
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDA Orange Book
0
Europe
0
Canada
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
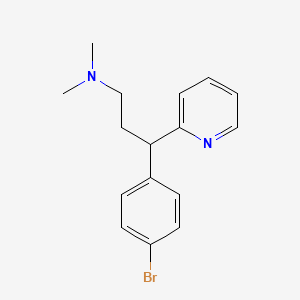

1. Brompheniramine Maleate
2. Brompheniramine Maleate (1:1)
3. Chlorphed
4. Dimetane
5. Dimetane Ten
6. Dimetane-ten
7. Dimetapp Allergy
8. Maleate, Brompheniramine
9. Oraminic 2
10. Oraminic-2
11. P Bromdylamine
12. P-bromdylamine
13. Para Bromdylamine
14. Para-bromdylamine
1. 86-22-6
2. Parabromdylamine
3. Brompheniraminum
4. Bromfeniramina
5. Ilvin
6. Bromfed
7. Parabromodylamine
8. Brotane
9. 1-(p-bromophenyl)-1-(2-pyridyl)-3-dimethylaminopropane
10. 3-(p-bromophenyl)-3-(2-pyridyl)-n,n-dimethylpropylamine
11. 2-(p-bromo-alpha-(2-dimethylaminoethyl)benzyl)pyridine
12. 3-(4-bromophenyl)-n,n-dimethyl-3-(2-pyridinyl)-1-propanamine
13. Veltane
14. Dimetane-ten
15. 3-(4-bromophenyl)-n,n-dimethyl-3-pyridin-2-ylpropan-1-amine
16. Antihistamine Compound
17. 3-(4-bromophenyl)-n,n-dimethyl-3-(pyridin-2-yl)propan-1-amine
18. Dimetapp Allergy
19. Brompheniramine (inn)
20. 2-pyridinepropanamine, .gamma.-(4-bromophenyl)-n,n-dimethyl-
21. Chembl811
22. Chebi:3183
23. H57g17p2fn
24. Lodrane
25. Bromfenex
26. Bromopheniramine
27. Brompheniramine [inn]
28. Brompheniramine [inn:ban]
29. Bromfeniramina [inn-spanish]
30. Brompheniraminum [inn-latin]
31. [3-(4-bromo-phenyl)-3-pyridin-2-yl-propyl]-dimethyl-amine
32. (+)-brompheniramine; (+)-parabromdylamine; (s)-brompheniramine
33. (+/-)-brompheniramine;bromphen; Brompheniramine; Parabromdylamine; Parabromodylamine
34. Brotane (tn)
35. Hsdb 3017
36. Einecs 201-657-8
37. Unii-h57g17p2fn
38. Gamma-(4-bromophenyl)-n,n-dimethyl-2-pyridinepropanamine
39. 2-pyridinepropanamine, Gamma-(4-bromophenyl)-n,n-dimethyl-
40. Allent (salt/mix)
41. (?)-brompheniramine
42. Drauxin (salt/mix)
43. Dimetane (salt/mix)
44. Spectrum_001682
45. Pyridine, 2-(p-bromo-alpha-(2-(dimethylamino)ethyl)benzyl)-
46. Specplus_000750
47. Prestwick0_000475
48. Prestwick1_000475
49. Prestwick2_000475
50. Prestwick3_000475
51. Spectrum2_001610
52. Spectrum3_000981
53. Spectrum4_001107
54. Spectrum5_001537
55. Schembl4814
56. Brompheniramine [mi]
57. Lopac0_000201
58. Oprea1_690847
59. Bspbio_000610
60. Bspbio_002641
61. Kbiogr_001554
62. Kbioss_002162
63. Brompheniramine [hsdb]
64. Divk1c_006846
65. Dexbrompheniramine Maleate Salt
66. Spbio_001619
67. Spbio_002549
68. Brompheniramine [vandf]
69. [3-(4-bromophenyl)-3-(2-pyridyl)propyl]dimethylamine
70. Bpbio1_000672
71. Gtpl7133
72. Brompheniramine [who-dd]
73. Dtxsid5022691
74. Dexbrompheniramine Brompheniramine
75. Hy-b0480a
76. Kbio1_001790
77. Kbio2_002162
78. Kbio2_004730
79. Kbio2_007298
80. Kbio3_001861
81. 3-(4-bromophenyl)-n,n-dimethyl-3-(2-pyridyl)propan-1-amine
82. Hms3604f03
83. A6b359
84. Bdbm50017666
85. Mfcd00865691
86. Pdsp1_000146
87. Pdsp2_000145
88. Stl058559
89. Akos005711313
90. Ccg-204296
91. Db00835
92. Sdccgsbi-0050189.p004
93. Ncgc00015146-04
94. Ncgc00015146-05
95. Ncgc00015146-07
96. Ncgc00015146-08
97. Ncgc00015146-17
98. Ncgc00015146-21
99. Ncgc00089782-02
100. Ac-15955
101. Sbi-0050189.p003
102. Db-056920
103. Ab00053703
104. Cs-0013144
105. Ft-0603299
106. Ft-0772799
107. C06857
108. D07543
109. Ab00053703_19
110. Ab00053703_20
111. L001031
112. Q2606497
113. Brd-a68723818-050-05-1
114. Brd-a68723818-050-18-4
115. [3-(4-bromophenyl)-3-(pyridin-2-yl)propyl]dimethylamine
116. 2-[p-bromo-.alpha.-[2-(dimethylamino)ethyl]benzyl]pyridine
117. Pyridine, 2-[p-bromo-.alpha.-[2-(dimethylamino)ethyl]benzyl]-
118. (6r,7r)-7-amino-8-oxo-3-(1-propenyl)-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid
119. 156428-33-0
| Molecular Weight | 319.24 g/mol |
|---|---|
| Molecular Formula | C16H19BrN2 |
| XLogP3 | 3.5 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 5 |
| Exact Mass | 318.07316 g/mol |
| Monoisotopic Mass | 318.07316 g/mol |
| Topological Polar Surface Area | 16.1 Ų |
