
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
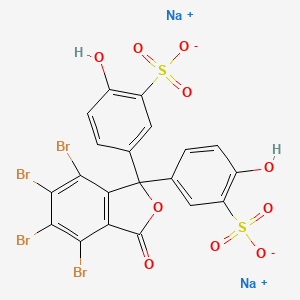

1. Bromosulphthalein
2. Bromsulphalein
3. Bromthalein
4. Disodium, Sulfobromophthalein
5. Sodium, Sulfobromophthalein
6. Sulfobromophthalein
7. Sulfobromophthalein Disodium
8. Sulfobromophthalein Sodium
9. Tetrabromsulphthalein
1. Bromosulfalein
2. 71-67-0
3. Bromsulphalein
4. Bromthalein
5. Bromosulphthalein
6. Hepatestabrome
7. Hepatosulfalein
8. Hepartest
9. Bromsulphthalein
10. Bsp Sodium
11. Bromsulfalein
12. Sodium Bromosulfalein
13. Sodium Bromsulphalein
14. Sodium Bromsulphthalein
15. Sodium Bromosulfophthalein
16. Sodium Sulphobromophthalein
17. Sulphobromophthalein Sodium
18. Disodium Bromosulfophthalein
19. Tetrabromophenolsulfophthalein
20. Sodium Phenol Tetrabromophthalein
21. Sodium Sulfobromophthalein
22. Sulfobromophthalein Disodium Salt
23. Disodium Phenoltetrabromophthalein Sulfonate
24. Chebi:63827
25. 4,5,6,7-tetrabromo-3',3''-disulfophenolphthalein Disodium Salt
26. Dsstox_cid_3621
27. Dsstox_rid_77114
28. Dsstox_gsid_23621
29. Bromsulfthalein
30. Hepartestabrome
31. Bromotaleina
32. Brom-tetragnost
33. Bromsulfophthalein
34. Bsf Simes
35. Cas-71-67-0
36. Benzenesulfonic Acid, 3,3'-(4,5,6,7-tetrabromo-3-oxo-1(3h)-isobenzofuranylidene)bis(6-hydroxy-, Disodium Salt
37. Benzenesulfonic Acid, 3,3'-(4,5,6,7-tetrabromo-3-oxo-1(3h)-isobenzofuranylidene)bis[6-hydroxy-, Disodium Salt
38. Bsf
39. Disodium 3,3'-(4,5,6,7-tetrabromo-3-oxo-1,3-dihydro-2-benzofuran-1,1-diyl)bis(6-hydroxybenzenesulfonate)
40. Cbsp
41. Bromsulphalein R Sodium
42. Ncgc00159391-02
43. Ncgc00159391-03
44. Unii-62e5ju30ov
45. Hsdb 2183
46. Disodium 2-hydroxy-5-[4,5,6,7-tetrabromo-1-(4-hydroxy-3-sulfonatophenyl)-3-oxo-1,3-dihydro-2-benzofuran-1-yl]benzene-1-sulfonate
47. Einecs 200-761-0
48. Sulfobromophthalein Sodium [usp:jan]
49. Bromsulphthalein Sodium
50. Bromosulfophthalein Sodium
51. [3h]bsp
52. Disodium 6,6'-dihydroxy-3,3'-(4,5,6,7-tetrabromo-1,3-dihydro-3-oxoisobenzofuran-1-ylidene)dibenzenesulphonate
53. Schembl487073
54. [3h]-bsp
55. Chembl453904
56. Gtpl4505
57. Gtpl4506
58. Bromosulfophthalein Disodium Salt
59. Dtxsid9023621
60. Sulfobromophthalein Sodium (jp17)
61. Sulfobromophthaleinsodiumtetrahydrate
62. Tox21_111628
63. Akos024373601
64. Tox21_111628_1
65. 5,5'-(4,5,6,7-tetrabromo-3-oxo-1(3h)-isobenzofuranylidene)bis(2-hydroxybenzenesulfonic Acid) Disodium Salt
66. Ft-0643010
67. D02210
68. Q926998
69. 4,5,6,7-tetrabromo-3',3'-disulfophenolphthalein Disodium Salt
| Molecular Weight | 838.0 g/mol |
|---|---|
| Molecular Formula | C20H8Br4Na2O10S2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 2 |
| Exact Mass | 837.60468 g/mol |
| Monoisotopic Mass | 833.60878 g/mol |
| Topological Polar Surface Area | 198 Ų |
| Heavy Atom Count | 38 |
| Formal Charge | 0 |
| Complexity | 994 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
Indicators and Reagents; Phthalein Dyes
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
FORTY-FIVE MIN AFTER INJECTION...4-5 ML OF BLOOD IS WITHDRAWN FROM OPPOSITE ARM TO AVOID CONTAMINATION OF SAMPLE. ...SOME CLINICIANS TAKE SINGLE SAMPLE @ 30 MIN & OTHERS @ 60 MIN. AMT OF DYE RETAINED IN THE SERUM IS DETERMINED COLORIMETRICALLY.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
USE...IN HEPATIC FUNCTION DETERMINATION IS NOT BASED ON ITS PHARMACOLOGIC EFFECTS BUT ON ITS DISTRIBUTION, METABOLISM, & EXCRETION FROM BODY.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
...HELPFUL IN DETECTING HEPATIC DAMAGE IN PT WHO HAVE BEEN EXPOSED TO HEPATOTOXINS & IN RECOGNIZING METASTATIC CARCINOMA. ...ALSO BEEN USED TO DISTINGUISH UPPER GI HEMORRHAGE DUE TO PEPTIC ULCER FROM THAT DUE TO ESOPHAGEAL VARICES & FOR DIAGNOSIS OF DUBIN-JOHNSON OR ROTOR SYNDROMES. ...USED TO EST HEPATIC BLOOD FLOW...
