
Synopsis
Synopsis
0
EU WC
0
KDMF
0
VMF
0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
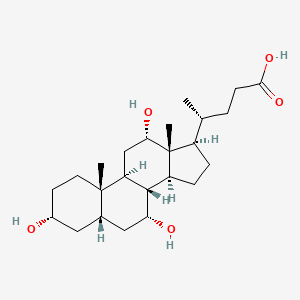

1. Cholbam
1. 81-25-4
2. Cholalic Acid
3. Cholate
4. Colalin
5. Cholalin
6. Cholbam
7. Cholsaeure
8. Nsc-6135
9. Kolbam
10. 3alpha,7alpha,12alpha-trihydroxy-5beta-cholanic Acid
11. Cholic Acid [usan]
12. Cholic Acid, 5beta-
13. 3,7,12-trihydroxycholanic Acid
14. 3alpha,7alpha,12alpha-trihydroxy-5beta-cholan-24-oic Acid
15. (r)-4-((3r,5s,7r,8r,9s,10s,12s,13r,14s,17r)-3,7,12-trihydroxy-10,13-dimethylhexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoic Acid
16. Sodium Cholate
17. Mfcd00003672
18. Hsdb 982
19. (4r)-4-[(3r,5s,7r,8r,9s,10s,12s,13r,14s,17r)-3,7,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoic Acid
20. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3a,5b,7a,12a)-
21. Terpenes And Terpenoids
22. Chembl205596
23. G1jo7801ae
24. Chebi:16359
25. Nsc6135
26. 3alpha,7alpha,12alpha-trihydroxy-5beta-cholanate
27. Chd
28. Cholic Acid (usan)
29. Orphacol
30. 3-alpha,7-alpha,12-alpha-trihydroxy-5-beta-cholan-24-oic Acid
31. 5beta-cholan-24-oic Acid, 3alpha,7alpha,12alpha-trihydroxy-
32. C24h40o5
33. 3,7,12-trihydroxycholan-24-oic Acid, (3alpha,5beta,7alpha,12alpha)-
34. 17-beta-(1-methyl-3-carboxypropyl)etiocholane-3alpha,7alpha,12alpha-triol
35. 3,7,12-trihydroxy-cholan-24-oic Acid (3-alpha,5-beta,7-alpha,12-alpha)
36. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3alpha,5beta,7alpha,12alpha)-
37. 129874-08-4
38. 4-[(3r,7r,8r,9s,10s,12s,13r,14s,17r)-3,7,12-trihydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoic Acid
39. (3alpha,5beta,7alpha,12alpha)-3,7,12-trihydroxycholan-24-oic Acid
40. (3alpha,5beta,7alpha,8x,12alpha)-3,7,12-trihydroxycholan-24-oic Acid
41. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3-alpha,5-beta,7-alpha,12-alpha)-
42. Smr000112165
43. Colalin (van)
44. Cholsaeure [german]
45. 5beta-cholanic Acid-3alpha,7alpha,12alpha-triol
46. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3.alpha.,5.beta.,7.alpha.,12.alpha.)-
47. Unii-g1jo7801ae
48. Cholalate
49. Orphacolreg
50. Cholic-acid
51. Ccris 1626
52. (3alpha,5beta,7alpha,8xi,12alpha)-3,7,12-trihydroxycholan-24-oic Acid
53. 5b-cholate
54. 5b-cholic Acid
55. Cholsa Currencyure
56. Cholbam (tn)
57. Einecs 201-337-8
58. Cholic Acid (8ci)
59. 5.beta.-cholic Acid
60. Brn 2822009
61. 361-09-1
62. Spectrum5_002005
63. Cholic Acid [mi]
64. 3alpha,7alpha,12alpha-trihydroxycholanic Acid
65. Bmse000650
66. Cholic Acid [fcc]
67. Cholic Acid [jan]
68. Ec 201-337-8
69. Cholic Acid [hsdb]
70. 3-alpha,7-alpha,12-alpha-trihydroxycholansaeure
71. Bidd:pxr0196
72. Schembl27461
73. Gtpl609
74. 3.alpha.,7.alpha.,12.alpha.-trihydroxy-5.beta.-cholanic Acid
75. 4-10-00-02072 (beilstein Handbook Reference)
