
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
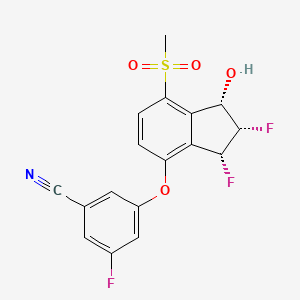

1. 3-(((1s,2s,3r)-2,3-difluoro-1-hydroxy-7-(methylsulfonyl)-2,3-dihydro-1h-inden-4-yl)oxy)-5-fluorobenzonitrile
2. Mk-6482
3. Mk6482
4. Pt-2977
5. Pt2977
6. Welireg
1. Pt2977
2. 1672668-24-4
3. Mk-6482
4. Welireg
5. Mk6482
6. Belzutifan [inn]
7. Belzutifan [usan]
8. Pt-2977
9. 3-(((1s,2s,3r)-2,3-difluoro-1-hydroxy-7-(methylsulfonyl)-2,3-dihydro-1h-inden-4-yl)oxy)-5-fluorobenzonitrile
10. 7k28nb895l
11. 3-[[(1s,2s,3r)-2,3-difluoro-1-hydroxy-7-methylsulfonyl-2,3-dihydro-1h-inden-4-yl]oxy]-5-fluorobenzonitrile
12. 3-{[(1s,2s,3r)-2,3-difluoro-1-hydroxy-7-(methylsulfonyl)-2,3-dihydro-1h-inden-4-yl]oxy}-5-fluorobenzonitrile
13. 3-((1s,2s,3r)-2,3-difluoro-1-hydroxy-7-methylsulfonylindan-4-yl)oxy-5-fluoro-benzonitrile
14. Benzonitrile, 3-(((1s,2s,3r)-2,3-difluoro-2,3-dihydro-1-hydroxy-7-(methylsulfonyl)-1h-inden-4-yl)oxy)-5-fluoro-
15. Belzutifan [who-dd]
16. Unii-7k28nb895l
17. Belzutifan [orange Book]
18. Chembl4585668
19. Schembl16560918
20. Gtpl11251
21. Pt 2977 [who-dd]
22. Bdbm373040
23. Dtxsid201334517
24. Pt 2977
25. Ex-a4441
26. Us9896418, Compound 289
27. Xrc66824
28. Nsc825217
29. Who 11196
30. At14994
31. Belzutifan (pt2977, Mk-6482)
32. Compound 2 [pmid: 31282155]
33. Nsc-825217
34. Mk-6482; Pt2977
35. Ac-35183
36. Hy-125840
37. Cs-0101119
38. A935088
39. Q27456641
40. 3-(((1s,2s,3r)-2,3-difluoro-1-hydroxy-7-(methanesulfonyl)-2,3-dihydro-1h-inden-4-yl)oxy)-5-fluorobenzonitrile
41. 3-((1s,2s,3r)-2,3-difluoro-1-hydroxy-7-(methylsulfonyl)-2,3-dihydro-1h-inden-4-yloxy)-5-fluorobenzonitrile
42. 72q
| Molecular Weight | 383.3 g/mol |
|---|---|
| Molecular Formula | C17H12F3NO4S |
| XLogP3 | 2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 3 |
| Exact Mass | 383.04391352 g/mol |
| Monoisotopic Mass | 383.04391352 g/mol |
| Topological Polar Surface Area | 95.8 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 675 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Belzutifan is indicated for the treatment of adult patients with von Hippel-Lindau (VHL) disease who require therapy for associated renal cell carcinoma (RCC), central nervous system (CNS) hemangioblastomas, or pancreatic neuroendocrine tumors (pNET), who do not require immediate surgery.
