
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. 3 Alpha Hydroxy 5 Alpha Pregnan 20 One
2. 3 Alpha Hydroxy 5 Beta Pregnan 20 One
3. 3 Alpha, 5 Beta Tetrahydroprogesterone
4. 3 Alpha, 5 Beta-tetrahydroprogesterone
5. 3 Alpha-hydroxy-5 Alpha-pregnan-20-one
6. 3 Alpha-hydroxy-5 Beta-pregnan-20-one
7. 3 Hydroxypregnan 20 One
8. 3-hydroxypregnan-20-one
9. 3beta Hydroxy 5alpha Pregnan 20 One
10. 3beta-hydroxy-5alpha-pregnan-20-one
11. Allopregnan 3 Beta Ol 20 One
12. Allopregnan-3 Beta-ol-20-one
13. Alpha-hydroxy-5 Alpha-pregnan-20-one, 3
14. Alpha-hydroxy-5 Beta-pregnan-20-one, 3
15. Alpha-pregnan-20-one, 3 Alpha-hydroxy-5
16. Beta-ol-20-one, Allopregnan-3
17. Beta-pregnan-20-one, 3 Alpha-hydroxy-5
18. Eltanolone
19. Epipregnanolone
20. Pregnan 3alpha Ol 20 One
21. Pregnan-3alpha-ol-20-one
22. Pregnanolone
23. Pregnanolone, (3alpha)-isomer
24. Pregnanolone, (3alpha, 5beta, 17-alpha)-isomer
25. Pregnanolone, (3alpha,5alpha)-isomer
26. Pregnanolone, (3alpha,5beta)-isomer
27. Pregnanolone, (3beta)-isomer
28. Pregnanolone, (3beta, 5alpha)-isomer
29. Pregnanolone, (3beta, 5alpha, 17alpha)-isomer
30. Pregnanolone, (3beta, 5alpha, 8alpha, 17beta)-isomer
31. Pregnanolone, (3beta, 5beta)-isomer
32. Pregnanolone, (3beta, 5beta, 17alpha)-isomer
33. Pregnanolone, (3beta, 5beta,14beta)-isomer
34. Pregnanolone, (5alpha)-isomer
35. Sepranolone
1. 516-54-1
2. Brexanolone
3. Allopregnan-3alpha-ol-20-one
4. Allotetrahydroprogesterone
5. Sage-547
6. Zulresso
7. 3alpha-hydroxy-5alpha-pregnan-20-one
8. 3alpha-oh Dhp
9. 3alpha,5alpha-thp
10. Sge-102
11. 3a,5a-thp
12. (3alpha,5alpha)-3-hydroxypregnan-20-one
13. Brexanolone [usan]
14. 3alpha-hydroxy-5alpha-dihydroprogesterone
15. 3a-hydroxy-5a-pregnan-20-one
16. S39xz5qv8y
17. Chembl207538
18. Pregnan-20-one, 3-hydroxy-, (3a,5a)-
19. Chebi:50169
20. 3alpha,5alpha-tetrahydroprogesterone
21. 1-[(3r,5s,8r,9s,10s,13s,14s,17s)-3-hydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]ethanone
22. 1-((3r,5s,8r,9s,10s,13s,14s,17s)-3-hydroxy-10,13-dimethylhexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)ethan-1-one
23. Allopregnan-3.alpha.-ol-20-one
24. C21h34o2
25. 3-alpha,5-alpha-pregnanolone
26. Sage547
27. (3
28. A)-allopregnanolone
29. Unii-s39xz5qv8y
30. 3-alpha,5-alpha-tetrahydroprogesterone
31. Allopregnan-3a-ol-20-one
32. Brexanolona
33. Brexanolonum
34. Brn 3211363
35. Allopregnalone
36. 3-alpha-hydroxy-5-alpha-pregnan-20-one
37. 3alpha-hydroxy-5alpha-pregnane-20-one
38. (3?,5?)-3-hydroxy-pregnan-20-one
39. Ncgc00163287-01
40. Zulresso (tn)
41. 3a,5a-pregnanolone
42. (3a)-allopregnanolone
43. 3a,5a-thprog
44. 5-alpha-pregnan-20-one, 3-alpha-hydroxy-
45. Brexanolone [inn]
46. (3alpha)-allopregnanolone
47. 3-a-tetrahydroprogesterone
48. Brexanolone (usan/inn)
49. Pregnan-20-one, 3-hydroxy-, (3.alpha.,5.alpha.)-
50. Biomol-nt_000266
51. Dsstox_cid_26342
52. Dsstox_rid_81546
53. 3a,5a-tetrahydroprogesterone
54. Bidd:pxr0069
55. Dsstox_gsid_46342
56. 5a-pregnan-3a-ol-20-one
57. Brexanolone [who-dd]
58. 3-alpha-tetrahydroprogesterone
59. 4-08-00-00664 (beilstein Handbook Reference)
60. 5a-pregnane-3a-ol-20-one
61. Schembl588060
62. Pregnan-20-one, 3-hydroxy-, (3-alpha,5-alpha)-
63. Bpbio1_000728
64. Gtpl4108
65. 3a-hydroxy-5a-pregnane-20-one
