
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
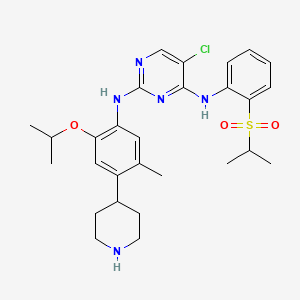

1. 5-chloro-n2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-n4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine
2. Ldk378
3. Zykadia
1. Ldk378
2. 1032900-25-6
3. Zykadia
4. Ldk-378
5. Nvp-ldk378-nx
6. Ldk 378
7. 5-chloro-n2-(2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl)-n4-(2-(isopropylsulfonyl)phenyl)pyrimidine-2,4-diamine
8. Ceritinib (ldk378)
9. Ceritinib(ldk378)
10. K418kg2get
11. Nvp-ldk-378-nx
12. Chembl2403108
13. Chebi:78432
14. 5-chloro-2-n-(5-methyl-4-piperidin-4-yl-2-propan-2-yloxyphenyl)-4-n-(2-propan-2-ylsulfonylphenyl)pyrimidine-2,4-diamine
15. 2,4-pyrimidinediamine, 5-chloro-n4-(2-((1-methylethyl)sulfonyl)phenyl)-n2-(5-methyl-2-(1-methylethoxy)-4-(4-piperidinyl)phenyl)-
16. 5-chloro-2-n-[5-methyl-4-(piperidin-4-yl)-2-(propan-2-yloxy)phenyl]-4-n-[2-(propane-2-sulfonyl)phenyl]pyrimidine-2,4-diamine
17. 5-chloro-n~2~-[5-methyl-4-(piperidin-4-yl)-2-(propan-2-yloxy)phenyl]-n~4~-[2-(propan-2-ylsulfonyl)phenyl]pyrimidine-2,4-diamine
18. 5-chloro-n2-(5-methyl-4-(piperidin-4-yl)-2-(propan-2-yloxy)phenyl)-n4-(2-(propane-2-sulfonyl)phenyl)pyrimidine-2,4-diamine
19. 5-chloro-n2-[2-isopropoxy-5-methyl-4-(4-piperidyl)phenyl]-n4-(2-isopropylsulfonylphenyl)pyrimidine-2,4-diamine
20. 5-chloro-n4-[2-[(1-methylethyl)sulfonyl]phenyl]-n2-[5-methyl-2-(1-methylethoxy)-4-(4-piperidinyl)phenyl]-2,4-pyrimidinediamine
21. 5-chloro-n4-[2-[(1methylethyl)sulfonyl]phenyl]-n2-[5-methyl-2-(1-methylethoxy)-4-(4-piperidinyl)phenyl]-2,4-pyrimidinediamine;5-chloro-n4-[2-[(1methylethyl)sulfonyl]phenyl]-n2-[5-methyl-2-(1-methylethoxy)-4-(4-piperidinyl)phenyl]-2,4-pyrimidinediamine
22. Ceritinib [usan:inn]
23. Unii-k418kg2get
24. Ceritinibum
25. Ceritinib[mi]
26. 5-chloro-n2-[5-methyl-4-(piperidin-4-yl)-2-(propan-2-yloxy)phenyl]-n4-[2-(propane-2-sulfonyl)phenyl]pyrimidine-2,4-diamine
27. Zykadia (tn)
28. Ldk378 Certinib
29. Ceritinib; Ldk378
30. Ldk378(ceritinib)
31. Ceritinib [inn]
32. Ceritinib [jan]
33. Eritinib (ldk378)
34. Ceritinib [mi]
35. Ceritinib [usan]
36. Ceritinib [vandf]
37. Ceritinib [who-dd]
38. Ceritinib (jan/usan/inn)
39. Gtpl7397
40. Schembl1014329
41. Ceritinib [orange Book]
42. Dtxsid10725373
43. Ex-a187
44. Hms3652h22
45. Hms3673i17
46. Hms3747a11
47. Amy10314
48. Bcp07611
49. Bdbm50436850
50. Mfcd26142648
51. Nsc776422
52. Nsc777193
53. Nsc800072
54. Zinc96272772
55. Akos025396438
56. Ccg-264762
57. Cs-1406
58. Db09063
59. Gs-6356
60. Nsc-776422
61. Nsc-777193
62. Nsc-800072
63. Sb16490
64. Compound 15b [pmid 23742252]
65. Ncgc00351603-10
66. Ncgc00351603-13
67. Ncgc00351603-15
68. Ac-27469
69. Hy-15656
70. Ft-0697208
71. S7083
72. Sw219725-1
73. D10551
74. A852144
75. J-690011
