

Synopsis
Synopsis
0
CEP/COS
0
VMF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
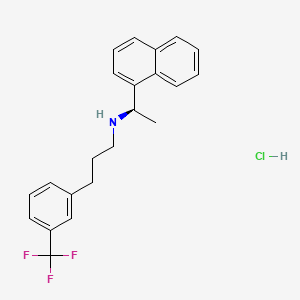

1. Alpha-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-1-naphthalenemethanamine, (alphar)-hydrochloride
2. Amg 073
3. Amg073
4. Cinacalcet
5. Krn 1493
6. Sensipar
1. 364782-34-3
2. Cinacalcet Hcl
3. Sensipar
4. Mimpara
5. Amg073 Hcl
6. Regpara
7. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hydrochloride
8. Cinacalcet, Hcl
9. Cinacalcet Hydrochloride [usan]
10. Cinacalcet (hydrochloride)
11. Amg-073 Hcl
12. Amg073 Hydrochloride
13. Chebi:48391
14. 1k860wsg25
15. Parareg
16. N-[(1r)-1-naphthalen-1-ylethyl]-3-[3-(trifluoromethyl)phenyl]propan-1-amine;hydrochloride
17. Amg-073 Hcl (cinacalcet Hydrochloride)
18. 364782-34-3 (hcl)
19. [(1r)-1-(naphthalen-1-yl)ethyl]({3-[3-(trifluoromethyl)phenyl]propyl})amine Hydrochloride
20. Dsstox_cid_26792
21. Dsstox_rid_81909
22. N-((1r)-1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hydrochloride
23. N-[(1r)-1-(1-naphthyl)ethyl]-3-[3-(trifluoromethyl)phenyl]propan-1-amine Hydrochloride
24. Dsstox_gsid_46792
25. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoro-methyl)phenyl)propan-1-amine Hydrochloride
26. Smr002530058
27. Cas-364782-34-3
28. Krn 1493
29. Ncgc00181002-01
30. Amg 073 Hcl
31. Unii-1k860wsg25
32. Sensipar (tn)
33. Mimpara (tn)
34. Cnc-hcl
35. Cinacelcet Hydrochloride
36. Sensipar(tm)
37. Amg-073 Hydrochloride
38. Amg-073.hcl
39. Mls004774045
40. Mls006010213
41. Chembl1200776
42. Dtxsid3046792
43. Bcpp000409
44. Bcp02533
45. Nps-1493
46. Tox21_112654
47. Cinacalcet Hydrochloride (jan/usan)
48. Cinacalcet Hydrochloride [mi]
49. Hy-70037a
50. Mfcd08067750
51. S1260
52. Cinacalcet Hydrochloride [jan]
53. Akos005146514
54. Akos015969126
55. Tox21_112654_1
56. Ac-1799
57. Am90312
58. Bcp9000286
59. Ccg-268579
60. Cs-0288
61. Gs-4171
62. Cinacalcet Hydrochloride [mart.]
63. Cinacalcet Hydrochloride [usp-rs]
64. Cinacalcet Hydrochloride [who-dd]
65. Ncgc00181002-04
66. 1-naphthalenemethanamine, Alpha-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-, (alphar)-, Hydrochloride
67. Sw219246-1
68. Cinacalcet Hydrochloride [orange Book]
69. D03505
70. 782c343
71. J-520045
72. Q27121179
73. Z1741977003
74. (r)-n-(1-(naphthalen-1-yl)ethyl)-3-(3-(trifluoromethyl)phenyl)propan-1-amine Hcl
75. (r)-n-(3-(3-(trifluoromethyl)phenyl)propyl)-1- -(1-napthyl)ethylamine Hydrochloride
76. (alphar)-alpha-methyl-n-[3-[3-(trifluoromethyl)phenyl)propyl]-1-napthalenemethanamine Hydrochloride
77. 1-naphthalenemethanamine, .alpha.-methyl-n-(3-(3-(trifluoromethyl)phenyl)propyl)-, (.alpha.r)-, Hydrochloride
78. 1-naphthalenemethanamine, Alpha-methyl-n-[3-[3-(trifluoromethyl)phenyl]propyl]-, Hydrochloride (1:1), (alphar)-
79. 1-naphthalenemethanamine,a-methyl-n-[3-[3-(trifluoromethyl)phenyl]propyl]-, Hydrochloride, (ar)-
| Molecular Weight | 393.9 g/mol |
|---|---|
| Molecular Formula | C22H23ClF3N |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 6 |
| Exact Mass | 393.1471119 g/mol |
| Monoisotopic Mass | 393.1471119 g/mol |
| Topological Polar Surface Area | 12 Ų |
| Heavy Atom Count | 27 |
| Formal Charge | 0 |
| Complexity | 422 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Cinacalcet hydrochloride |
| PubMed Health | Cinacalcet (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 60mg; 30mg; 90mg |
| Market Status | Tentative Approval |
| Company | Teva Pharms; Barr |
| 2 of 4 | |
|---|---|
| Drug Name | Sensipar |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 90mg base; eq 30mg base; eq 60mg base |
| Market Status | Prescription |
| Company | Amgen |
| 3 of 4 | |
|---|---|
| Drug Name | Cinacalcet hydrochloride |
| PubMed Health | Cinacalcet (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | oral |
| Strength | 60mg; 30mg; 90mg |
| Market Status | Tentative Approval |
| Company | Teva Pharms; Barr |