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
For more Therapeutic Uses (Complete) data for SODIUM SULFOBROMOPHTHALEIN (8 total), please visit the HSDB record page.
HIGH BLOOD LEVELS OF BILIRUBIN MAY INTERFERE WITH MEASUREMENT OF SULFOBROMOPHTHALEIN... HEPARIN...INCR COLOR INTENSITY...IN SERUM & CAUSES SHIFT IN ABSORPTION... ... SULFOBROMOPHTHALEIN MAY PRODUCE...INCR IN OTHER LABORATORY TEST RESULTS...SERUM CALCIUM, SERUM CREATINE & CREATININE, UROBILINOGEN, BLOOD PROTEIN...
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
PARENTERAL CHOLECYSTOGRAPHIC AGENTS ARE SATURATED SOLN, & PT ALMOST INVARIABLY EXPERIENCE THROMBOPHLEBITIS IF SULFOBROMOPHTHALEIN IS INJECTED INTO SAME ARM WITHIN FEW DAYS FOLLOWING INJECTION OF RADIOPAQUE SUBSTANCE.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
VET WARNING: IRRITANT TO VEINS. USE DILUTE SOLN VIA SMALL GAUGE NEEDLES OR CANNULAS. ... ICTERIC STATES PREVENT INTERPRETATION OF TEST.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 578
...USED WITH EXTREME CAUTION, IF @ ALL, IN PT WHO HAVE HAD PREVIOUS SULFOBROMOPHTHALEIN TESTS & IN PT WITH HISTORY OF ALLERGY OR ASTHMA... COMPLETE MEDICAL HISTORY...SHOULD BE OBTAINED BEFORE DYE IS ADMIN.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
For more Drug Warnings (Complete) data for SODIUM SULFOBROMOPHTHALEIN (7 total), please visit the HSDB record page.
Indicators and Reagents
Substances used for the detection, identification, analysis, etc. of chemical, biological, or pathologic processes or conditions. Indicators are substances that change in physical appearance, e.g., color, at or approaching the endpoint of a chemical titration, e.g., on the passage between acidity and alkalinity. Reagents are substances used for the detection or determination of another substance by chemical or microscopical means, especially analysis. Types of reagents are precipitants, solvents, oxidizers, reducers, fluxes, and colorimetric reagents. (From Grant and Hackh's Chemical Dictionary, 5th ed, p301, p499) (See all compounds classified as Indicators and Reagents.)
Coloring Agents
Chemicals and substances that impart color including soluble dyes and insoluble pigments. They are used in INKS; PAINTS; and as INDICATORS AND REAGENTS. (See all compounds classified as Coloring Agents.)
