
Synopsis
Synopsis
0
JDMF
0
NDC API
0
VMF
0
Europe

0
Canada

0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Anspor
2. Cefradine
3. Cephradine Dihydrate
4. Cephradine, Non Stoichiometric Hydrate
5. Cephradine, Non-stoichiometric Hydrate
6. Dexef
7. Dihydrate, Cephradine
8. Kelsef
9. Maxisporin
10. Nicef
11. Non-stoichiometric Hydrate Cephradine
12. Sefril
13. Septacef
14. Sq 11436
15. Sq-11436
16. Sq11436
17. Velocef
18. Velosef
19. Zeefra G
1. Cefradine
2. 38821-53-3
3. Cephradin
4. Anspor
5. Sefril
6. Velosef
7. Cefradina
8. Cefradinum
9. Eskacef
10. Sq 11436
11. Cefradin
12. Sq-11436
13. (6r,7r)-7-((r)-2-amino-2-(1,4-cyclohexadien-1-yl)acetamido)-3-methyl-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid
14. Cefradine [inn]
15. Cephradine Anhydrous
16. 7-(d-2-amino-2-(1,4-cyclohexadienyl)acetamide)desacetoxycephalosporanicacid
17. Chebi:3547
18. (6r,7r)-7-[[(2r)-2-amino-2-cyclohexa-1,4-dien-1-ylacetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
19. 9ya6sx5s4d
20. (6r,7r)-7-{[(2r)-2-amino-2-cyclohexa-1,4-dien-1-ylacetyl]amino}-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
21. Nsc-756672
22. Ced
23. Cephradine Hydrate
24. Ncgc00183036-01
25. Dsstox_cid_2785
26. Cefradinum [inn-latin]
27. Cephradine [usan:ban]
28. Dsstox_rid_76729
29. Cefradina [inn-spanish]
30. Dsstox_gsid_22785
31. (6r,7r)-7-[(2r)-2-amino-2-(cyclohexa-1,4-dien-1-yl)acetamido]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
32. (7r)-7-[[(2r)-2-amino-2-cyclohexa-1,4-dien-1-ylacetyl]amino]-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
33. Cekodin
34. Megacef
35. Velocef
36. Megace F
37. Skf D 39304
38. Cephradine (usp)
39. Velosef (tn)
40. Velosef '125'
41. Velosef '250'
42. Velosef '500'
43. Anspor (tn)
44. Cas-38821-53-3
45. Hsdb 3216
46. Cefradine (jan/inn)
47. Einecs 254-137-8
48. Unii-9ya6sx5s4d
49. Brn 6075388
50. Infexin
51. Cephradine,(s)
52. (6r,7r)-7-(((2r)-2-amino-2-cyclohexa-1,4-dien-1-ylacetyl)amino)-3-methyl-8-oxo-5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid
53. (6r,7r)-7-((r)-2-amino-2-(cyclohexa-1,4-dienyl)acetamido)-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
54. Mfcd00865048
55. Sk&f D-39304
56. Sk-d-39304
57. 5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid, 7-((amino-1,4-cyclohexadien-1-ylacetyl)amino)-3-methyl-8-oxo-, (6r-(6alpha,7beta(r*)))-
58. 7-(d-2-amino-2-(1,4-cyclohexadien-1-yl)acetamido)-3-methyl-8-oxo-5-thia-1-azabicyclo(4.2.0)-oct-2-ene-2-caboxylic Acid
