
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
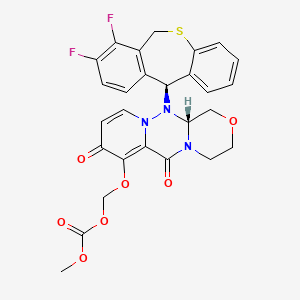

1. (((12ar)-12-((11s)-7,8-difluoro-6,11-dihydrodibenzo(b,e)thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12ahexahydro-1h-(1,4)oxazino(3,4-c)pyrido(2,1-f)(1,2,4)triazin-7-yl)oxy)methyl Methyl Carbonate
2. (12ar)-12-((11s)-7,8-difluoro-6,11-dihydrodibenzo(b,e)thiepin-11-yl)-7-hydroxy-3,4,12,12a-tetrahydro-1h-(1,4)oxazino(3,4-c)pyrido(2,1-f)(1,2,4)triazine-6,8-dione
3. Baloxavir
4. Xofluza
1. 1985606-14-1
2. Xofluza
3. Baloxavir-marboxil
4. Baloxavir Marboxil [inn]
5. S-033188
6. 505cxm6ohg
7. (((r)-12-((s)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12a-hexahydro-1h-[1,4]oxazino[3,4-c]pyrido[2,1-f][1,2,4]triazin-7-yl)oxy)methyl Methyl Carbonate
8. (((12ar)-12-((11s)-7,8-difluoro-6,11-dihydrodibenzo(b,e)thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12ahexahydro-1h-(1,4)oxazino(3,4-c)pyrido(2,1-f)(1,2,4)triazin-7-yl)oxy)methyl Methyl Carbonate
9. ({(12ar)-12-[(11s)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl]-6,8-dioxo-3,4,6,8,12,12ahexahydro-1h-[1,4]oxazino[3,4-c]pyrido[2,1-f][1,2,4]triazin-7-yl}oxy)methyl Methyl Carbonate
10. [(3r)-2-[(11s)-7,8-difluoro-6,11-dihydrobenzo[c][1]benzothiepin-11-yl]-9,12-dioxo-5-oxa-1,2,8-triazatricyclo[8.4.0.03,8]tetradeca-10,13-dien-11-yl]oxymethyl Methyl Carbonate
11. Carbonic Acid, (((12ar)-12-((11s)-7,8-difluoro-6,11-dihydrodibenzo(b,e)thiepin-11-yl)-3,4,6,8,12,12a-hexahydro-6,8-dioxo-1h-(1,4)oxazino(3,4-c)pyrido(2,1-f)(1,2,4)triazin-7-yl)oxy)methyl Methyl Ester
12. Xofluza (tn)
13. Unii-505cxm6ohg
14. Baloxavir Marboxil [mi]
15. Baloxavir Marboxil [jan]
16. Chembl4297503
17. Schembl20108731
18. Gtpl11748
19. Baloxavir Marboxil [usan]
20. Dtxsid601027758
21. Amy16544
22. Baloxavir Marboxil [who-dd]
23. Ex-a1867
24. Baloxavir Marboxil (jan/usan/inn)
25. S5952
26. Akos037652423
27. Db13997
28. Dt-0012
29. Baloxavir Marboxil [orange Book]
30. Ac-30675
31. Hy-109025
32. Cs-0030527
33. D11021
34. D70046
35. A937215
36. S033188
37. Q48333728
38. (((12ar)-12-((11s)-7,8-difluoro-6,11-dihydrodibenzo(b,e)thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12a-hexahydro-1h -(1,4)oxazino(3,4-c)pyrido(2,1-f)(1,2,4)triazin-7-yl)oxy)methyl Methylcarbonate
39. (((r)-12-((s)-7,8-difluoro-6,11-dihydrodibenzo[b,e]thiepin-11-yl)-6,8-dioxo-3,4,6,8,12,12a-hexahydro-1h-[1,4]oxazino[3,4-c]pyrido[2,1-f][1,2,4]triazin-7-yl)oxy)methylmethylcarbonate
40. 1830312-72-5
| Molecular Weight | 571.6 g/mol |
|---|---|
| Molecular Formula | C27H23F2N3O7S |
| XLogP3 | 3.8 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 6 |
| Exact Mass | 571.12247758 g/mol |
| Monoisotopic Mass | 571.12247758 g/mol |
| Topological Polar Surface Area | 123 Ų |
| Heavy Atom Count | 40 |
| Formal Charge | 0 |
| Complexity | 1090 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
For the treatment of influenza A and B virus infection, in patients 12 and older who have been symptomatic for no more than 48 hours. Clinical trials of this drug did not include subjects 65 years of age and older to determine whether they respond in a different way than younger subjects.
FDA Label
* Treatment of influenza:
Xofluza is indicated for the treatment of uncomplicated influenza in patients aged 12 years and above.
* Post exposure prophylaxis of influenza:
Xofluza is indicated for post-exposure prophylaxis of influenza in individuals aged 12 years and above. Xofluza should be used in accordance with official recommendations.
