
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Listed Dossiers
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Alunbrig
2. Ap26113
1. 1197953-54-0
2. Alunbrig
3. Ap-26113
4. Brigatinib [usan]
5. Brigatinib (ap-26113)
6. Hyw8db273j
7. Ap 26113
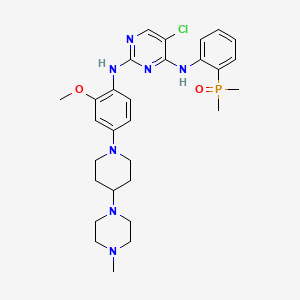
8. (2-((5-chloro-2-((2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)amino)pyrimidin-4-yl)amino)phenyl)dimethylphosphine Oxide
9. 5-chloro-4-n-(2-dimethylphosphorylphenyl)-2-n-[2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl]pyrimidine-2,4-diamine
10. 2,4-pyrimidinediamine, 5-chloro-n4-(2-(dimethylphosphinyl)phenyl)-n2-(2-methoxy-4-(4-(4-methyl-1-piperazinyl)-1-piperidinyl)phenyl)-
11. 5-chloro-n4-(2-(dimethylphosphoryl)phenyl)-n2-(2-methoxy-4-(4-(4-methylpiperazin-1- Yl)piperidin-1-yl)phenyl)pyrimidine-2,4-diamine
12. 2,4-pyrimidinediamine, 5-chloro-n4-[2-(dimethylphosphinyl)phenyl]-n2-[2-methoxy-4-[4-(4-methyl-1-piperazinyl)-1-piperidinyl]phenyl]-
13. 5-chloro-n~4~-[2-(dimethylphosphoryl)phenyl]-n~2~-{2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}pyrimidine-2,4-diamine
14. Unii-hyw8db273j
15. Brigatiib
16. Alunbrig (tn)
17. 5-chloro-n4-(2-(dimethylphosphoryl)phenyl)-n2-(2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)pyrimidine-2,4-diamine
18. 5-chloro-n4-[2-(dimethylphosphoryl)phenyl]-n2-{2-methoxy-4-[4-(4-methylpiperazin-1-yl)piperidin-1-yl]phenyl}pyrimidine-2,4-diamine
19. 6gy
20. Brigatinib [mi]
21. Brigatinib [inn]
22. Brigatinib [jan]
23. Brigatinib (jan/usan)
24. Brigatinib [usan:inn]
25. Brigatinib; Ap26113
26. Brigatinib (ap26113)
27. Brigatinib [who-dd]
28. 5-chloro-n4-[2-(dimethylphosphinyl)phenyl]-n2-[2-methoxy-4-[4-(4-methyl-1-piperazinyl)-1-piperidinyl]phenyl]-2,4-pyrimidinediamine
29. Gtpl7741
30. Brigatinib [orange Book]
31. Chembl3545311
32. Schembl11916361
33. Ex-a775
34. Dtxsid501027929
35. Amy10294
36. Bcp17214
37. Bdbm50185140
38. Mfcd29472221
39. Nsc784728
40. Nsc787457
41. Akos030257612
42. Zinc148723177
43. Cs-4278
44. Db12267
45. Nsc-784728
46. Nsc-787457
47. Sb40412
48. Compound 11q [pmid: 27144831]
49. Ncgc00483924-01
50. (2-((5-chloro-2-((2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)amino)pyrimidin-4-yl)amino)phenyl)dimethyl-,delta5-phosphanone
51. Ac-29958
52. As-75176
53. Hy-12857
54. Db-118419
55. Ft-0700112
56. S8229
57. J3.535.964h
58. D10866
59. Q27456393
60. (2-((5-chloro-2-((2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)phenyl)amino)pyrimidin-4-yl)amino)phenyl)dimethyl-,.delta.5-phosphanone
| Molecular Weight | 584.1 g/mol |
|---|---|
| Molecular Formula | C29H39ClN7O2P |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 8 |
| Exact Mass | 583.2591382 g/mol |
| Monoisotopic Mass | 583.2591382 g/mol |
| Topological Polar Surface Area | 85.9 Ų |
| Heavy Atom Count | 40 |
| Formal Charge | 0 |
| Complexity | 835 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 1 | |
|---|---|
| Drug Name | ALUNBRIG |
| Active Ingredient | BRIGATINIB |
| Company | ARIAD (Application Number: N208772. Patents: 9012462, 9273077, 9611283) |
The anaplastic lymphoma kinase positive, metastatic non-small cell lung cancer (ALK+ NSCLC), represents only 3-5% of the NSCLC cancer cases, but the ALK mutation, overexpression and presence in several oncogenic fusion proteins in solid and hematologic tumors have pointed out the importance as well as its potential as a cancer therapy target. The ALK-related cases of NSCLC are associated with the presence of the fusion gene EML4-ALK which fused the ALK protein with the echinoderm microtubule-associated protein like-4 whose original function is the correct formation of microtubules. The presence of the aberrant fusion protein results in abnormal signaling that provokes increased cell growth, proliferation and survival. Crizotinib is indicated for the treatment of such cases but the presence of ALK kinase domain mutations confer resistance to the treatment. Thus, brigatinib is indicated for the treatment of patients with ALK+ NSCLC with intolerance to Crizotinib.