| 4 of 4 | |
|---|---|
| Drug Name | Sensipar |
| Drug Label | Sensipar (cinacalcet) is a calcimimetic agent that increases the sensitivity of the calcium-sensing receptor to activation by extracellular calcium. Its empirical formula is C22H22F3NHCl with a molecular weight of 393.9 g/mol (hydrochloride salt... |
| Active Ingredient | Cinacalcet hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 90mg base; eq 30mg base; eq 60mg base |
| Market Status | Prescription |
| Company | Amgen |
* Secondary hyperparathyroidism :
Adults
Treatment of secondary hyperparathyroidism (HPT) in adult patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Paediatric population
Treatment of secondary hyperparathyroidism (HPT) in children aged 3 years and older with end-stage renal disease (ESRD) on maintenance dialysis therapy in whom secondary HPT is not adequately controlled with standard of care therapy (see section 4. 4).
Cinacalcet Accordpharma may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate (see section 5. 1).
* Parathyroid carcinoma and primary hyperparathyroidism in adults:
Reduction of hypercalcaemia in adult patients with:
- parathyroid carcinoma.
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of secondary hyperparathyroidism (HPT) in patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Cinacalcet Mylan may be used as part of a therapeutic regimen including phosphate binders and/or vitamin D sterols, as appropriate.
Reduction of hypercalcaemia in patients with:
- parathyroid carcinoma
- primary HPT for whom parathyroidectomy
would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
* Secondary hyperparathyroidism :
Adults
Treatment of secondary hyperparathyroidism (HPT) in adult patients with end stage renal disease (ESRD) on maintenance dialysis therapy.
Paediatric population
Treatment of secondary hyperparathyroidism (HPT) in children aged 3 years and older with end stage renal disease (ESRD) on maintenance dialysis therapy in whom secondary HPT is not adequately controlled with standard of care therapy.
Mimpara may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate.
Parathyroid carcinoma and primary hyperparathyroidism in adults.
Reduction of hypercalcaemia in adult patients with:
- parathyroid carcinoma;
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of secondary hyperparathyroidism (HPT) in patients with end-stage renal disease (ESRD) on maintenance dialysis therapy.
Mimpara may be used as part of a therapeutic regimen including phosphate binders and/or Vitamin D sterols, as appropriate (see section 5. 1).
Reduction of hypercalcaemia in patients with:
-parathyroid carcinoma.
- primary HPT for whom parathyroidectomy would be indicated on the basis of serum calcium
levels (as defined by relevant treatment guidelines), but in whom parathyroidectomy is not clinically appropriate or is contraindicated.
Treatment of parathyroid carcinoma, Treatment of primary hyperparathyroidism , Treatment of secondary hyperparathyroidism in patients with end-stage renal disease
Calcimimetic Agents
Small organic molecules that act as allosteric activators of the calcium sensing receptor (CaSR) in the PARATHYROID GLANDS and other tissues. They lower the threshold for CaSR activation by extracellular calcium ions and diminish PARATHYROID HORMONE (PTH) release from parathyroid cells. (See all compounds classified as Calcimimetic Agents.)
Calcium-Regulating Hormones and Agents
Hormones and molecules with calcium-regulating hormone-like actions that modulate OSTEOLYSIS and other extra-skeletal activities to maintain calcium homeostasis. (See all compounds classified as Calcium-Regulating Hormones and Agents.)
H05BX01
H05BX01
H05BX01
H05BX01
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2013-09-22
Pay. Date : 2013-07-03
DMF Number : 27211
Submission : 2013-06-28
Status : Active
Type : II