59. Sq-22022
60. Cefradine Hydrate
61. Cefradine [jan]
62. Cephradine [mi]
63. Cephradine [hsdb]
64. Cefradine [mart.]
65. Epitope Id:116210
66. Cefradine [who-dd]
67. Schembl3244
68. Chembl1604
69. Divk1c_000739
70. Cephradine, Unspecified Hydrate
71. Amy500
72. Gtpl4830
73. Cefradine [ep Monograph]
74. Dtxsid4022785
75. Hms502e21
76. Kbio1_000739
77. Ninds_000739
78. Bcpp000291
79. Sq 22022 [dihydrate]
80. Hy-b1156
81. Zinc3830515
82. Tox21_113488
83. Bdbm50370585
84. S4671
85. Akos015961130
86. Tox21_113488_1
87. Ac-1409
88. Bcp9000506
89. Ccg-208520
90. Cs-4608
91. Db01333
92. Nsc 756672
93. Idi1_000739
94. Cefradine 100 Microg/ml In Acetonitrile
95. Ncgc00183036-02
96. Ncgc00263658-01
97. 7beta-[(2r)-2-(cyclohexa-1,4-dienyl)-2-phenylacetamido]-3-methyl-3,4-didehydrocepham-4-carboxylic Acid
98. As-75158
99. C06897
100. C74975
101. D00264
102. 821c533
103. Q2734674
104. Cefradine, European Pharmacopoeia (ep) Reference Standard
105. Cefradine For Peak Identification, European Pharmacopoeia (ep) Reference Standard
106. (6r,7r)-7-((r)-2-amino-2-(cyclohexa-1,4-dien-1-yl)acetamido)-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid
107. (6r,7r)-7-((r)-2-amino-2-(cyclohexa-1,4-dien-1-yl)acetamido)-3-methyl-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylicacid
108. 5-thia-1-azabicyclo(4.2.0)oct-2-ene-2-carboxylic Acid, 7-((amino-1,4-cyclohexadien-1-ylacetyl)amino)-3-methyl-8-oxo-, (6r-(6.alpha.,7.beta.(r*)))-
109. 5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid,7-[[(2r)-2-amino-2-(1,4-cyclohexadien-1-yl)acetyl]amino]-3-methyl-8-oxo-,(6r,7r)-
110. 5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic Acid,7-[[(2r)-amino-1,4-cyclohexadien-1-ylacetyl]amino]-3-methyl-8-oxo-,(6r,7r)-
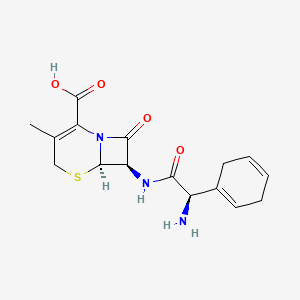
| Molecular Weight | 349.4 g/mol |
|---|---|
| Molecular Formula | C16H19N3O4S |
| XLogP3 | 0.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Exact Mass | 349.10962727 g/mol |
| Monoisotopic Mass | 349.10962727 g/mol |
| Topological Polar Surface Area | 138 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 697 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cephalosporins
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Cephradine /is/ indicated in the treatment of bacterial urinary tract infections caused by susceptible organisms. /Included in US product labeling/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 768
Cephradine /is/ indicated in the treatment of bacterial pharyngitis caused by susceptible organisms. /Included in US product labeling/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 768
Cephradine /is/ indicated in the treatment of skin and soft tissue infections caused by susceptible organisms. /Included in US product labeling/
USP Convention. USPDI - Drug Information for the Health Care Professional. 16th ed. Volume I. Rockville, MD: U.S. Pharmaceutical Convention, Inc. 1996 (Plus updates)., p. 268
For more Therapeutic Uses (Complete) data for CEPHRADINE (18 total), please visit the HSDB record page.
Cephalosporins should be used with caution in patients with a history of GI disease, particularly colitis. Because antibiotic-associated pseudomembranous colitis has been reported with the use of cephalosporins, it should be considered in the differential diagnosis of patients who develop diarrhea during or following therapy with the drugs. /Cephalosporins/
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 109
Prolonged use of a cephalosporin may result in the overgrowth of nonsusceptible organisms, especially Enterobacter, Pseudomonas, enterococci, or Candida. If superinfection occurs, appropriate therapy should be instituted. /Cephalosporins/
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 109
Other adverse effects reported with cephalosporin therapy include chest pain, pleural effusion, dyspnea or respiratory distress, cough, and rhinitis. Increased or decreased serum glucose concentration also has been reported. /Cephalosporins/
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 109
The most frequent adverse reactions to orally administered cephalosporins are nausea, vomiting, and diarrhea. These effects are usually mild and transient, but rarely may be severe enough to require discontinuance of the drug. Other adverse GI effects which have occurred with some of the oral cephalosporins include abdominal pain, tenesmus, epigastric pain/dyspepsia, decreased appetite/anorexia, glossitis, flatulence, candidiasis (eg, oral thrush), taste alteration, decreased salivation, and heartburn. Adverse GI effects can also occur with IM or IV cephalosporins. /Cephalosporins/
McEvoy G.K. (ed.). American Hospital Formulary Service-Drug Information 96. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 1996 (Plus Supplements)., p. 109
For more Drug Warnings (Complete) data for CEPHRADINE (9 total), please visit the HSDB record page.
Anti-Bacterial Agents
Substances that inhibit the growth or reproduction of BACTERIA. (See all compounds classified as Anti-Bacterial Agents.)
J - Antiinfectives for systemic use
J01 - Antibacterials for systemic use
J01D - Other beta-lactam antibacterials
J01DB - First-generation cephalosporins
J01DB09 - Cefradine
Route of Elimination
Over 90 percent of the drug is excreted unchanged in the urine within six hours.
CEPHRADINE IS ALMOST COMPLETELY ABSORBED AFTER ORAL ADMIN; IN PRESENCE OF FOOD, ABSORPTION IS SLOWED BUT NOT DECR. ORAL DOSES OF 250 & 500 MG YIELD PEAK PLASMA LEVELS OF ABOUT 9 & 16.5 UG/ML, WITHIN 40 MIN FROM EMPTY STOMACH. ABSORPTION BY IM ROUTE IS CONSIDERABLY SLOWER.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 1171
AFTER SINGLE ORAL ADMIN, CEPHRADINE WAS ABSORBED RAPIDLY FROM GI TRACT & PEAK SERUM CONCN ACHIEVED WITHIN 1 HR. 75-100% OF DOSE EXCRETED UNCHANGED IN URINE IN FIRST 6 HOURS.