76. Mls001066422
77. Mls002207051
78. Cholic Acid [usp-rs]
79. Cholic Acid [who-dd]
80. 3a,7a,12a-trihydroxycholanate
81. 3alpha,7alpha,12alpha-trihydroxy-5beta-cholanoic Acid
82. Cholic Acid [ema Epar]
83. Dtxsid6040660
84. Bdbm21680
85. 3-alpha,7-alpha,12-alpha-trihydroxycholansaeure [german]
86. Cholic Acid [ep Impurity]
87. Cholic Acid [orange Book]
88. Hms2268l18
89. 3a,7a,12a-trihydroxy-b-cholanate
90. 3a,7a,12a-trihydroxycholanic Acid
91. Hy-n0324
92. Zinc6858022
93. 3a,7a,12a-trihydroxy-5b-cholanate
94. 5b-cholanic Acid-3a,7a,12a-triol
95. Bbl029799
96. Lmst04010001
97. S3742
98. Stk801993
99. 3a,7a,12a-trihydroxy-5b-cholanoate
100. 3a,7a,12a-trihydroxy-beta-cholanate
101. Akos005622502
102. 3a,7a,12a-trihydroxy-b-cholanic Acid
103. Ccg-268746
104. Cs-6608
105. Db02659
106. 3a,7a,12a-trihydroxy-5a-cholanic Acid
107. 3a,7a,12a-trihydroxy-5b-cholanic Acid
108. Ncgc00142384-03
109. 3a,7a,12a-trihydroxy-5b-cholanoic Acid
110. 3a,7a,12a-trihydroxy-beta-cholanic Acid
111. 4-((1s,2s,7s,11s,16s,5r,9r,10r,14r,15r)-5,9,16-trihydroxy-2,15-dimethyltetracy Clo[8.7.0.0<2,7>.0<11,15>]heptadec-14-yl)pentanoic Acid
112. As-69849
113. Bp-30084
114. 3a,7a,12a-trihydroxy-5b-cholan-24-oate
115. Cholic Acid, Vetec(tm) Reagent Grade, 98%
116. N1680
117. Cholic Acid, From Ox Or Sheep Bile, >=98%
118. 3a,7a,12a-trihydroxy-5b-cholan-24-oic Acid
119. C-5900
120. C00695
121. D10699
122. A830252
123. Q287415
124. Sr-01000765698
125. 3alpha,7alpha,12alpha-trihydroxy-beta-cholanic Acid
126. Sr-01000765698-4
127. 3.alpha.,12.alpha.-trihydroxy-5.beta.-cholanic Acid
128. Ursodeoxycholic Acid Impurity B [ep Impurity]
129. 3-.alpha.,7-.alpha.,12-.alpha.-trihydroxycholansaeure
130. 3alpha, 7alpha, 12alpha-trihydroxy-5beta-cholanic Acid
131. 3alpha, 7alpha, 12alpha-trihydroxy-5beta-cholanoic Acid
132. 3-&alpha,7-&alpha,12-&alpha-trihydroxy-5-&beta-cholanate
133. Cholic Acid, British Pharmacopoeia (bp) Reference Standard
134. Cholic Acid, European Pharmacopoeia (ep) Reference Standard
135. 17b-[1-methyl-3-carboxypropyl]etiocholane-3a,7a,12a-triol
136. 5.beta.-cholan-24-oic Acid,7.alpha.,12.alpha.-trihydroxy-
137. 5beta-cholanic Acid, 3alpha,7alpha,12alpha-trihydroxy- (7ci)
138. 5beta-cholanic Acid-3alpha,7alpha,12alpha-triol 5beta-cholic Acid
139. Cholic Acid, 500 Mug/ml In Methanol, Certified Reference Material
140. Cholic Acid, United States Pharmacopeia (usp) Reference Standard
141. 17beta-[1-methyl-3-carboxypropyl]etiocholane-3alpha,7alpha,12alpha-triol
142. 3-.alpha.,7-.alpha.,12-.alpha.-trihydroxy-5-.beta.-cholan-24-oic Acid
143. 3alpha,7alpha,12alpha-trihydroxy-5beta-cholan-24-oic Acid (cholic Acid)
144. 5.beta.-cholan-24-oic Acid, 3.alpha.,7.alpha.,12.alpha.-trihydroxy-
145. (3.alpha.,5.beta.,7.alpha.,12.alpha.)-3,7,12-trihydroxycholan-24-oic Acid Sodium Salt