| Heavy Atom Count | 19 |
| Formal Charge | 0 |
| Complexity | 249 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Veltane |
| Active Ingredient | Brompheniramine maleate |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 4mg |
| Market Status | Prescription |
| Company | Lannett |
| 2 of 2 | |
|---|---|
| Drug Name | Veltane |
| Active Ingredient | Brompheniramine maleate |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 4mg |
| Market Status | Prescription |
| Company | Lannett |
Anti-Allergic Agents; Histamine H1 Antagonists
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Brompheniramine and dexbrompheniramine share the actions and uses of other antihistamines. Preparations containing brompheniramine maleate or dexbrompheniramine maleate in fixed combination with other agents (e.g., dextromethorphan, guaifenesin, phenylephrine, pseudoephedrine) are used for relief of rhinorrhea, sneezing, lacrimation, itching eyes, oronasopharyngeal itching, and/or other symptoms (e.g., nasal/sinus congestion, cough) associated with seasonal (e.g., hay fever) or perennial (nonseasonal) allergic rhinitis, nonallergic (vasomotor) rhinitis, other upper respiratory allergies, or the common cold. Combination preparations generally should only be used when symptoms amenable to each ingredient are present concurrently.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 9
Like other antihistamines, brompheniramine and dexbrompheniramine should not be used in premature or full-term neonates. Conventional or extended-release preparations of brompheniramine maleate should be used in children younger than 2 or 6 years of age, respectively, only under the direction of a clinician. Brompheniramine maleate should not be used for self-medication in children younger than 6 years of age.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 9
Overdosage and toxicity (including death) have been reported in children younger than 2 years of age receiving nonprescription (over-the-counter, OTC) preparations containing antihistamines, cough suppressants, expectorants, and nasal decongestants alone or in combination for relief of symptoms of upper respiratory tract infection. There is limited evidence of efficacy for these preparations in this age group, and appropriate dosages (i.e., approved by the US Food and Drug Administration (FDA)) for the symptomatic treatment of cold and cough have not been established. Therefore, FDA stated that nonprescription cough and cold preparations should not be used in children younger than 2 years of age; the agency continues to assess safety and efficacy of these preparations in older children. Meanwhile, because children 2-3 years of age also are at increased risk of overdosage and toxicity, some manufacturers of oral nonprescription cough and cold preparations recently have agreed to voluntarily revise the product labeling to state that such preparations should not be used in children younger than 4 years of age. Because FDA does not typically request removal of products with previous labeling from pharmacy shelves during a voluntary label change, some preparations will have the new recommendation ("do not use in children younger than 4 years of age"), while others will have the previous recommendation ("do not use in children younger than 2 years of age"). FDA recommends that parents and caregivers adhere to the dosage instructions and warnings on the product labeling that accompanies the preparation if administering to children and consult with their clinician about any concerns. Clinicians should ask caregivers about use of nonprescription cough and cold preparations to avoid overdosage.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 9