76. Q21011233
77. N-{2-methyl-5-[(methylamino)methyl]phenyl}-4-[(4-phenyl-2-quinazolinyl)amino]benzamide
78. 4mk
79. 5-chloro-n(2)-{5-methyl-4-(piperidin-4-yl)-2-[(propan-2-yl)oxy]phenyl}-n(4)-[2-(propane-2-sulfonyl)phenyl]pyrimidine-2,4-diamine
80. 5-chloro-n2-(2-isopropoxy-5-methyl-4-piperidin-4-yl-phenyl)-n4-[2-(propane-2-sulfonyl)-phenyl]-pyrimidine-2,4-diamine
81. 5-chloro-n2-(5-methyl-4-(piperidin-4-yl)-2-(propan-2-yloxy)phenyl)-n4-(2-(propane-2- Sulfonyl)phenyl)pyrimidine-2,4-diamine
82. 5-chloro-n4-(2-((1-methylethyl)sulfonyl)phenyl)-n2-(5-methyl-2-(1-methylethoxy)-4- (piperidin-4-yl)phenyl)pyrimidine-2,4-diamine
83. Ceritinib;5-chloro-n2-[2-isopropoxy-5-methyl-4-(4-piperidyl)phenyl]-n4-(2-isopropylsulfonylphenyl)pyrimidine-2,4-diamine;ceritinib
84. N-{2-[(5-chloro-2-{[2-isopropoxy-5-methyl-4-(piperidin-4-yl)phenyl]amino}pyrimidin-4-yl)amino]phenyl}propane-2-sulfonamide
| Molecular Weight | 558.1 g/mol |
|---|---|
| Molecular Formula | C28H36ClN5O3S |
| XLogP3 | 6.4 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Exact Mass | 557.2227389 g/mol |
| Monoisotopic Mass | 557.2227389 g/mol |
| Topological Polar Surface Area | 114 Ų |
| Heavy Atom Count | 38 |
| Formal Charge | 0 |
| Complexity | 835 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Ceritinib is a kinase inhibitor indicated for the treatment of patients with anaplastic lymphoma kinase (ALK)-positive metastatic non-small cell lung cancer (NSCLC) who have progressed on or are intolerant to crizotinib. This indication is approved under accelerated approval based on tumor response rate and duration of response. An improvement in survival or disease-related symptoms has not been established. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
FDA Label
Zykadia is indicated for the treatment of adult patients with anaplastic lymphoma kinase (ALK) positive advanced non small cell lung cancer (NSCLC) previously treated with crizotinib.
Antineoplastic Agents
Substances that inhibit or prevent the proliferation of NEOPLASMS. (See all compounds classified as Antineoplastic Agents.)
Protein Kinase Inhibitors
Agents that inhibit PROTEIN KINASES. (See all compounds classified as Protein Kinase Inhibitors.)
L01XE
L01XE28
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01E - Protein kinase inhibitors
L01ED - Anaplastic lymphoma kinase (alk) inhibitors
L01ED02 - Ceritinib
Absorption
After oral administration of ceritinib, peak concentrations were achieved after approximately 4 to 6 hours.
Route of Elimination
Following oral administration of a single 750 mg radiolabeled ceritinib dose, 92.3% of the administered dose was recovered in the feces (with 68% as unchanged parent compound) while 1.3% of the administered dose was recovered in the urine.