146. (3alpha,5beta,7alpha,8alpha,12alpha,14beta,17alpha)-3,7,12-trihydroxycholan-24-oic Acid
147. 17.beta.-(1-methyl-3-carboxypropyl)etiocholane-3.alpha.,7.alpha.,12.alpha.-triol
148. 3,7,12-trihydroxy-cholan-24-oic Acid, (3.alpha.,5.beta.,7.alpha.,12.alpha.)-
149. 3.alpha.,7.alpha.,12.alpha.-trihydroxy-5.beta.-cholan-24-oic Acid
150. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3alpha,5beta,7alpha,12alpha)- (9ci)
151. Cholan-24-oic Acid,7,12-trihydroxy-, (3.alpha.,5.beta.,7.alpha.,12.alpha.)-
152. (4r)-4-((3r,7r,8r,9s,10s,12s,13r,14s,17r)-3,7,12-trihydroxy-10,13-dimethylhexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoic Acid
153. (4r)-4-[(1r,3as,3br,4r,5as,7r,9as,9bs,11s,11ar)-4,7,11-trihydroxy-9a,11a-dimethyl-hexadecahydro-1h-cyclopenta[a]phenanthren-1-yl]pentanoic Acid
154. (4r)-4-[(1s,2s,5r,7s,9r,10r,11s,14r,15r,16s)-5,9,16-trihydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadecan-14-yl]pentanoic Acid
155. (r)-4-((3r,5s,7r,8r,10s,12s,13r,14s,17r)-3,7,12-trihydroxy-10,13-dimethyl-hexadecahydro-1h-cyclopenta[a]phenanthren-17-yl)pentanoic Acid
156. 10321-98-9
157. Cholan-24-oic Acid, 3,7,12-trihydroxy-, (3-.alpha.,5-.beta.,7-.alpha.,12-.alpha.)-
| Molecular Weight | 408.6 g/mol |
|---|---|
| Molecular Formula | C24H40O5 |
| XLogP3 | 3.6 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Exact Mass | 408.28757437 g/mol |
| Monoisotopic Mass | 408.28757437 g/mol |
| Topological Polar Surface Area | 98 Ų |
| Heavy Atom Count | 29 |
| Formal Charge | 0 |
| Complexity | 637 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 11 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Cholic acid is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of July 18, 2015: https://clinicaltrials.gov/search/intervention=cholic+acid
Cholbam is indicated for the treatment of bile acid synthesis disorders due to single enzyme defects (SEDs). /Included in US product label/
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
Cholbam is indicated for adjunctive treatment of peroxisomal disorders (PDs) including Zellweger spectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption. /Included in US product label/
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 1, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
The safety and effectiveness of Cholbam on extrahepatic manifestations of bile acid synthesis disorders due to single enzyme defects (SEDs) or peroxisomal disorders (PDs) including Zellweger spectrum disorders have not been established.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 1, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
For more Therapeutic Uses (Complete) data for CHOLIC ACID (7 total), please visit the HSDB record page.