Date of Issue : 2022-06-17
Valid Till : 2025-07-07
Written Confirmation Number : WC-0067
Address of the Firm :

NDC Package Code : 55111-959
Start Marketing Date : 2015-06-10
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : EU, CA, RU |
 ASolution Pharmaceuticals: Your Partner for cGMP Development & Manufacturing Services – Supplying APIs, FDFs, and Specialty Molecules.
ASolution Pharmaceuticals: Your Partner for cGMP Development & Manufacturing Services – Supplying APIs, FDFs, and Specialty Molecules.
 Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.

GDUFA
DMF Review : Reviewed
Rev. Date : 2014-03-25
Pay. Date : 2014-03-14
DMF Number : 21405
Submission : 2008-03-06
Status : Active
Type : II

NDC Package Code : 16436-0082
Start Marketing Date : 2010-06-03
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (75kg/75kg)
Marketing Category : BULK INGREDIENT
 Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.

GDUFA
DMF Review : Reviewed
Rev. Date : 2013-02-23
Pay. Date : 2012-12-24
DMF Number : 24881
Submission : 2011-04-22
Status : Active
Type : II

Date of Issue : 2022-07-27
Valid Till : 2025-07-21
Written Confirmation Number : WC-0087-(Annexure-1)
Address of the Firm :

| Available Reg Filing : ASMF, CA, AU, BR, CN |

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 29494
Submission : 2015-06-24
Status : Active
Type : II

NDC Package Code : 49711-1517
Start Marketing Date : 2014-11-19
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2014-10-30
Pay. Date : 2014-08-04
DMF Number : 28156
Submission : 2014-03-27
Status : Active
Type : II

Date of Issue : 2022-07-15
Valid Till : 2025-07-25
Written Confirmation Number : WC-0126
Address of the Firm :

Registrant Name : V-ON BIO Co., Ltd.
Registration Date : 2020-06-05
Registration Number : No. 4579-5-ND(2)
Manufacturer Name : Solara Active Pharma Sciences Limited
Manufacturer Address : RS No.33 & 34 Mather Road, Periyakalapet, Puducherry-605014. India


GDUFA
DMF Review : Reviewed
Rev. Date : 2016-04-27
Pay. Date : 2015-09-28
DMF Number : 29162
Submission : 2015-03-25
Status : Active
Type : II

Date of Issue : 2022-06-29
Valid Till : 2025-07-02
Written Confirmation Number : WC-0123
Address of the Firm :

NDC Package Code : 65085-0052
Start Marketing Date : 2018-11-21
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (25kg/25kg)
Marketing Category : BULK INGREDIENT

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
GDUFA
DMF Review : Complete
Rev. Date : 2013-09-22
Pay. Date : 2013-07-03
DMF Number : 27211
Submission : 2013-06-28
Status : Active
Type : II
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
GDUFA
DMF Review : Complete
Rev. Date : 2014-03-25
Pay. Date : 2014-03-14
DMF Number : 21405
Submission : 2008-03-06
Status : Active
Type : II
 Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
GDUFA
DMF Review : Complete
Rev. Date : 2013-02-23
Pay. Date : 2012-12-24
DMF Number : 24881
Submission : 2011-04-22
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23910
Submission : 2010-06-23
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-09-30
Pay. Date : 2013-08-21
DMF Number : 27364
Submission : 2013-08-22
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2015-11-06
Pay. Date : 2015-08-21
DMF Number : 28051
Submission : 2014-05-05
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-08-12
Pay. Date : 2014-05-17
DMF Number : 24297
Submission : 2010-10-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 25889
Submission : 2012-03-29
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-10-30
Pay. Date : 2014-08-04
DMF Number : 28156
Submission : 2014-03-27
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-03-31
Pay. Date : 2013-11-19
DMF Number : 27592
Submission : 2013-11-28
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Registration Number : 230MF10003
Registrant's Address : 7-2-A2, Hetero Corporate, Industrial Estates, Sanath Nagar, Hyderabad-500 018, Telang...
Initial Date of Registration : 2018-01-10
Latest Date of Registration : 2018-01-10