WHEN HEPATIC FUNCTION IS SEVERELY IMPAIRED, UP TO 10-25%...MAY BE EXCRETED IN URINE. NORMALLY...LESS THAN 2% OF DOSE IS EXCRETED IN THIS WAY, & EXTRAHEPATIC REMOVAL OF SULFOBROMOPTHALEIN CAN BE DISREGARDED.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
NORMAL METABOLISM & EXCRETION OF SULFOBROMOPHTHALEIN DEPEND ON HEPATIC BLOOD FLOW, ABILITY OF LIVER CELLS TO TAKE UP & CONCN THE DYE, & INTAKE BILIARY EXCRETION.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
...METABOLIZED IN LIVER BY CONJUGATION WITH GLUTATHIONE, & BOTH FREE & CONJUGATED...ARE EXCRETED INTO BILE. ... DYE DOES NOT UNDERGO ENTEROHEPATIC CIRCULATION...& IS EXCRETED PRIMARILY IN FECES. ... EXCRETION OF DYE IS USUALLY COMPLETE WITHIN 24 HR.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
FOLLOWING IV ADMIN...ALMOST COMPLETELY BOUND TO PLASMA PROTEINS IN BLOOD, MAINLY ALBUMIN & ALPHA1 LIPOPROTEINS. DYE IS RAPIDLY TAKEN UP & BRIEFLY STORED BY HEPATIC PARENCHYMAL CELLS. ... SMALL AMT...MAY BE TAKEN UP BY SKELETAL MUSCLE, BUT EXTRA-HEPATIC TISSUE UPTAKE IS MINIMAL. ...DOES NOT CROSS PLACENTA.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
For more Absorption, Distribution and Excretion (Complete) data for SODIUM SULFOBROMOPHTHALEIN (6 total), please visit the HSDB record page.
S-ARYLTRANSFERASE IS THOUGHT TO BE INVOLVED IN METABOLISM OF SULFOBROMOPHTHALEIN...
The Chemical Society. Foreign Compound Metabolism in Mammals. Volume 2: A Review of the Literature Published Between 1970 and 1971. London: The Chemical Society, 1972., p. 368
IT IS EXCRETED IN BILE OF RATS AS BSP-GLUTATHIONE & OTHER AMINOACYL CONJUGATES, FORMED BY CONDENSATION OF BSP WITH GLUTATHIONE, PROBABLY WITHOUT ELIMINATION OF ANY OF BROMINE ATOMS.
Parke, D. V. The Biochemistry of Foreign Compounds. Oxford: Pergamon Press, 1968., p. 25
SULFOBROMOPHTHALEIN /WAS METABOLIZED TO THE FOLLOWING/ S-(3,3-BIS(4-HYDROXY-3-SULFOPHENYL) TRIBROMOPHTHALIDYL)GLUTATHIONE IN DOG; JAVITT NB, ET AL, J CLIN INVEST, 39, 1570 (1960). SULFOBROMOPHTHALEIN-BETA-D-GLUCURONIDE IN LIVER; HIRAYAMA C, ET AL, SAISHIN IGAKU, 17, 2520 (1962).
Goodwin, B.L. Handbook of Intermediary Metabolism of Aromatic Compounds. New York: Wiley, 1976., p. S-15
IN ONE STUDY IN SMALL NUMBER OF NORMAL SUBJECTS, PLASMA HALF-LIFE AVG ABOUT 4 MIN. LIVER HAS STORAGE CAPACITY...WHICH IS PROPORTIONAL TO PLASMA CONCN OF DYE, & RANGES FROM 50-75 MG OF SULFOBROMOPHTHALEIN...PER MG OF DYE PER 100 ML OF PLASMA IN PT WITH NORMAL LIVER FUNCTION.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
SULFOBROMOPHTHALEIN TEST FOR LIVER FUNCTION IS BASED ON THE FACT THAT IN PT WITH HEPATIC DYSFUNCTION, PERCENTAGE DISAPPEARANCE RATE & EST STORAGE CAPACITY...ARE DECR & PLASMA HALF-LIFE & EST EXCRETORY TIME OF DYE ARE INCR.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 36:44
Global Sales Information

ABOUT THIS PAGE
80
PharmaCompass offers a list of Bromthalein API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Bromthalein manufacturer or Bromthalein supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Bromthalein manufacturer or Bromthalein supplier.
PharmaCompass also assists you with knowing the Bromthalein API Price utilized in the formulation of products. Bromthalein API Price is not always fixed or binding as the Bromthalein Price is obtained through a variety of data sources. The Bromthalein Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Bromthalein manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Bromthalein, including repackagers and relabelers. The FDA regulates Bromthalein manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Bromthalein API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Bromthalein supplier is an individual or a company that provides Bromthalein active pharmaceutical ingredient (API) or Bromthalein finished formulations upon request. The Bromthalein suppliers may include Bromthalein API manufacturers, exporters, distributors and traders.
Bromthalein Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Bromthalein GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Bromthalein GMP manufacturer or Bromthalein GMP API supplier for your needs.
A Bromthalein CoA (Certificate of Analysis) is a formal document that attests to Bromthalein's compliance with Bromthalein specifications and serves as a tool for batch-level quality control.
Bromthalein CoA mostly includes findings from lab analyses of a specific batch. For each Bromthalein CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Bromthalein may be tested according to a variety of international standards, such as European Pharmacopoeia (Bromthalein EP), Bromthalein JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Bromthalein USP).