The safety and effectiveness of Cholbam has been established in pediatric patients 3 weeks of age and older for the treatment of bile acid synthesis disorders due to single enzyme defects (SEDs), and for adjunctive treatment of patients with peroxisomal disorders (PDs) including Zellweger spectrum disorders who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
Monitor liver function and discontinue Cholbam in patients who develop worsening of liver function while on treatment. Concurrent elevations of serum gamma glutamyltransferase (GGT), alanine aminotransferase (ALT) may indicate Cholbam overdose. Discontinue treatment with Cholbam at any time if there are clinical or laboratory indicators of worsening liver function or cholestasis.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
The developmental and health benefits of breastfeeding should be considered along with the mother's clinical need for Cholbam and any potential adverse effects on the breastfed infant from Cholbam or from the underlying maternal condition.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
No studies in pregnant women or animal reproduction studies have been conducted with Cholbam. Limited published case reports discuss pregnancies in women taking cholic acid for 3beta-HSD deficiency resulting in healthy infants. These reports may not adequately inform the presence or absence of drug-associated risk with the use of Cholbam during pregnancy. The background risk of major birth defects and miscarriage for the indicated population is unknown. However, the background risk in the U.S. general population of major birth defects is 2-4% and of miscarriage is 15-20% of clinically recognized pregnancies.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
For more Drug Warnings (Complete) data for CHOLIC ACID (6 total), please visit the HSDB record page.
Oral cholic acid is indicated for: treatment of bile acid synthesis disorders due to single enzyme defects; and as adjunctive treatment of peroxisomal disorders including Zellweger spectrum disorders in patients who exhibit manifestations of liver disease, steatorrhea or complications from decreased fat soluble vitamin absorption.
Orphacol is indicated for the treatment of inborn errors in primary bile-acid synthesis due to 3-hydroxy-5-C27-steroid oxidoreductase deficiency or 4-3-oxosteroid-5-reductase deficiency in infants, children and adolescents aged one month to 18 years and adults.
Cholic Acid FGK is indicated for the treatment of inborn errors of primary bile acid synthesis, in infants from one month of age for continuous lifelong treatment through adulthood, encompassing the following single enzyme defects:
- sterol 27-hydroxylase (presenting as cerebrotendinous xanthomatosis, CTX) deficiency;
- 2- (or alpha-) methylacyl-CoA racemase (AMACR) deficiency;
- cholesterol 7 alpha-hydroxylase (CYP7A1) deficiency.
Treatment of other inborn errors of bile acid synthesis, Treatment of oxysterol 7a-hydrolylase deficiency and defective amida
A05AA03
A05AA03
A - Alimentary tract and metabolism
A05 - Bile and liver therapy
A05A - Bile therapy
A05AA - Bile acids and derivatives
A05AA03 - Cholic acid
Following ingestion, absorption of cholic acid will first be by the small intestine, and is then transported to the liver by the blood for further processing.
European Medicines Agency (EMA), Committee for Medicinal Products for Human Use (CHMP), European Public Assessment Report (EPAR): Orphacol (Cholic Acid) p.19 (2011). Available from, as of June 2, 2015: https://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/001250/WC500131542.pdf
Orally administered cholic acid is subject to the same metabolic pathway as endogenous cholic acid. Cholic acid is absorbed by passive diffusion along the length of the gastrointestinal tract. Once absorbed, cholic acid enters into the body's bile acid pool and undergoes enterohepatic circulation mainly in conjugated forms. In the liver, cholic acid is conjugated with glycine or taurine by bile acid-CoA synthetase and bile acid-CoA: amino acid N-acetyltransferase. Conjugated cholic acid is actively secreted into bile mainly by the Bile Salt Efflux Pump (BSEP), and then released into the small intestine, along with other components of bile. Conjugated cholic acid is mostly re-absorbed in the ileum mainly by the apical-sodium-dependent-bile acid transporter, passed back to the liver by transporters including sodium-taurocholate cotransporting polypeptide and organic anion transport protein and enters another cycle of enterohepatic circulation. Any conjugated cholic acid not absorbed in the ileum passes into the colon where deconjugation and 7-dehydroxylation are mediated by bacteria to form cholic acid and deoxycholic acid which may be re-absorbed in the colon or excreted in the feces. The loss of cholic acid is compensated by de-novo synthesis of cholic acids from cholesterol to maintain the bile acid pool in healthy subjects.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
Excretion studies in rat showed that cholic acid (CA) is almost exclusively excreted in the feces in the form of metabolite. Only minor amounts of CA were found in the unconjugated form in rat feces. Urinary excretion of bile acids is minimal and in mice fed a 1% CA diet, the excretion of bile acids was 2000-fold higher in feces than in urine
European Medicines Agency (EMA), Committee for Medicinal Products for Human Use (CHMP), European Public Assessment Report (EPAR): Orphacol (Cholic Acid) p.20 (2011). Available from, as of June 2, 2015: https://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/001250/WC500131542.pdf
Cholic acid was shown to be mainly absorbed in the distal (ileal) rather than proximal segments of the small intestine in the guinea pig.