Registration Number : 301MF10033
Registrant's Address : 343, Sapyeong-daero, Seocho-gu, Seoul, Korea
Initial Date of Registration : 2019-07-19
Latest Date of Registration : 2019-07-19

Registration Number : 230MF10125
Registrant's Address : #1516, Geumgang Penterium IT Tower, 282, Hagui-ro, Dongan-gu, Anyang-si, Gyeonggi-do,...
Initial Date of Registration : 2018-09-03
Latest Date of Registration : 2020-03-02

Registration Number : 301MF10098
Registrant's Address : 148-1 Tajii, Mihara-ku, Sakai City, Osaka Prefecture
Initial Date of Registration : 2019-11-27
Latest Date of Registration : 2019-11-27

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Date of Issue : 2022-06-17
Valid Till : 2025-07-07
Written Confirmation Number : WC-0067
Address of the Firm : Chemical Technical Operations Unit-VI, APIIC Industrial Estate, Pydibhimavaram V...
 Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
Date of Issue : 2023-01-06
Valid Till : 2025-07-29
Written Confirmation Number : WC-0099
Address of the Firm : Plot No. E-50, Unit-IV, MIDC, Tarapur - 401506, Taluka: Palghar, District: Thane...
 Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
Date of Issue : 2022-07-27
Valid Till : 2025-07-21
Written Confirmation Number : WC-0087-(Annexure-1)
Address of the Firm : Plot No.1 to 5, 31 to 35 & 48 to 51, 26 & K/201, Village Lakhmapur, Taluka Dindo...
Date of Issue : 2019-06-26
Valid Till : 2022-06-25
Written Confirmation Number : WC-0023
Address of the Firm : Sy.No.1/22, 2/1 To 5, 6 to 18, 61 to 69, Pydibhimavaram Village, Ranasthalam, Sr...

Date of Issue : 2022-06-08
Valid Till : 2025-09-08
Written Confirmation Number : WC-0144
Address of the Firm : Plot. D-7 MIDC, Kurkumbh, Dist. Pune-413802

Date of Issue : 2019-07-22
Valid Till : 2022-07-02
Written Confirmation Number : WC-0135
Address of the Firm : Survey No. 47, Paiki Sub Plot No. 1, Village Lodariyal, Taluka Sanand, Dist. Ahm...

Date of Issue : 2019-08-09
Valid Till : 2025-08-08
Written Confirmation Number : WC-0066
Address of the Firm : Plot No. 1, Hetero SEZ Infrastructure Ltd., Narasapuram, Visakhapatnam-531 081, ...

Date of Issue : 2022-06-03
Valid Till : 2025-05-27
Written Confirmation Number : WC-0003
Address of the Firm : Vill.- Bhagwanpur, Barwala Road, Near Dera Bassi, Distt.- SAS Nagar (Mohali), Pu...

Date of Issue : 2019-11-28
Valid Till : 2022-07-02
Written Confirmation Number : WC-0079
Address of the Firm : Plot No.2209, GIDC, Industrial Estate, At & Post Sarigam, Tal-Umbergaon, Valsad-...