Some patients, especially children, receiving antihistamines may experience paradoxical excitement characterized by restlessness, insomnia, tremors, euphoria, nervousness, delirium, palpitation, and even seizures. /Antihistamine drugs/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2
Adverse effects, which vary in incidence and severity with the individual drug, are caused by all antihistamines, although serious toxicity rarely occurs. Individual patients vary in their susceptibility to the adverse effects of these drugs, and such effects may disappear despite continued therapy. Geriatric patients may be particularly susceptible to dizziness, sedation, and hypotension. Most mild reactions may be relieved by a reduction in dosage or changing to another antihistamine. /Antihistamine drugs/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 2
For more Drug Warnings (Complete) data for BROMPHENIRAMINE (17 total), please visit the HSDB record page.
/CASE REPORTS/ The case history and toxicological findings of an infant fatality involving pseudoephedrine, brompheniramine, and dextromethorphan are presented. Concentrations of brompheniramine ... measured in both postmortem blood and liver /was/ .... 0.40 mg/L in blood and 0.16 mg/kg in liver. ...
PMID:14607011 Boland DM et al; J Anal Toxicol 27 (7): 523-6 (2003)
There is limited information about toxic and lethal concentrations of ... /antihistamines/ in children. .... a case of a pediatric fatality due to a common brompheniramine and phenylpropanolamine preparation /is presented/ and .... available Pediatric Toxicology Registry data on alkylamine antihistamines /were reviewed/. ... A review of the Registry data suggests that postmortem blood brompheniramine concentrations of 0.4 mg/L and greater in children is indicative of brompheniramine poisoning. ...
PMID:9095304 Jumbelic MI et al; Am J Forensic Med Pathol 18 (1): 65-9 (1997)
For the treatment of the symptoms of the common cold and allergic rhinitis, such as runny nose, itchy eyes, watery eyes, and sneezing.
Brompheniramine is an antihistaminergic medication of the propylamine class. It is a first-generation antihistamine. In allergic reactions an allergen interacts with and cross-links surface IgE antibodies on mast cells and basophils. Once the mast cell-antibody-antigen complex is formed, a complex series of events occurs that eventually leads to cell-degranulation and the release of histamine (and other chemical mediators) from the mast cell or basophil. Once released, histamine can react with local or widespread tissues through histamine receptors. Histamine, acting on H1-receptors, produces pruritis, vasodilatation, hypotension, flushing, headache, tachycardia, and bronchoconstriction. Histamine also increases vascular permeability and potentiates pain. Brompheniramine is a histamine H1 antagonist (or more correctly, an inverse histamine agonist) of the alkylamine class. It provides effective, temporary relief of sneezing, watery and itchy eyes, and runny nose due to hay fever and other upper respiratory allergies.
Anti-Allergic Agents
Agents that are used to treat allergic reactions. Most of these drugs act by preventing the release of inflammatory mediators or inhibiting the actions of released mediators on their target cells. (From AMA Drug Evaluations Annual, 1994, p475) (See all compounds classified as Anti-Allergic Agents.)
Histamine H1 Antagonists
Drugs that selectively bind to but do not activate histamine H1 receptors, thereby blocking the actions of endogenous histamine. Included here are the classical antihistaminics that antagonize or prevent the action of histamine mainly in immediate hypersensitivity. They act in the bronchi, capillaries, and some other smooth muscles, and are used to prevent or allay motion sickness, seasonal rhinitis, and allergic dermatitis and to induce somnolence. The effects of blocking central nervous system H1 receptors are not as well understood. (See all compounds classified as Histamine H1 Antagonists.)