European Medicines Agency (EMA), Committee for Medicinal Products for Human Use (CHMP), European Public Assessment Report (EPAR): Orphacol (Cholic Acid) p.19 (2011). Available from, as of June 2, 2015: https://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/001250/WC500131542.pdf
For more Absorption, Distribution and Excretion (Complete) data for CHOLIC ACID (9 total), please visit the HSDB record page.
The mechanism and sequence of side chain hydroxylation of cholesterol in bile acid synthesis was studied in the isolated perfused rabbit liver. A comparison was made between the importance of 26- and 25-hydroxylation in cholic acid biosynthesis in the rabbit. The formation of [G-(3)H]cholic acid was observed when the liver was perfused with 5beta-[G-(3)H]cholestane-3alpha, 7alpha-diol, 5beta-[G-(3)H]cholestane-3alpha, 7alpha-12alpha-triol, and 5beta-[G-(3)H]cholestane-3alpha, 7alpha, 26-triol. No [G-(3)H]chenodeoxycholic acid was detected in the bile. These findings indicate that potential precursors of chenodeoxycholic acid were hydroxylated at position 12alpha either subsequent to or before hydroxylation of the cholesterol side chain. In addition, no other intermediates (tetrahydroxy or pentahydroxy bile alcohols) were found in the bile when these compounds were perfused in the liver. Bile acid precursors were detected in bile when the rabbit liver was perfused with 5beta-[24-(14)C]cholestane-3alpha, 7alpha, 25-triol. The 5beta-[24-(14)C]cholestane-3alpha, 7alpha, 25-triol was hydroxylated in the liver at the 12alpha position to yield the corresponding 5beta-cholestane-3alpha, 7alpha, 12alpha, 25-tetrol. The tetrol was further metabolized to a series of pentols (5beta-cholestane-3alpha, 7alpha, 12alpha, 22, 25-pentol; 5beta-cholestane-3alpha, 7alpha, 12alpha, 23, 25-pentol; 5beta-cholestane-3alpha, 7alpha, 12alpha, 24, 25-pentol; and 5beta-cholestane-3alpha, 7alpha, 12alpha, 25, 26-pentol). The major bile acid obtained from the perfusion of the 5beta-cholestane-3alpha, 7alpha, 25-triol was cholic acid. The experiments indicated that in the rabbit liver 12alpha-hydroxylation can occur after hydroxylation of the cholesterol side chain at either C-25 (5 beta-cholestane-3alpha, 7alpha, 25-triol) or C-26 (5beta-cholestane-3alpha, 7alpha-26-triol). Apparently, the rabbit can form cholic acid via the classical 26-hydroxylation pathway as well as via 25-hydroxylated intermediates.