PMID:4591487 ZAKI A ET AL; J CLIN PHARMACOL 14: 118-26 (1974)
SIX SUBJECTS RECEIVED 1 G EACH OF CEPHRADINE (I) & CEPHALEXIN (II) EVERY 4 HR FOR 7 DOSES & 5 RECEIVED 2 G EVERY 6 HR FOR 5 DOSES. PEAK SERUM LEVELS FOR I & II WERE 29 & 35 UG/ML, RESPECTIVELY, FOLLOWING 1 G & 45-50 UG/ML FOLLOWING 2 G.
PMID:438352 CHOW M ET AL; J CLIN PHARMACOL 19: 185-194 (1979)
Cephalosporins are primarily excreted by the kidney ... . /Cephalosporins/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 1088
For more Absorption, Distribution and Excretion (Complete) data for CEPHRADINE (18 total), please visit the HSDB record page.
Cephradine is not metabolized and, after rapid absorption from the gastrointestinal tract, is excreted unchanged in the urine.
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 1089
Cefradine is a first generation cephalosporin antibiotic with a spectrum of activity similar to Cefalexin. Cefradine, like the penicillins, is a beta-lactam antibiotic. By binding to specific penicillin-binding proteins (PBPs) located inside the bacterial cell wall, it inhibits the third and last stage of bacterial cell wall synthesis. Cell lysis is then mediated by bacterial cell wall autolytic enzymes such as autolysins; it is possible that Cefradine interferes with an autolysin inhibitor.
Cephalosporins and cephamycins inhibit bacterial cell wall synthesis in a manner similar to that of penicillin. /Cephalosporins/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 1085
The first generation cephalosporins ... have activity against gram positive bacteria and relatively modest activity against gram negative microorganisms. /Cephalosporins/
Gilman, A.G., T.W. Rall, A.S. Nies and P. Taylor (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 8th ed. New York, NY. Pergamon Press, 1990., p. 1085
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
20
PharmaCompass offers a list of Cephradine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Cephradine manufacturer or Cephradine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cephradine manufacturer or Cephradine supplier.
PharmaCompass also assists you with knowing the Cephradine API Price utilized in the formulation of products. Cephradine API Price is not always fixed or binding as the Cephradine Price is obtained through a variety of data sources. The Cephradine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Cephradine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Cephradine, including repackagers and relabelers. The FDA regulates Cephradine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Cephradine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Cephradine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Cephradine supplier is an individual or a company that provides Cephradine active pharmaceutical ingredient (API) or Cephradine finished formulations upon request. The Cephradine suppliers may include Cephradine API manufacturers, exporters, distributors and traders.
click here to find a list of Cephradine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Cephradine DMF (Drug Master File) is a document detailing the whole manufacturing process of Cephradine active pharmaceutical ingredient (API) in detail. Different forms of Cephradine DMFs exist exist since differing nations have different regulations, such as Cephradine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Cephradine DMF submitted to regulatory agencies in the US is known as a USDMF. Cephradine USDMF includes data on Cephradine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Cephradine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Cephradine suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Cephradine Drug Master File in Korea (Cephradine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Cephradine. The MFDS reviews the Cephradine KDMF as part of the drug registration process and uses the information provided in the Cephradine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Cephradine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Cephradine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Cephradine suppliers with KDMF on PharmaCompass.
A Cephradine CEP of the European Pharmacopoeia monograph is often referred to as a Cephradine Certificate of Suitability (COS). The purpose of a Cephradine CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Cephradine EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Cephradine to their clients by showing that a Cephradine CEP has been issued for it. The manufacturer submits a Cephradine CEP (COS) as part of the market authorization procedure, and it takes on the role of a Cephradine CEP holder for the record. Additionally, the data presented in the Cephradine CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Cephradine DMF.
A Cephradine CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Cephradine CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Cephradine suppliers with CEP (COS) on PharmaCompass.
A Cephradine written confirmation (Cephradine WC) is an official document issued by a regulatory agency to a Cephradine manufacturer, verifying that the manufacturing facility of a Cephradine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Cephradine APIs or Cephradine finished pharmaceutical products to another nation, regulatory agencies frequently require a Cephradine WC (written confirmation) as part of the regulatory process.
click here to find a list of Cephradine suppliers with Written Confirmation (WC) on PharmaCompass.
Cephradine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Cephradine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Cephradine GMP manufacturer or Cephradine GMP API supplier for your needs.
A Cephradine CoA (Certificate of Analysis) is a formal document that attests to Cephradine's compliance with Cephradine specifications and serves as a tool for batch-level quality control.
Cephradine CoA mostly includes findings from lab analyses of a specific batch. For each Cephradine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Cephradine may be tested according to a variety of international standards, such as European Pharmacopoeia (Cephradine EP), Cephradine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Cephradine USP).