R - Respiratory system
R06 - Antihistamines for systemic use
R06A - Antihistamines for systemic use
R06AB - Substituted alkylamines
R06AB01 - Brompheniramine
Absorption
Antihistamines are well absorbed from the gastrointestinal tract after oral administration.
Brompheniramine and dexbrompheniramine maleates appear to be well absorbed from the GI tract.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 9
Distribution of brompheniramine into human body tissues and fluids has not been fully characterized, but the drug appears to be widely distributed. Following oral administration of a single dose of the drug in healthy adults, the apparent volume of distribution reportedly averaged 11.7 L/kg.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 9
Following oral administration of a single 0.13-mg/kg dose of brompheniramine maleate in healthy, fasting adults in one study, peak serum brompheniramine concentrations of 7.7-15.7 ng/mL occurred within 2-5 hours; in most of these individuals, a second lower peak, possibly secondary to enterohepatic circulation, also was observed. The antihistamine effect of brompheniramine, as determined by suppression of the wheal and flare responses induced by intradermal administration of histamine, appears to be maximal within 3-9 hours after a single oral dose of the drug, but suppression of the flare response may persist for up to at least 48 hours; the antipruritic effect appears to be maximal within 9-24 hours.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 10
Brompheniramine and its metabolites are excreted principally in urine. About 40% of an oral dose of brompheniramine is excreted in urine and about 2% in feces within 72 hours in healthy individuals. In healthy individuals, about 5-10% of an oral dose is excreted in urine as unchanged drug ... .
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 10
The pharmacokinetics and antihistaminic effect of brompheniramine in seven normal adults /were assessed/. The mean peak serum brompheniramine concentration of 11.6 +/- 3.0 ng/mL occurred at a mean time of 3.1 +/- 1.1 hr. The mean serum half-life value was 24.9 +/- 9.3 hr, the mean clearance rate was 6.0 +/- 2.3 mL/min/kg, and the mean volume of distribution was 11.7 +/- 3.1 L/kg. The mean wheal size was significantly suppressed (P less than or equal to 0.1) at 3, 6, and 9 hr after the brompheniramine dose when mean concentrations ranged from 10.2 +/- 2.9 to 7.0 +/- 2.2 ng/mL. Significant suppression (P less than or equal to 0.05) of mean flare size was found from 3 to 48 hr after the brompheniramine dose, when mean concentrations ranged from 10.2 +/- 2.9 to 2.5 +/- 0.6 nL/mL. The mean pruritus score was significantly suppressed at 9 and 12 hr (P less than or equal to 0.1) and at 24 hr (P less than or equal to 0.05). Brompheniramine had a long half-life and large volume of distribution in normal adults. It also had a prolonged antihistaminic effect in the skin as evidenced by suppression of the wheal and flare response to histamine and by suppression of pruritus.
PMID:6128358 Simons EE et al; J Allergy Clin Immunol 70 (6): 458-64 (1982)
Hepatic (cytochrome P-450 system), some renal.
The metabolic and excretory fate of the drug has not been fully characterized. Brompheniramine undergoes N-dealkylation to form monodesmethylbrompheniramine and didesmethylbrompheniramine, and is metabolized to the propionic acid derivative, which is partially conjugated with glycine, and to other unidentified metabolites. Brompheniramine and its metabolites are excreted principally in urine. About 40% of an oral dose of brompheniramine is excreted in urine and about 2% in feces within 72 hours in healthy individuals. In healthy individuals, about 5-10% of an oral dose is excreted in urine as unchanged drug, 6-10% as monodesmethylbrompheniramine, 6-10% as didesmethylbrompheniramine, small amounts as the propionic acid derivative and its glycine conjugate, and the remainder as unidentified metabolites.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 10
the pharmacokinetics and antihistaminic effect of brompheniramine in seven normal adults /were assessed/. ... The mean serum half-life value was 24.9 +/- 9.3 hr ...
PMID:6128358 Simons EE et al; J Allergy Clin Immunol 70 (6): 458-64 (1982)
In healthy adults, the half-life of brompheniramine reportedly ranges from 11.8-34.7 hours.
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 10
Brompheniramine is an antagonist of the H1 histamine receptors with moderate antimuscarinic actions, as with other common antihistamines such as diphenhydramine. Due to its anticholindergic effects, brompheniramine may cause drowsiness, sedation, dry mouth, dry throat, blurred vision, and increased heart rate.