PMID:1262339 Cohen B et al; J Biol Chem 251 (9): 2709-15 (1976)
In classic cholic acid biosynthesis, a series of ring modifications of cholesterol precede side chain cleavage and yield 5beta-cholestane-3alpha, 7alpha, 12alpha-triol. Side chain reactions of the triol then proceed either by the mitochondrial 27-hydroxylation pathway or by the microsomal 25-hydroxylation pathway. We have developed specific and precise assay methods to measure the activities of key enzymes in both pathways, 5beta-cholestane-3alpha, 7alpha, 12alpha-triol 25- and 27-hydroxylases and 5beta-cholestane-3alpha, 7alpha, 12alpha, 25-tetrol 23R-, 24R-, 24S- and 27-hydroxylases. The extracts from either the mitochondrial or microsomal incubation mixtures were purified by means of a disposable silica cartridge column, derivatized into trimethylsilyl ethers, and quantified by gas chromatography;-mass spectrometry with selected-ion monitoring in a high resolution mode. Compared with the addition of substrates in acetone, those in 2-hydroxypropyl-beta-cyclodextrin increased mitochondrial triol 27-hydroxylase activity 132% but decreased activities of the enzymes in microsomal 25-hydroxylation pathway (triol 25-hydroxylase and 5beta-cholestane-3alpha, 7alpha, 12alpha, 25-tetrol 23R-, 24R-, 24S- and 27-hydroxylases) 13;-60% in human liver. The enzyme activities in both pathways were generally 2- to 4-times higher in mouse and rabbit livers compared with human liver. In all species, microsomal triol 25-hydroxylase activities were 4- to 11-times larger than mitochondrial triol 27-hydroxylase activities but the activities of tetrol 24S-hydroxylase were similar to triol 27-hydroxylase activities in our assay conditions. The regulation of both pathways in rabbit liver was studied after bile acid synthesis was perturbed. Cholesterol feeding up-regulated enzyme activities involved in both 25- (64;-142%) and 27- (77%) hydroxylation pathways, while bile drainage up-regulated only the enzymes in the 25-hydroxylation pathway (178;-371%). Using these new assays, we demonstrated that the 25- and 27-hydroxylation pathways for cholic acid biosynthesis are more active in mouse and rabbit than human livers and are separately regulated in rabbit liver.
PMID:10706592 Honda A et al; J Lipid Res 41 (3): 442-51 (2000)
Deoxycholic acid is the main metabolite of cholic acid. Patients with 3alpha-HSD deficiency and delta4-3-oxoR deficiency and subjects with a normal bile acid metabolism have shown that upon treatment with cholic acid, serum and bile predominantly contain cholic acid and deoxycholic acid, while chenodeoxycholic acid and its metabolites appear to be reduced. Under cholic acid treatment, patients are therefore exposed to higher than normal deoxycholic acid concentrations, although the exact quantifications of these concentrations have not been described. In single- and repeat-dose studies, deoxycholic acid showed lethal effects, gastrointestinal and hepatic toxicities at approximately half the doses needed for cholic acid to produce the same effects. It is therefore considered that deoxycholic acid is more toxic than cholic acid and may in fact be the causative agent of some of cholic acid's toxicity. Mutagenicity data from bacterial test for deoxycholic acid is ambiguous but deoxycholic acid was genotoxic in an in vitro micronucleus assay. Additionally, ... the genotoxic potential of BA (focusing on chenodeoxycholic acid and deoxycholic acid) on human colonocytes and colon tumor cells HT 29 by a comet assay /was investigated/. In both cell types a clear dose-dependent genotoxic effect induced by the two bile acids was observed, with deoxycholic acid being more genotoxic. Viability of cells appeared to be greater than 75%. Use of a nuclease III modified comet assay suggested that the DNA damage could be mediated by reactive oxygen species production but was somewhat protected by inclusion of anti-oxidants. Short term carcinogenicity studies suggest that deoxycholic acid like cholic acid has carcinogenicity promoting properties. In rat liver, deoxycholic acid (75-150 mg/kg) exerted promoting activity as evidenced by significantly increased values of alpha-glutamyl transpeptidase-positive (alpha-GT+) liver foci compared with the corresponding controls given the carcinogen, diethylnitrosamine (DEN) alone. Deoxycholic acid (20 mg/kg) enhanced the development and growth of azoxymethane (AOM)-induced aberrant crypt foci in rat colons. In a parallel study, deoxycholic acid in the absence of AOM did not significantly induce aberrant crypt foci. However, /a study/ concluded that deoxycholic acid may act not only as promoters but also initiators of the multistage process of carcinogenesis.
