
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
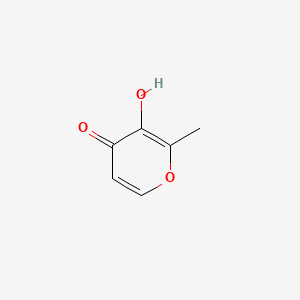

1. 3-hydroxy-2-methyl-4-pyrone
1. 118-71-8
2. 3-hydroxy-2-methyl-4-pyrone
3. 3-hydroxy-2-methyl-4h-pyran-4-one
4. Larixinic Acid
5. Palatone
6. Talmon
7. Larixic Acid
8. Vetol
9. Veltol
10. Corps Praline
11. 3-hydroxy-2-methylpyran-4-one
12. 4h-pyran-4-one, 3-hydroxy-2-methyl-
13. 2-methyl Pyromeconic Acid
14. 2-methylpyromeconic Acid
15. 2-methyl-3-hydroxy-4-pyrone
16. 2-methyl-3-hydroxypyrone
17. Maltol (natural)
18. 3-hydroxy-2-methyl-gamma-pyrone
19. 2-methyl-3-oxy-gamma-pyrone
20. Fema No. 2656
21. 3-hydroxy-2-methylpyrone
22. 3-hydroxy-2-methyl-pyran-4-one
23. Mfcd00006578
24. Nsc 2829
25. 2-methyl-3-hydroxy-4-pyranone
26. 3-hydroxy-2-methyl-4-pyranone
27. Chebi:69438
28. Nsc2829
29. 3-hydroxy-2-methyl-1,4-pyrone
30. Ins No.636
31. Nsc-2829
32. 3-hydroxy-2-methyl-.gamma.-pyrone
33. Nsc-404458
34. Ins-636
35. Mls000069412
36. 3a9rd92bs4
37. 5-hydroxy-6-methyl-4h-pyran-4-one
38. Maltol (3-hydroxy-2-methyl-4-pyrone)
39. E636
40. Smr000059093
41. E-636
42. Dsstox_cid_5523
43. Dsstox_rid_77818
44. Dsstox_gsid_25523
45. Wln: T6o Dvj B1 Cq
46. Cas-118-71-8
47. Maltol [nf]
48. Ccris 3467
49. Einecs 204-271-8
50. Brn 0112169
51. Unii-3a9rd92bs4
52. Methylmaltol
53. Methyl Maltol
54. Laricinic Acid
55. Ai3-18547
56. Natural Maltol
57. Spectrum_001419
58. Opera_id_338
59. Specplus_000443
60. Maltol [fhfi]
61. Maltol [inci]
62. Maltol [fcc]
63. Maltol [usp-rs]
64. Spectrum2_001795
65. Spectrum3_001351
66. Spectrum4_001871
67. Spectrum5_000462
68. Maltol [ii]
69. Maltol [mi]
70. Maltol [mart.]
71. Bmse000538
72. Maltol, Analytical Standard
73. Schembl4815
74. 3-hydroxy-2-pyran-4-one
75. Bspbio_003161
76. Kbiogr_002365
77. Kbioss_001899
78. Spectrum310025
79. 5-18-01-00114 (beilstein Handbook Reference)
80. Mls001424145
81. Mls002415738
82. 3-hydroxy-2-methyl-g-pyrone
83. Chembl31422
84. Divk1c_006539
85. 3-hydroxy-2-methylpyr-4-one
86. Spbio_001749
87. Qspl 180
88. Dtxsid0025523
89. 2-methyl-3-oxy-.gamma.-pyrone
90. 3-hydroxy-2-methyl-4-oxopyrane
91. 3-hydroxyl-2-methyl-4-pyranone
92. Fema 2656
93. Hsdb 8320
94. Kbio1_001483
95. Kbio2_001899
96. Kbio2_004467
97. Kbio2_007035
98. Kbio3_002381
99. Xpctzqvdejyugt-uhfffaoysa-
100. 3-hydroxy-2-methyl-gamma -pyrone
101. Hms2052k09
102. Hms3394k09
103. Kuc106764n
104. Zinc164488
105. Str01642
106. Tox21_202215
107. Tox21_300118
108. Bbl011669
109. Bdbm50227434
110. Ccg-38443
111. Maltol, Natural, >=98.5%, Fg
112. Nsc404458
113. S4940
114. Stk801686
115. 2-methyl-3-hydroxy-4h-pyran-4-one
116. Maltol, >=99.0%, Fcc, Fg
117. 3-hydroxy-2-methyl-4-pyrone, 99%
118. Akos005607790
119. 3-hydroxy-2-methyl-4-pyrone, Natural
120. Cs-w013504
121. Hy-w012788
122. Nc00350
123. Ps-4578
124. Sdccgmls-0066563.p001
125. 4-(a-d-glucopyranosido)-a-glucopyranose
126. Ncgc00091223-01
127. Ncgc00091223-02
128. Ncgc00091223-03
129. Ncgc00091223-04
130. Ncgc00091223-05
131. Ncgc00178231-01
132. Ncgc00254046-01
133. Ncgc00259764-01
134. Bp-11468
135. Ksc-11-228-8
136. Nci60_002320
137. Sy011358
138. Db-002512
139. Am20080119
140. Ft-0615804
141. M0673
142. Maltol Solution, 1 Wt. % In Benzyl Alcohol
143. A804081
144. Q420648
145. Sr-01000712383
146. Sr-01000712383-3
147. W-108539
148. Brd-k40619305-001-12-1
149. Z1868163292
150. Maltol, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 126.11 g/mol |