H1 antagonists inhibit most of the effects of histamine on smooth muscles, especially the constriction of respiratory smooth muscle. /Histamine Antagonists: H1 Antagonists/
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill, 2006., p. 636
H1 antagonists strongly block the increased capillary permeability and formation of edema and wheal brought about by histamine. /Histamine Antagonists: H1 Antagonists/
Hardman, J.G., L.E. Limbird, P.B., A.G. Gilman. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 11th ed. New York, NY: McGraw-Hill, 2006., p. 637
Antihistamines competitively antagonize most of the smooth muscle stimulating actions of histamine on the H1-receptors of the GI tract, uterus, large blood vessels, and bronchial muscle. Contraction of the sphincter of Oddi and bile duct may be mediated in part by H1-receptors, and opiate-induced contraction of biliary smooth muscle has been antagonized by antihistamines. The drugs only are feebly antagonistic to bronchospasm induced by antigen-antibody reactions. Antihistamines also effectively antagonize the action of histamine that results in increased capillary permeability and the formation of edema. H1-receptor antagonists also suppress flare and pruritus that accompany the endogenous release of histamine. Antihistamines appear to act by blocking H1-receptor sites, thereby preventing the action of histamine on the cell; they do not chemically inactivate or physiologically antagonize histamine nor do they prevent the release of histamine. Antihistamines do not block the stimulating effect of histamine on gastric acid secretion, which is mediated by H2-receptors of the parietal cells. /Antihistamine drugs/
American Society of Health System Pharmacists; AHFS Drug Information 2009. Bethesda, MD. (2009), p. 7
... Affinities of two commonly used over-the-counter antihistamines, brompheniramine and chlorpheniramine, as well as terfenadine in comparison with atropine at the five human muscarinic cholinergic receptor subtypes using CHO cells stably transfected with the individual subtypes /were evaluated/. Atropine was more potent than all three drugs at m1-m5 (p<0.01). No significant difference was observed between chlorpheniramine and brompheniramine. Atropine, brompheniramine, and chlorpheniramine could not discriminate between m1-m5. ...
PMID:10212017 Yasuda SU, Yasuda RP; Pharmacotherapy 19 (4): 447-51 (1999)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
30
PharmaCompass offers a list of Brompheniramine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Brompheniramine manufacturer or Brompheniramine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Brompheniramine manufacturer or Brompheniramine supplier.
PharmaCompass also assists you with knowing the Brompheniramine API Price utilized in the formulation of products. Brompheniramine API Price is not always fixed or binding as the Brompheniramine Price is obtained through a variety of data sources. The Brompheniramine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Brompheniramine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Brompheniramine, including repackagers and relabelers. The FDA regulates Brompheniramine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Brompheniramine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Brompheniramine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Brompheniramine supplier is an individual or a company that provides Brompheniramine active pharmaceutical ingredient (API) or Brompheniramine finished formulations upon request. The Brompheniramine suppliers may include Brompheniramine API manufacturers, exporters, distributors and traders.
click here to find a list of Brompheniramine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Brompheniramine DMF (Drug Master File) is a document detailing the whole manufacturing process of Brompheniramine active pharmaceutical ingredient (API) in detail. Different forms of Brompheniramine DMFs exist exist since differing nations have different regulations, such as Brompheniramine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Brompheniramine DMF submitted to regulatory agencies in the US is known as a USDMF. Brompheniramine USDMF includes data on Brompheniramine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Brompheniramine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Brompheniramine suppliers with USDMF on PharmaCompass.
Brompheniramine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Brompheniramine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Brompheniramine GMP manufacturer or Brompheniramine GMP API supplier for your needs.
A Brompheniramine CoA (Certificate of Analysis) is a formal document that attests to Brompheniramine's compliance with Brompheniramine specifications and serves as a tool for batch-level quality control.
Brompheniramine CoA mostly includes findings from lab analyses of a specific batch. For each Brompheniramine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Brompheniramine may be tested according to a variety of international standards, such as European Pharmacopoeia (Brompheniramine EP), Brompheniramine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Brompheniramine USP).