Baloxavir marboxil is a selective inhibitor of influenza cap-dependent endonuclease which prevents polymerase function and therefore influenza virus mRNA replication,. It has shown therapeutic activity against influenza A and B virus infections, including strains resistant to current antiviral agents. This drug inhibits an enzyme required for viral replication, thus rapidly treating flu virus infection, and alleviating the symptoms associated with infection. A single dose of this agent was shown to be superior to placebo in relieving influenza symptoms and superior to both oseltamivir and placebo drug in virologic outcomes (marked by decreased viral load). The safety profile of Baloxavir marboxil compared favorably with that of oseltamivir, making it an effective treatment option for treatment of the flu virus, in one single dose,.
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AX - Other antivirals
J05AX25 - Baloxavir marboxil
Absorption
Tmax: 4h
Route of Elimination
14.7 % of a single dose is excreted in the urine, and 80.1% excreted in the feces
Volume of Distribution
1180 (V/F, L).
Clearance
10.3 L/h
Baloxavir marboxil is a prodrug that is converted by hydrolysis to baloxavir, the active form that exerts anti-influenza virus activity.
Terminal elimination half-life: 79.1 h.
This drug is a CAP endonuclease inhibitor. The influenza endonuclease is an essential subdomain of the viral RNA polymerase enzyme. CAP endonuclease processes host pre-mRNAs to serve as primers for viral mRNA and therefore has been a common target for studies of anti-influenza drugs. Inhibiting the activity of endonuclease can block the transcription of mRNA and inactivate the influenza virus. Viral gene transcription is primed by short-capped oligonucleotides that are cleaved from host cell pre mRNA by endonuclease activity. Translation of viral mRNAs by the host ribosome requires that they are capped at the 5' end, and this is achieved in cells infected with influenza virus by a cap-snatching mechanism, whereby the endonuclease cleaves 5 caps from host mRNA which then act as primers for transcription. The N-terminal domain of PA subunit (PAN) has been confirmed to accommodate the endonuclease activity residues, which is highly preserved among subtypes of influenza A virus and is able to fold functionally. Translation of viral mRNAs by the host ribosome requires that they are capped at the 5' end, and this is achieved in cells infected with influenza virus by a cap-snatching mechanism, whereby the endonuclease cleaves 5 caps from host mRNA which then act as primers for transcription. The endonuclease domain binds the N-terminal half of PA (PAN) and contains a two-metal (Mn2+) active site that selectively cleaves the pre-mRNA substrate at the 3 end of a guanine. The administration of a cap-endonuclease inhibitor, such as Baloxavir marboxil, prevents the above process from occurring, exhibiting its action at the beginning of the pathway before CAP endonuclease may exert its action.
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
48
PharmaCompass offers a list of Baloxavir Marboxil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Baloxavir Marboxil manufacturer or Baloxavir Marboxil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Baloxavir Marboxil manufacturer or Baloxavir Marboxil supplier.
A Baloxavir Marboxil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Baloxavir Marboxil, including repackagers and relabelers. The FDA regulates Baloxavir Marboxil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Baloxavir Marboxil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Baloxavir Marboxil manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Baloxavir Marboxil supplier is an individual or a company that provides Baloxavir Marboxil active pharmaceutical ingredient (API) or Baloxavir Marboxil finished formulations upon request. The Baloxavir Marboxil suppliers may include Baloxavir Marboxil API manufacturers, exporters, distributors and traders.
click here to find a list of Baloxavir Marboxil suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Baloxavir Marboxil DMF (Drug Master File) is a document detailing the whole manufacturing process of Baloxavir Marboxil active pharmaceutical ingredient (API) in detail. Different forms of Baloxavir Marboxil DMFs exist exist since differing nations have different regulations, such as Baloxavir Marboxil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Baloxavir Marboxil DMF submitted to regulatory agencies in the US is known as a USDMF. Baloxavir Marboxil USDMF includes data on Baloxavir Marboxil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Baloxavir Marboxil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Baloxavir Marboxil suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Baloxavir Marboxil Drug Master File in Korea (Baloxavir Marboxil KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Baloxavir Marboxil. The MFDS reviews the Baloxavir Marboxil KDMF as part of the drug registration process and uses the information provided in the Baloxavir Marboxil KDMF to evaluate the safety and efficacy of the drug.
After submitting a Baloxavir Marboxil KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Baloxavir Marboxil API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Baloxavir Marboxil suppliers with KDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Baloxavir Marboxil as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Baloxavir Marboxil API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Baloxavir Marboxil as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Baloxavir Marboxil and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Baloxavir Marboxil NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Baloxavir Marboxil suppliers with NDC on PharmaCompass.
Baloxavir Marboxil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Baloxavir Marboxil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Baloxavir Marboxil GMP manufacturer or Baloxavir Marboxil GMP API supplier for your needs.
A Baloxavir Marboxil CoA (Certificate of Analysis) is a formal document that attests to Baloxavir Marboxil's compliance with Baloxavir Marboxil specifications and serves as a tool for batch-level quality control.
Baloxavir Marboxil CoA mostly includes findings from lab analyses of a specific batch. For each Baloxavir Marboxil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Baloxavir Marboxil may be tested according to a variety of international standards, such as European Pharmacopoeia (Baloxavir Marboxil EP), Baloxavir Marboxil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Baloxavir Marboxil USP).