|---|---|
| Molecular Formula | C6H6O3 |
| XLogP3 | 0.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 126.031694049 g/mol |
| Monoisotopic Mass | 126.031694049 g/mol |
| Topological Polar Surface Area | 46.5 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 200 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
/EXPL THER/ /The objective of the study was/ to evaluate the neuroprotective and neurite outgrowth effects of maltol, a natural aroma compound, on retinal ganglion cells (RGCs) under oxidative stress in vitro. Mouse primary RGCs were isolated using immunopanning-magnetic separation and exposed to H2O2 in the presence of maltol. The cell viability and apoptosis were determined by using adenosine 5'-triphosphate (ATP) assay and terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate (dUTP) nick end labeling (TUNEL), respectively. Neurite outgrowth was assessed by immunofluorescence for alpha-tubulin. The activation of nuclear factor-kappaB (NF-kappaB) was also evaluated using immunofluorescence. When the RGCs were exposed to 20 uM of H2O2 for 16 hr, their viability dropped to 40.3 +/- 3.4%. However, the maltol treatment restored the cells in a dose-dependent manner. The viability recovered to 73.9 +/- 5.1% with 10 uM of maltol and even reached 175.1 +/- 11.3% with 2 mM of maltol, as measured by ATP assay. This oxidative stress significantly increased the number of TUNEL-positive RGCs, but the maltol drastically reduced the proportion of those apoptotic cells. The oxidative stress hampered the neurite outgrowth of the RGCs, whereas maltol restored their ability to sprout neurites. Regarding NF-kappaB, the active form of phosphorylated NF-kappaB (pNF-kappaB) increased the oxidative stress level but the maltol treatment again reduced it to an unstressful level. Our data revealed that maltol attenuated the oxidative stress-induced injury in the primary mouse RGCs. Its neuroprotective and neurite outgrowth effects seemed to be related to NF-kappaB signaling. Maltol has potential as a new neuroprotective therapeutic agent for oxidative stress-related ocular diseases, including glaucoma.
PMID:25352751 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4203576 Hong S et al; Mol Vis 20: 1456-62 (2014)
Groups of two beagle dogs of each sex were given a single intravenous injection of 10 mg/kg bw maltol, and urine samples were collected for 72 hr. An average of 58.5% of the administered dose was excreted as a mixture of sulfate and glucuronic acid conjugates of maltol. About 98% of the total urinary excretion of conjugates occurred within the first 24 hr, males and females excreting an average of 42% and 73% of the administered dose, respectively.
WHO/JEFCA; WHO Food Additives Series 56; Maltol and Related Substances p. 83-4 (2006). Available from, as of May 26, 2016: https://www.inchem.org/pages/jecfa.html
Maltol and derivatives contain a gamma-pyrone ring system. Gamma-pyrones are relatively basic, and the behavior as a base is partly due to the aromatic character and relative stability of the conjugate acid. As the gamma-pyrone ring also contains a 3-hydroxy substituent, it is expected that maltol and its derivatives will be readily conjugated with glucuronic acid or sulfate. In addition, maltol may form a complex with metal ions (e.g. Fe++), like phenols.