European Medicines Agency (EMA), Committee for Medicinal Products for Human Use (CHMP), European Public Assessment Report (EPAR): Orphacol (Cholic Acid) p.24-5 (2011). Available from, as of June 2, 2015: https://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Public_assessment_report/human/001250/WC500131542.pdf
Cholic acid has known human metabolites that include Cholic acid glucuronide.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
Bile acids (BAs) are known to regulate BA synthesis and transport by the farnesoid X receptor in the liver (FXR-SHP) and intestine (FXR-Fgf15). However, the relative importance of individual BAs in regulating these processes is not known. Therefore, mice were fed various doses of five individual BAs, including cholic acid (CA), chenodeoxycholic acid (CDCA), deoxoycholic acid (DCA), lithocholic acid (LCA), and ursodeoxycholic acid (UDCA) in their diets at various concentrations for one week to increase the concentration of one BA in the enterohepatic circulation. The mRNA of BA synthesis and transporting genes in liver and ileum were quantified. In the liver, the mRNA of SHP, which is the prototypical target gene of FXR, increased in mice fed all concentrations of BAs. In the ileum, the mRNA of the intestinal FXR target gene Fgf15 was increased at lower doses and to a higher extent by CA and DCA than by CDCA and LCA. Cyp7a1, the rate-limiting enzyme in BA synthesis, was decreased more by CA and DCA than CDCA and LCA. Cyp8b1, the enzyme that 12-hydroxylates BAs and is thus responsible for the synthesis of CA, was decreased much more by CA and DCA than CDCA and LCA. Surprisingly, neither a decrease in the conjugated BA uptake transporter (Ntcp) nor increase in BA efflux transporter (Bsep) was observed by FXR activation, but an increase in the cholesterol efflux transporter (Abcg5/Abcg8) was observed with FXR activation. Thus in conclusion, CA and DCA are more potent FXR activators than CDCA and LCA when fed to mice, and thus they are more effective in decreasing the expression of the rate limiting gene in BA synthesis Cyp7a1 and the 12-hydroxylation of BAs Cyp8b1, and are also more effective in increasing the expression of Abcg5/Abcg8, which is responsible for biliary cholesterol excretion. However, feeding BAs do not alter the mRNA or protein levels of Ntcp or Bsep, suggesting that the uptake or efflux of BAs is not regulated by FXR at physiological and pharmacological concentrations of BAs.
PMID:25582706 Song P et al; Toxicol Appl Pharmacol 283 (1): 57-64 (2015)
Cholic acid is a primary bile acid synthesized from cholesterol in the liver. In bile acid synthesis disorders due to single enzyme defects (SEDs) in the biosynthetic pathway, and in peroxisomal disorders (PDs) including Zellweger spectrum disorders, deficiency of primary bile acids leads to unregulated accumulation of intermediate bile acids and cholestasis. Bile acids facilitate fat digestion and absorption by forming mixed micelles, and facilitate absorption of fat-soluble vitamins in the intestine. Endogenous bile acids including cholic acid enhance bile flow and provide the physiologic feedback inhibition of bile acid synthesis. The mechanism of action of cholic acid has not been fully established; however, it is known that cholic acid and its conjugates are endogenous ligands of the nuclear receptor, farnesoid X receptor (FXR). FXR regulates enzymes and transporters that are involved in bile acid synthesis and in the enterohepatic circulation to maintain bile acid homeostasis under normal physiologic conditions.
NIH; DailyMed. Current Medication Information for Cholbam (Cholic Acid) Capsule (Updated: March 2015). Available from, as of May 28, 2015: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=e5b67402-8550-4604-97a0-c7b149fbf753
 ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.
ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.

