
52. Pei-600
53. Pei 1000
54. Pei 1120
55. 54p5fex9fh
56. Polyaziridine
57. Poly(ethyleneimine)(approx. 30% In Water)
58. Dtxsid8020599
59. Chebi:30969
60. Ethylenimine Polymer
61. Dihydroazirine
62. Aziridin [german]
63. Ei
64. Aethylenimin [german]
65. Ethyleenimine [dutch]
66. Etilenimina [italian]
67. Pei 600
68. Dow Pei-6
69. Ethylenimine, Homopolymer
70. Dow Pei-600e
71. Dow Pei-18
72. Polyethylenimine, M.w.600
73. Pei
74. Ccris 296
75. Polyethylenimine, M.w.1800
76. Hsdb 540
77. P 1000
78. Einecs 205-793-9
79. Un1185
80. Rcra Waste No. P054
81. Unii-54p5fex9fh
82. Brn 0102380
83. Ai3-50324
84. Aziridine, Homopolymer, Ethoxylated
85. Ethyleneimine;aziridine;ethylenimine
86. Aziridine;ethylenimine
87. Polyethylenimine Linear
88. Polyethyleneimine Linear
89. Ethyleneimine, Inhibited
90. Aziridine [iarc]
91. Aziridine [inci]
92. Ethylenimine [mi]
93. Ec 205-793-9
94. Ethyleneimine [hsdb]
95. Wln: /t3mtj/
96. 5-20-01-00003 (beilstein Handbook Reference)
97. Un 1185 (salt/mix)
98. Chembl540990
99. Aziridine, Homopolymer, Ethoxylated, Phosphonomethylated
100. Ethoxylated Polyethylenimine,000
101. Bcp29842
102. Str07159
103. Mfcd00039669
104. Nsc124034
105. Nsc124035
106. Nsc124036
107. Nsc134422
108. Nsc196335
109. Stl168030
110. Aziridine 100 Microg/ml In Methanol
111. Akos005366671
112. Nsc-124034
113. Nsc-124035
114. Nsc-124036
115. Nsc-134422
116. Nsc-196335
117. 68130-98-3
118. 68130-99-4
119. Cs-0058423
120. Ft-0688199
121. Ft-0699349
122. Ethyleneimine, Inhibited [un1185] [poison]
123. 151e564
124. Q409141
125. Aziridine (ethyleneimine), Stabilized With 1-2% Naoh
126. 49553-93-7
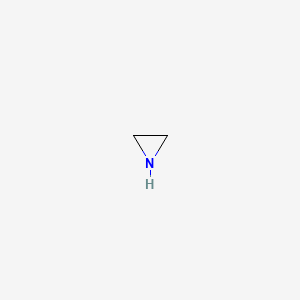
| Molecular Weight | 43.07 g/mol |
|---|---|
| Molecular Formula | C2H5N |
| XLogP3 | -0.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 0 |
| Exact Mass | 43.042199164 g/mol |
| Monoisotopic Mass | 43.042199164 g/mol |
| Topological Polar Surface Area | 21.9 Ų |
| Heavy Atom Count | 3 |
| Formal Charge | 0 |
| Complexity | 10.3 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Goups of 5-6 male Dow-Wistar rats were injected intraperitoneally with 0.3-0.42 mg/kg 14-C-aziridine (ethyleneimine) and were sacrificed after 24 and 96 hours. In both cases, ca. 50% of the administered dose was excreted in the urine; small amounts were present in the feces and expired air (3-5%). A small amount of the test substance itself was detected in urine, the major part of the radioactivity was due to unknown metabolites. Radioactivity was distributed throughout the rats, with a slight accumulation in liver, intestine, caecum, spleen, and kidneys.
European Chemicals Bureau; IUCLID Dataset, Ethyleneimine (151-56-4) (2000 CD-ROM edition). Available from, as of February 14, 2006: https://esis.jrc.ec.europa.eu/
... /It/ penetrates animal skin so rapidly that its percutaneous toxicity is not decreased if it is washed off 1 min after contact.
International Labour Office. Encyclopedia of Occupational Health and Safety. Vols. I&II. Geneva, Switzerland: International Labour Office, 1983., p. 231
When (14)C-ethyleneimine (0.30-0.42 mg/kg body weight) was injected ip into rats, approx one-half of the (14)C was excreted into the urine and 2-6% in feces after 96 hr. Only a small amount of urinary (14)C was ethyleneimine; the major portion of the urinary (14)C was found in a number of unidentified products. Three to 5% of the (14)C was expired as CO2, and 1-3% was expired as ethyleneimine.
PMID:5586366 Wright GJ, Rowe VK; Toxicol Appl Pharmacol 11: 575-84 (1967)
No reports found; [TDR, p. 688]
TDR - Ryan RP, Terry CE, Leffingwell SS (eds). Toxicology Desk Reference: The Toxic Exposure and Medical Monitoring Index, 5th Ed. Washington DC: Taylor & Francis, 1999., p. 688
The ethylenimines are classic alkylating agents with biologic effects similar to the beta-chloroethylamines. /Ethylenimines/
Clayton, G. D. and F. E. Clayton (eds.). Patty's Industrial Hygiene and Toxicology: Volume 2A, 2B, 2C: Toxicology. 3rd ed. New York: John Wiley Sons, 1981-1982., p. 2672
Ethyleneimine reacted with guanosine in aqueous medium to yield two identified products: imidazole-ring-opened 7-alkylguanosine (80%) and intact 1-alkylguanosine (14%). Incubation of ethyleneimine with guanosine or deoxyguanosine at pH 6.0 (1 hr, 37 C) yielded some intact 7-alkylated products. Half-life of imidazole ring opening of 7-alkylguanosine was 11, 5, and 2.8 min at pH 7.0, 7.7, and was 21 min at pH 7.7.
Hemminki K; Chem-Biol Interactions 48: 249-260 (1984)
... Examples of direct-acting mutagens include alkylating agents such as ... ethyleneimine ... Direct-acting carcinogens are typically carcinogenic at multiple sites and in all species examined ...
Klaassen, C.D. (ed). Casarett and Doull's Toxicology. The Basic Science of Poisons. 6th ed. New York, NY: McGraw-Hill, 2001., p. 257
ABOUT THIS PAGE
25
PharmaCompass offers a list of Aziridine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Aziridine manufacturer or Aziridine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Aziridine manufacturer or Aziridine supplier.
PharmaCompass also assists you with knowing the Aziridine API Price utilized in the formulation of products. Aziridine API Price is not always fixed or binding as the Aziridine Price is obtained through a variety of data sources. The Aziridine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Aziridine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Aziridine, including repackagers and relabelers. The FDA regulates Aziridine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Aziridine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Aziridine supplier is an individual or a company that provides Aziridine active pharmaceutical ingredient (API) or Aziridine finished formulations upon request. The Aziridine suppliers may include Aziridine API manufacturers, exporters, distributors and traders.
Aziridine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Aziridine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Aziridine GMP manufacturer or Aziridine GMP API supplier for your needs.
A Aziridine CoA (Certificate of Analysis) is a formal document that attests to Aziridine's compliance with Aziridine specifications and serves as a tool for batch-level quality control.
Aziridine CoA mostly includes findings from lab analyses of a specific batch. For each Aziridine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Aziridine may be tested according to a variety of international standards, such as European Pharmacopoeia (Aziridine EP), Aziridine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Aziridine USP).