WHO/JEFCA; WHO Food Additives Series 56; Maltol and Related Substances p. 83 (2006). Available from, as of May 26, 2016: https://www.inchem.org/pages/jecfa.html
Maltol (3-hydroxy-2-methyl-4-pyrone) produced reactive oxygen species as a complex with transition metals. Maltol/iron complex inactivated aconitase the most sensitive enzyme to oxidative stress. The inactivation of aconitase was iron-dependent, and prevented by TEMPOL, a scavenger of reactive oxygen species, suggesting that the maltol/iron-mediated generation of superoxide anion is responsible for the inactivation of aconitase. Addition of maltol effectively enhanced the ascorbate/copper-mediated formation of 8-hydroxy-2'-deoxyguanosine in DNA. Oxidation of ascorbic acid by CuSO(4) was effectively stimulated by addition of maltol, and the enhanced oxidation rate was markedly inhibited by the addition of catalase and superoxide dismutase. These results suggest that maltol can stimulate the copper reduction coupled with the oxidation of ascorbate, resulting in the production of superoxide radical which in turn converts to hydrogen peroxide and hydroxyl radical. Cytotoxic effect of maltol can be explained by its prooxidant properties: maltol/transition metal complex generates reactive oxygen species causing the inactivation of aconitase and the production of hydroxyl radical causing the formation of DNA base adduct.
PMID:16799863 Murakami K et al; Biometals 19(3):253-7 (2006)
... We examined the ability of maltol to induce the cytochrome P450 1a1 (Cyp1a1), an enzyme known to play an important role in the chemical activation of xenobiotics to carcinogenic derivatives. Our results showed that treatment of Hepa 1c1c7 cells with maltol significantly induced Cyp1a1 at mRNA, protein, and activity levels in a concentration-dependent manner. The RNA synthesis inhibitor, actinomycin D, completely blocked the Cyp1a1 mRNA induction by maltol, indicating a requirement of de novo RNA synthesis through transcriptional activation. In addition, maltol induced aryl hydrocarbon receptor (AhR)-dependent luciferase reporter gene expression in stably transfected H1L1.1c2 cells, suggesting an AhR-dependent mechanism. This is the first demonstration that the food flavoring agent, maltol, can directly induce Cyp1a1 gene expression in an AhR-dependent manner and represents a novel mechanism by which maltol promotes carcinogenicity and toxicity.
PMID:17317091 Anwar-Mohamed A, El-Kadi AO; Toxicol In Vitro 21 (4): 685-90 (2007)
Maltol has antioxidant properties, presumably through its ability to complex metal ions such as Fe++ and to promote the formation of reduced glutathione (GSH). Maltol at a concentration of 130 umol/L inhibited iron-mediated lipid peroxidation and increased scavenging of reactive oxygen species by enhancing the supply of NADPH required for regeneration of GSH. Maltol inhibited the formation of thiobarbituric acid-reactive substances when incubated with rat liver microsomes in the presence of Fe++ and ascorbate. Maltol at concentrations of 130-140 umol/L also effectively inhibited the inactivation of NADP-isocitrate dehydrogenase, the principal NADPH-generating enzyme, by Fe++. Maltol significantly increased the oxidation of Fe++, while dimethylpyrone had no effect. The latter results suggest that the 3-hydroxy substituent in maltol is necessary to promote Fe++ oxidation.
WHO/JEFCA; WHO Food Additives Series 56; Maltol and Related Substances p. 84 (2006). Available from, as of May 26, 2016: https://www.inchem.org/pages/jecfa.html
Global Sales Information

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
87
PharmaCompass offers a list of Maltol API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Maltol manufacturer or Maltol supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Maltol manufacturer or Maltol supplier.
PharmaCompass also assists you with knowing the Maltol API Price utilized in the formulation of products. Maltol API Price is not always fixed or binding as the Maltol Price is obtained through a variety of data sources. The Maltol Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A CAS-118-71-8 manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of CAS-118-71-8, including repackagers and relabelers. The FDA regulates CAS-118-71-8 manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. CAS-118-71-8 API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A CAS-118-71-8 supplier is an individual or a company that provides CAS-118-71-8 active pharmaceutical ingredient (API) or CAS-118-71-8 finished formulations upon request. The CAS-118-71-8 suppliers may include CAS-118-71-8 API manufacturers, exporters, distributors and traders.
CAS-118-71-8 Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of CAS-118-71-8 GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right CAS-118-71-8 GMP manufacturer or CAS-118-71-8 GMP API supplier for your needs.
A CAS-118-71-8 CoA (Certificate of Analysis) is a formal document that attests to CAS-118-71-8's compliance with CAS-118-71-8 specifications and serves as a tool for batch-level quality control.
CAS-118-71-8 CoA mostly includes findings from lab analyses of a specific batch. For each CAS-118-71-8 CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
CAS-118-71-8 may be tested according to a variety of international standards, such as European Pharmacopoeia (CAS-118-71-8 EP), CAS-118-71-8 JP (Japanese Pharmacopeia) and the US Pharmacopoeia (CAS-118-71-8 USP).