Certificate Number : CEP 2000-202 - Rev 08
Issue Date : 2024-04-03
Type : TSE
Substance Number :
Status : Valid

Registration Number : 304MF10051
Registrant's Address : Via Sicilia, 8/10 42122 Reggio Emilia, ITALY
Initial Date of Registration : 2022-03-10
Latest Date of Registration :

Certificate Number : CEP 2012-026 - Rev 06
Issue Date : 2023-11-27
Type : TSE
Substance Number :
Status : Valid

NDC Package Code : 12598-9622
Start Marketing Date : 2024-08-27
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Sanofi is a pioneer in Diabetes Solutions, Human Vaccines, Innovative Drugs, Consumer Healthcare, and the new Genzyme.
Certificate Number : R0-CEP 2001-021 - Rev 01
Issue Date : 2005-02-18
Type : TSE
Substance Number :
Status : Expired
 ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.
ICE Pharma offers a unique range of bile acid products and best-in-class services for the pharmaceutical industry.

Certificate Number : CEP 2000-334 - Rev 07
Issue Date : 2024-04-03
Type : TSE
Substance Number :
Status : Valid

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31884
Submission : 2017-07-14
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31885
Submission : 2017-07-14
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27181
Submission : 2013-06-07
Status : Active
Type : II

Registration Number : 306MF10023
Registrant's Address : 68 Weld Street, RD2 Palmerston North 4472, New Zealand
Initial Date of Registration : 2024-02-07
Latest Date of Registration :

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2843
Submission : 1977-02-07
Status : Inactive
Type : II

Certificate Number : R1-CEP 2004-036 - Rev 01
Issue Date : 2011-10-17
Type : TSE
Substance Number :
Status : Withdrawn by Holder


Certificate Number : R1-CEP 2009-252 - Rev 00
Issue Date : 2015-07-08
Type : TSE
Substance Number :
Status : Valid

NDC Package Code : 46438-0669
Start Marketing Date : 2024-11-28
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 27181
Submission : 2013-06-07
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31885
Submission : 2017-07-14
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 31884
Submission : 2017-07-14
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2843
Submission : 1977-02-07
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
28
PharmaCompass offers a list of Cholic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Cholic Acid manufacturer or Cholic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Cholic Acid manufacturer or Cholic Acid supplier.
PharmaCompass also assists you with knowing the Cholic Acid API Price utilized in the formulation of products. Cholic Acid API Price is not always fixed or binding as the Cholic Acid Price is obtained through a variety of data sources. The Cholic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)-, including repackagers and relabelers. The FDA regulates CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- supplier is an individual or a company that provides CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- active pharmaceutical ingredient (API) or CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- finished formulations upon request. The CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers may include CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- API manufacturers, exporters, distributors and traders.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- DMF (Drug Master File) is a document detailing the whole manufacturing process of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- active pharmaceutical ingredient (API) in detail. Different forms of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- DMFs exist exist since differing nations have different regulations, such as CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- DMF submitted to regulatory agencies in the US is known as a USDMF. CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- USDMF includes data on CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)-'s chemical properties, information on the facilities and procedures used, and details about packaging and storage. The CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- Drug Master File in Japan (CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- JDMF) empowers CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- JDMF during the approval evaluation for pharmaceutical products. At the time of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers with JDMF on PharmaCompass.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP of the European Pharmacopoeia monograph is often referred to as a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- Certificate of Suitability (COS). The purpose of a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- to their clients by showing that a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP has been issued for it. The manufacturer submits a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP (COS) as part of the market authorization procedure, and it takes on the role of a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP holder for the record. Additionally, the data presented in the CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- DMF.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- suppliers with NDC on PharmaCompass.
CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- GMP manufacturer or CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- GMP API supplier for your needs.
A CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CoA (Certificate of Analysis) is a formal document that attests to CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)-'s compliance with CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- specifications and serves as a tool for batch-level quality control.
CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CoA mostly includes findings from lab analyses of a specific batch. For each CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- may be tested according to a variety of international standards, such as European Pharmacopoeia (CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- EP), CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- JP (Japanese Pharmacopeia) and the US Pharmacopoeia (CHOLAN-24-OIC ACID, 3,7,12-TRIHYDROXY-, (3-.ALPHA.,5-.BETA.,7-.ALPHA.,12-.ALPHA.)- USP).