66. Brexanolone [orange Book]
67. 3a-hydroxy-5a-dihydroprogesterone
68. 5
69. A-pregnan-3
70. A-ol-20-one
71. 5alpha Pregnan 3alpha Ol 20 One
72. Dtxsid901016239
73. Hms3886d05
74. 5alpha-pregnane-3alpha-ol-20-one
75. Amy16827
76. Ex-a1195
77. Zinc4081043
78. Tox21_112042
79. (3a,5a)-3-hydroxypregnan-20-one
80. Bdbm50191342
81. Lmst02030156
82. S5805
83. (+)-3a-hydroxy-5a-pregnan-20-one
84. Akos024285177
85. Pregnane-3alpha-ol-20-one, 5alpha-
86. Cs-6401
87. Db11859
88. 3
89. A,5
90. A-thp;sage-547;brexanolone
91. Ncgc00163287-02
92. 5alpha-pregnan-3alpha-ol-20-one, Solid
93. Ac-30353
94. As-76408
95. Cas-128-20-1
96. (3alpha,5alpha)-3-hydroxy-pregnan-20-one
97. Hy-101107
98. B7456
99. Allopregnan-3.alpha.-ol-20-one [mi]
100. D11149
101. 516a541
102. 3.alpha.-hydroxy-5.alpha.-pregnan-20-one
103. Brd-k18172896-001-01-7
104. Q28487680
105. (+)-3.alpha.-hydroxy-5.alpha.-pregnan-20-one
106. 8e5d696a-eebc-495d-a8c9-98fc9a593150
| Molecular Weight | 318.5 g/mol |
|---|---|
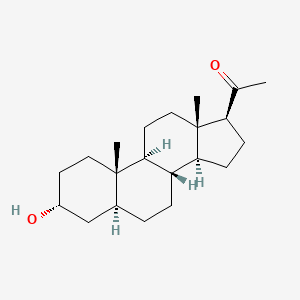
| Molecular Formula | C21H34O2 |
| XLogP3 | 4.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 1 |
| Exact Mass | 318.255880323 g/mol |
| Monoisotopic Mass | 318.255880323 g/mol |
| Topological Polar Surface Area | 37.3 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 500 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 8 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Brexanolone is a synthetic neuroactive steroid gamma-aminobutyric acid A (GABA(a)) receptor positive modulator indicated for the treatment of postpartum depression (PPD) in adult women.
FDA Label
Treatment of postpartum depression
Brexanolone potentiated GABA-mediated currents from recombinant human GABA(a) receptors in mammalian cells expressing 122 receptor subunits, 43 receptor subunits, and 63 receptor subunits. Moreover, it was determined during a Phase 1 randomized, placebo and positive-controlled, double-blind, three-period crossover thorough QT study in 30 healthy adult subjects that brexanolone use did not prolong the QT interval to any clinically relevant extent when administered at 1.9-times the exposure occurring at the highest recommended infusion rate (90 mcg/kg/hour).
Anesthetics
Agents capable of inducing a total or partial loss of sensation, especially tactile sensation and pain. They may act to induce general ANESTHESIA, in which an unconscious state is achieved, or may act locally to induce numbness or lack of sensation at a targeted site. (See all compounds classified as Anesthetics.)
Absorption
It has been determined that brexanolone has a low oral bioavailability of approximately <5% in adults, which suggests infant exposure would also be expected to be low.
Route of Elimination
Following the administration of radiolabeled brexanolone, it was observed that 47% of the administrated dose was recovered largely as metabolites in the feces and 42% in urine, where less than 1% as recovered as unchanged brexanolone.
Volume of Distribution
The volume of distribution documented for brexanolone is approximately 3 L/kg, a value which suggests relatively extensive distribution into tissues.
Clearance
The total plasma clearance determined for brexanolone is approximately 1 L/h/kg.