Volume of Distribution
The apparent volume of distribution (Vd/F) is 4230 L following a single 750 mg dose.
Clearance
The geometric mean apparent clearance (CL/F) of ceritinib was lower at steady-state (33.2 L/h) after 750 mg daily dosing than after a single 750 mg dose (88.5 L/h).
In vitro studies demonstrated that CYP3A was the major enzyme involved in the metabolic clearance of ceritinib. Following oral administration of a single 750 mg radiolabeled ceritinib dose, ceritinib as the parent compound was the main circulating component (82%) in human plasma.
The terminal half life is 41 hours.
Ceritinib inhibits Anaplastic lymphoma kinase (ALK) also known as ALK tyrosine kinase receptor or CD246 (cluster of differentiation 246), which is an enzyme that in humans is encoded by the ALK gene. About 4-5% of NSCLCs have a chromosomal rearrangement that generates a fusion gene between EML4 (echinoderm microtubule-associated protein-like 4) and ALK (anaplastic lymphoma kinase), which results in constitutive kinase activity that contributes to carcinogenesis and seems to drive the malignant phenotype. Ceritinib exerts its therapeutic effect by inhibiting autophosphorylation of ALK, ALK-mediated phosphorylation of the downstream signaling protein STAT3, and proliferation of ALK-dependent cancer cells. Ceritinib has been shown to inhibit in vitro proliferation of cell lines expressing EML4-ALK and NPM-ALK fusion proteins and demonstrated dose-dependent inhibition of EML4-ALK-positive NSCLC xenograft growth in mice and rats.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
23
PharmaCompass offers a list of Ceritinib API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Ceritinib manufacturer or Ceritinib supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ceritinib manufacturer or Ceritinib supplier.
PharmaCompass also assists you with knowing the Ceritinib API Price utilized in the formulation of products. Ceritinib API Price is not always fixed or binding as the Ceritinib Price is obtained through a variety of data sources. The Ceritinib Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Ceritinib manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ceritinib, including repackagers and relabelers. The FDA regulates Ceritinib manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ceritinib API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Ceritinib manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Ceritinib supplier is an individual or a company that provides Ceritinib active pharmaceutical ingredient (API) or Ceritinib finished formulations upon request. The Ceritinib suppliers may include Ceritinib API manufacturers, exporters, distributors and traders.
click here to find a list of Ceritinib suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Ceritinib DMF (Drug Master File) is a document detailing the whole manufacturing process of Ceritinib active pharmaceutical ingredient (API) in detail. Different forms of Ceritinib DMFs exist exist since differing nations have different regulations, such as Ceritinib USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ceritinib DMF submitted to regulatory agencies in the US is known as a USDMF. Ceritinib USDMF includes data on Ceritinib's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ceritinib USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ceritinib suppliers with USDMF on PharmaCompass.
A Ceritinib written confirmation (Ceritinib WC) is an official document issued by a regulatory agency to a Ceritinib manufacturer, verifying that the manufacturing facility of a Ceritinib active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Ceritinib APIs or Ceritinib finished pharmaceutical products to another nation, regulatory agencies frequently require a Ceritinib WC (written confirmation) as part of the regulatory process.
click here to find a list of Ceritinib suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Ceritinib as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Ceritinib API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Ceritinib as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Ceritinib and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Ceritinib NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Ceritinib suppliers with NDC on PharmaCompass.
Ceritinib Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ceritinib GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Ceritinib GMP manufacturer or Ceritinib GMP API supplier for your needs.
A Ceritinib CoA (Certificate of Analysis) is a formal document that attests to Ceritinib's compliance with Ceritinib specifications and serves as a tool for batch-level quality control.
Ceritinib CoA mostly includes findings from lab analyses of a specific batch. For each Ceritinib CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ceritinib may be tested according to a variety of international standards, such as European Pharmacopoeia (Ceritinib EP), Ceritinib JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ceritinib USP).