Belzutifan exerts its therapeutic effects by inhibiting a transcription factor necessary for the growth of solid tumors associated with VHL disease. It is taken once daily at approximately the same time each day, with or without food. Both severe anemia and hypoxia have been observed following therapy with belzutifan, and patients should be monitored closely before and during therapy to ensure patients can be managed as clinically indicated. There are no data regarding the use of erythropoiesis-stimulating agents for the treatment of belzutifan-induced anemia, and as such these therapies should be avoided. Belzutifan may cause embryo-fetal toxicity when administered to pregnant women. Female patients and male patients with female partners of reproductive potential should ensure that an effective form of contraception is used throughout therapy and for one week after the last dose - as belzutifan appears to decrease the efficacy of systemic hormonal contraceptives, patients should be advised to use an additional method of contraception (e.g. condoms) to eliminate the possibility of pregnancy during therapy.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01X - Other antineoplastic agents
L01XX - Other antineoplastic agents
L01XX74 - Belzutifan
Absorption
In patients with VHL disease-associated renal cell carcinoma, the mean Cmax and AUC0-24h at steady-state - which was achieved after approximately three days of therapy - were 1.3 g/mL and 16.7 ghr/mL, respectively. The median Tmax is one to two hours following oral administration. The administration of belzutifan with food has a negligible effect on drug disposition - when given alongside a high-calorie, high-fat meal, the Tmax was delayed by approximately 2 hours with no other clinically meaningful effects observed.
Volume of Distribution
The steady-state volume of distribution of belzutifan following oral administration is approximately 130 L.
Clearance
The mean clearance of belzutifan following oral administration is 7.3 L/h.
Belzutifan is primarily metabolized by UGT2B17 and CYP2C19, and to a lesser extent by CYP3A4.
The mean elimination half-life of belzutifan is 14 hours.
Hypoxia-inducible factor 2 (HIF-2) is a transcription factor which aids in oxygen sensing by regulating genes that promote adaptation to hypoxia. In healthy patients, when oxygen levels are normal, HIF-2 is broken down via ubiquitin-proteasomal degradation by von-Hippel Lindau (VHL) proteins. In the presence of hypoxia, HIF-2 translocates into cell nuclei and forms a transcriptional complex with hypoxia-inducible factor 1 (HIF-1) - this complex then induces the expression of downstream genes associated with cellular proliferation and angiogenesis. Patients with von-Hippel Lindau (VHL) disease lack functional VHL proteins, leading to an accumulation of HIF-2, and this accumulation is what drives the growth of VHL-associated tumors. Belzutifan is an inhibitor of HIF-2 that prevents its complexation with HIF-1 in conditions of hypoxia or impaired VHL protein function, thereby reducing the expression of HIF-2 target genes and slowing/stopping the growth of VHL-associated tumors.

16.5k
<10
21.2
3,49,113.2
API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|---|---|---|
| CANADA | 1.20 | 1,18,263.4 | <10 |
| INDIA | 20.00 | 10,359.9 | <10 |
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
84
PharmaCompass offers a list of Belzutifan API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Belzutifan manufacturer or Belzutifan supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Belzutifan manufacturer or Belzutifan supplier.
PharmaCompass also assists you with knowing the Belzutifan API Price utilized in the formulation of products. Belzutifan API Price is not always fixed or binding as the Belzutifan Price is obtained through a variety of data sources. The Belzutifan Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Belzutifan manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Belzutifan, including repackagers and relabelers. The FDA regulates Belzutifan manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Belzutifan API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Belzutifan manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Belzutifan supplier is an individual or a company that provides Belzutifan active pharmaceutical ingredient (API) or Belzutifan finished formulations upon request. The Belzutifan suppliers may include Belzutifan API manufacturers, exporters, distributors and traders.
click here to find a list of Belzutifan suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Belzutifan DMF (Drug Master File) is a document detailing the whole manufacturing process of Belzutifan active pharmaceutical ingredient (API) in detail. Different forms of Belzutifan DMFs exist exist since differing nations have different regulations, such as Belzutifan USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Belzutifan DMF submitted to regulatory agencies in the US is known as a USDMF. Belzutifan USDMF includes data on Belzutifan's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Belzutifan USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Belzutifan suppliers with USDMF on PharmaCompass.
Belzutifan Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Belzutifan GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Belzutifan GMP manufacturer or Belzutifan GMP API supplier for your needs.
A Belzutifan CoA (Certificate of Analysis) is a formal document that attests to Belzutifan's compliance with Belzutifan specifications and serves as a tool for batch-level quality control.
Belzutifan CoA mostly includes findings from lab analyses of a specific batch. For each Belzutifan CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Belzutifan may be tested according to a variety of international standards, such as European Pharmacopoeia (Belzutifan EP), Belzutifan JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Belzutifan USP).