Brexanolone is extensively metabolized by non-cytochrome (CYP) based pathways by way of three main routes - keto-reduction (via aldo-keto reductases), glucuronidation (via UDP-glucuronosyltransferases), and sulfation (via sulfotransferases). Three predominant circulating metabolites result from such metabolic pathways and they are all pharmacologically inactive and ultimately do not contribute to the overall efficacy of the medication.
The terminal half-life observed for brexanolone is approximately 9 hours.
Brexanolone is a neuroactive steroid that occurs naturally (referred to as natural allopregnanolone) in the body when the female sex hormone progesterone is metabolized. This steroid compound is also believed to exhibit activity as a barbiturate-like, positive allosteric modulator of both synaptic and extrasynaptic GABA(a) receptors. In doing so, brexanolone can enhance the activity of GABA at such receptors by having GABA(a) receptor calcium channels open more often and for longer periods of time. Furthermore, it is believed that brexanolone elicits such action on GABA(a) receptors at a binding site that is distinct from those associated with benzodiazepines. Concurrently, GABA is considered the principal inhibitory neurotransmitter in the human body. When GABA binds to GABA(a) receptors found in neuron synapses, chloride ions are conducted across neuron cell membranes via an ion channel in the receptors. With enough chloride ions conducted, the local, associated neuron membrane potentials are hyperpolarized - making it more difficult or less likely for action potentials to fire, ultimately resulting in less excitation of the neurons, like those involved in neuronal pathways that may be in part responsible for eliciting certain traits of PPD like stress, anxiety, etc. Postpartum depression (PPD) is a mood disorder that can affect women after childbirth. Women with PPD experience feelings of extreme sadness, anxiety, and exhaustion that can make it difficult or even dangerous for them to perform various daily activities or care for themselves or for others, including newborn. Although the exact pathophysiology of PPD remains unknown, it is believed that altered profiles and rapid, unpredictable fluctuations in the blood concentrations of neuroactive steroids like endogenous brexanolone (among others), GABA, and GABA receptors occur in women who are at risk of PPD after childbirth. In particular, within the context of PPD, it is proposed that endogenous brexanolone levels can quickly drop or fluctuate variedly after childbirth and that GABA(a) receptor levels and expression are decreased and down-regulated throughout pregnancy. Such fluctuations and decreases may consequently leave women susceptible to the possibility of PPD. As a medication, synthetic brexanolone can subsequently facilitate a return of positive allosteric modulator GABA(a) modulation while GABA(a) receptor levels and expression gradually return to normal in the time following postpartum. As such, studies suggest the potential for the development of brexanolone as a new mechanism for treatment of PPD that is directly related to the underlying pathophysiology as opposed to many other antidepressant medications whose pharmacological actions are usually entirely unrelated.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
63
PharmaCompass offers a list of Brexanolone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Brexanolone manufacturer or Brexanolone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Brexanolone manufacturer or Brexanolone supplier.
PharmaCompass also assists you with knowing the Brexanolone API Price utilized in the formulation of products. Brexanolone API Price is not always fixed or binding as the Brexanolone Price is obtained through a variety of data sources. The Brexanolone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Brexanolone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Brexanolone, including repackagers and relabelers. The FDA regulates Brexanolone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Brexanolone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Brexanolone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Brexanolone supplier is an individual or a company that provides Brexanolone active pharmaceutical ingredient (API) or Brexanolone finished formulations upon request. The Brexanolone suppliers may include Brexanolone API manufacturers, exporters, distributors and traders.
click here to find a list of Brexanolone suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Brexanolone DMF (Drug Master File) is a document detailing the whole manufacturing process of Brexanolone active pharmaceutical ingredient (API) in detail. Different forms of Brexanolone DMFs exist exist since differing nations have different regulations, such as Brexanolone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Brexanolone DMF submitted to regulatory agencies in the US is known as a USDMF. Brexanolone USDMF includes data on Brexanolone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Brexanolone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Brexanolone suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Brexanolone as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Brexanolone API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Brexanolone as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Brexanolone and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Brexanolone NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Brexanolone suppliers with NDC on PharmaCompass.
Brexanolone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Brexanolone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Brexanolone GMP manufacturer or Brexanolone GMP API supplier for your needs.
A Brexanolone CoA (Certificate of Analysis) is a formal document that attests to Brexanolone's compliance with Brexanolone specifications and serves as a tool for batch-level quality control.
Brexanolone CoA mostly includes findings from lab analyses of a specific batch. For each Brexanolone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Brexanolone may be tested according to a variety of international standards, such as European Pharmacopoeia (Brexanolone EP), Brexanolone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Brexanolone USP).