FDA Label
Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase (ALK)positive advanced nonsmall cell lung cancer (NSCLC) previously not treated with an ALK inhibitor.
Alunbrig is indicated as monotherapy for the treatment of adult patients with anaplastic lymphoma kinase ALKpositive advanced NSCLC previously treated with crizotinib.
Brigitanib inhibits proliferation and in vitro viability of cells expressing the fusion protein EML4-ALK as well as 17 crizotinib-resistant ALK mutants. Its action is expanded to cells expressing EGFR deletions, ROS1-L2026M, FLT3-F691L and FLT3-D835Y. Brigitanib presents a dose-dependent inhibition of tumor growth, tumor burden and prolonged survival in mice EML4-ALK xenograft models. Time course of Brigatinib and exposure-response studies are still unknown.
L01XE43
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01ED - Anaplastic lymphoma kinase (alk) inhibitors
L01ED04 - Brigatinib
Absorption
Administration of brigatinib at a concentration of 90 mg generates a Cmax of 552 ng/ml and AUC of 8165 ng h/ml while the administration of 180 mg presents a Cmax of 1452 ng/ml and AUC of 20276 ng h/ml. It has a dose proportional exposure with an accumulation ratio on the range of 1.9 to 2.4. Following oral administration of brigatinib, the Tmax is presented in a range from 1 to 4 hours. Consumption of a high-fat meal compared to overnight fasting reduces Cmax by 13% without presenting an effect on AUC.
Route of Elimination
The elimination of brigatinib is divided in 65% in feces and 25% in urine. From the elimination in both compartments, the unchanged for of brigatinib represented 41% of the total in feces and 86% in urine.
Volume of Distribution
The apparent volume of distribution at steady state is 153 L.
Clearance
After oral administration of180 mg of brigatinib, the apparent oral clearance at steady-state is 12.7 L/h.
Brigatinib is metabolized by CYP2C8 (72.4%) and CYP3A4 (27.6%) in human liver microsomes and hepatocytes. The two major metabolites generated are the N-demethylated form and the cysteine conjugated form. Oral administration of radiolabelled brigatinib showed the systemic presence of 91.5% in the unchanged form and 3.5% of the primary metabolite AP26123. The AUC of AP26123 is less than 10% of the AUC of brigatinib and presented an inhibitory effect 3 fold lower.
The half-life of brigatinib at steady-state was 25 hours.
Brigitanib acts as a tyrosine kinase inhibitor with activity against multiple kinases including ALK, ROS1, insulin-like growth factor 1 receptor and against EGFR deletions and point mutations. It acts by inhibiting ALK phosphorylation and the activation of downstream signaling proteins.
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
50
PharmaCompass offers a list of Brigatinib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Brigatinib manufacturer or Brigatinib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Brigatinib manufacturer or Brigatinib supplier.
A Brigatinib manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Brigatinib, including repackagers and relabelers. The FDA regulates Brigatinib manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Brigatinib API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Brigatinib manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Brigatinib supplier is an individual or a company that provides Brigatinib active pharmaceutical ingredient (API) or Brigatinib finished formulations upon request. The Brigatinib suppliers may include Brigatinib API manufacturers, exporters, distributors and traders.
click here to find a list of Brigatinib suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Brigatinib Drug Master File in Korea (Brigatinib KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Brigatinib. The MFDS reviews the Brigatinib KDMF as part of the drug registration process and uses the information provided in the Brigatinib KDMF to evaluate the safety and efficacy of the drug.
After submitting a Brigatinib KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Brigatinib API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Brigatinib suppliers with KDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Brigatinib as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Brigatinib API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Brigatinib as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Brigatinib and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Brigatinib NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Brigatinib suppliers with NDC on PharmaCompass.
Brigatinib Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Brigatinib GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Brigatinib GMP manufacturer or Brigatinib GMP API supplier for your needs.
A Brigatinib CoA (Certificate of Analysis) is a formal document that attests to Brigatinib's compliance with Brigatinib specifications and serves as a tool for batch-level quality control.
Brigatinib CoA mostly includes findings from lab analyses of a specific batch. For each Brigatinib CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Brigatinib may be tested according to a variety of international standards, such as European Pharmacopoeia (Brigatinib EP), Brigatinib JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Brigatinib USP).