Date of Issue : 2020-02-14
Valid Till : 2022-07-02
Written Confirmation Number : WC-0214
Address of the Firm : Gut No. 546, 571, 519 & 520, Village: Kumbhavali, Tarapur, Boisar, Tal & DIst- P...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
About the Company : Founded in 1984, DRL is well-known for its generic APIs and its track record in drug product development. It is one of the earliest pharma API manufacturers with a diverse portfoli...
 ASolution Pharmaceuticals: Your Partner for cGMP Development & Manufacturing Services – Supplying APIs, FDFs, and Specialty Molecules.
ASolution Pharmaceuticals: Your Partner for cGMP Development & Manufacturing Services – Supplying APIs, FDFs, and Specialty Molecules.
About the Company : Since 2010, our dedication to our clientele has been unwavering in addressing pharmaceutical hurdles, regardless of scale. We prioritize integrity, responsiveness, and punctuality,...
 Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
Aarti Pharmalabs is a partner of choice for APIs & Intermediates and the largest Indian manufacturer of Xanthine Derivatives.
About the Company : Aarti Pharmalab, earlier the pharma division of Aarti Industries, is a leading Indian manufacturer of APIs. It has dedicated facilities to manufacture HPAPIs, corticosteroids, cyto...
About the Company : HRV Global is a leading global manufacturer, seller & exporter of a wide range of APIs, advanced intermediates, pellets, food grade chemicals, food additives & food ingredients. It...
 Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
Medichem is a vertically integrated pharmaceutical company specializing in the development & manufacturing of APIs & FDFs.
About the Company : Medichem is an indepently-owned company based in Barcelona. Founded in 1972 and with more than 45 years, Medichem has been engaged in the process development and manufacture of Act...
 Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
Megafine Pharma is engaged in the production of APIs, and advanced intermediates, and offers contract manufacturing services.
About the Company : Megafine, established in 1995, is a privately owned and professionally managed enterprise with its corporate office in MUMBAI and two multipurpose manufacturing sites at VAPI & NAS...
About the Company : Bal Pharma is a leading Indian pharmaceutical company with 30+ years of experience, specializing in prescription drugs, generic and OTC products, intravenous infusions, and bulk ac...
About the Company : Ningbo Zhenlei Chemical Co.,ltd. located in ningbo city, zhejiang province. We are specializes in R&D, manufacturing and marketing. Our main product is R-3-amino-1-butanol, Sitagli...

About the Company : We are Rampex Labs, a fully cGMP compliant pharmaceutical intermediates plant located in the outskirts of Hyderabad, India. Our company was founded in 2004 with commercial operatio...

About the Company : Solara Active Pharma Sciences is a young, dynamic, entrepreneurial and customer oriented API manufacturer. We have a legacy of over three decades and trace our origins to the API e...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Details:
Cinacalcet Tablets is a therapeutic equivalent generic version of Sensipar (cinacalcet) Tablets, approved by the U.S. Food and Drug Administration (USFDA).
Lead Product(s): Cinacalcet Hydrochloride
Therapeutic Area: Endocrinology Brand Name: Undisclosed
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable October 01, 2020
Lead Product(s) : Cinacalcet Hydrochloride
Therapeutic Area : Endocrinology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Dr. Reddy's Laboratories Announces the Launch of Cinacalcet Tablets in the U.S. Market
Details : Cinacalcet Tablets is a therapeutic equivalent generic version of Sensipar (cinacalcet) Tablets, approved by the U.S. Food and Drug Administration (USFDA).
Brand Name : Undisclosed
Molecule Type : Small molecule
Upfront Cash : Not Applicable
October 01, 2020
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results](R )-(+)-1-(1-Naphtyl)-ethylamine Hydrochloride
CAS Number : 82572-04-1
End Use API : Cinacalcet Hydrochloride
About The Company : Minakem is a fully integrated development partner and commercial manufacturer for pharma companies. With around 70 years of experience, Minakem is a technology-...
1 ACETYL NAPHTHALENE (1 ACETONAPHTHONE)
CAS Number : 941-98-0
End Use API : Cinacalcet Hydrochloride
About The Company : DKSH, founded with the goal of improving people's lives, assists businesses with market expansion and business growth in both existing and emerging markets. It ...
CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : Malladi Drugs & Pharmaceuticals Ltd was founded in 1980 by microbiologist Mr. M L N Sastry. Within a few years, Malladi established itself as a leader in manufa...
CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : Established in 2016, a state-of-the-art R&D facility engaged in the development of Novel processes for Bulk Actives including Deuterated Actives, Advanced Inter...

R)-(+)-1-(1-Naphthyl)ethylamine
CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : Arran is an independent fine chemical company located near Athlone, Ireland. Our range of pharmaceutical intermediates includes alkylamines and hydrochlorides, ...

CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : Based on the excellent enzymatic technology platform, Enzymaster Chemical applies innovative technology to realize its commercial production of chiral compounds...

(R)-1-(Naphthalen-1-yl)ethanamine
CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : We believe in attaining the highest levels of customer satisfaction and social welfare with the philosophy of keeping first things first. Our systematic interve...

3-(3'-Trifluoromethylphenyl)propanol
CAS Number : 78573-45-2
End Use API : Cinacalcet Hydrochloride
About The Company : Mehta API Pvt Ltd engaged in manufacturing and marketing of APIs & Advance Drug Intermediates.... Globally. Mehta Api has been inordinately proud of its achieve...

3-(3-(Trifluoromethyl)phenyl)propan-1-ol
CAS Number : 78573-45-2
End Use API : Cinacalcet Hydrochloride
About The Company : RR LIFESCIENCES manufactures products purely by organic synthesis to offer its customers a choice of products in their areas. Our facilities are equipped with a...

R-(+)-l-(l-naphthyl) ethylamine
CAS Number : 3886-70-2
End Use API : Cinacalcet Hydrochloride
About The Company : Founded in 1995, Shenzhen Oriental Pharma focussed on the production of Active Pharma Ingredients (APIs), Now the main product is Phenylephrine Hcl (PE), Doxyla...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Film Coated Tablet
Dosage Strength : 30MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 60MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Cohance Lifesciences, offers full range of CDMO services for small molecule APIs, intermediates, ADCs, Pellets and Formulations.
Regulatory Info :
Registration Country : India
Brand Name :
Dosage Form : Tablet
Dosage Strength : 90MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : India
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Packaging :
Approval Date : 2020-09-18
Application Number : 208368
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 60MG BASE
Packaging :
Approval Date : 2020-09-18
Application Number : 208368
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info : RX
Registration Country : USA
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 90MG BASE
Packaging :
Approval Date : 2020-09-18
Application Number : 208368
Regulatory Info : RX
Registration Country : USA
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 30 mg
Packaging : 28 UNITS 30 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 60 mg
Packaging : 28 UNITS 60 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 90 mg
Packaging : 28 UNITS 90 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
Regulatory Info : RX
Registration Country : USA
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Packaging :
Approval Date : 2018-04-30
Application Number : 209226
Regulatory Info : RX
Registration Country : USA
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Approval Date : 2020-09-18
Application Number : 208368
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 60MG BASE
Approval Date : 2020-09-18
Application Number : 208368
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 90MG BASE
Approval Date : 2020-09-18
Application Number : 208368
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Approval Date : 2018-04-30
Application Number : 209226
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 60MG BASE
Approval Date : 2018-04-30
Application Number : 209226
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 90MG BASE
Approval Date : 2018-04-30
Application Number : 209226
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Approval Date : 2020-05-15
Application Number : 211892
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Approval Date : 2018-12-27
Application Number : 204377
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD :
TE Code :
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET; ORAL
Dosage Strength : 90MG
Approval Date :
Application Number : 90476
RX/OTC/DISCN :
RLD :
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : CINACALCET HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : EQ 30MG BASE
Approval Date : 2018-03-08
Application Number : 208915
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 30 mg
Packaging : 28 UNITS 30 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 60 mg
Packaging : 28 UNITS 60 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET DR REDDY'S
Dosage Form : Film-Coated Tablets
Dosage Strength : 90 mg
Packaging : 28 UNITS 90 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Brand Name : Cinacalcet Accord
Dosage Form : Antic-calc Tablet, Film Coated
Dosage Strength : 90 mg
Packaging : Blister
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Sweden
Brand Name : Mimpara
Dosage Form : FILMDRAGERAD TABLETT
Dosage Strength : 60 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : MIMPARA
Dosage Form : Film-Coated Tablets
Dosage Strength : 30 mg
Packaging : 28 UNITS 30 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : CINACALCET AUROBINDO
Dosage Form : Film-Coated Tablets
Dosage Strength : 30 mg
Packaging : 28 UNITS 30 MG - ORAL USE
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : Turkey
Brand Name :
Dosage Form : Film Coated Tablet
Dosage Strength : 90MG
Packaging :
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : Turkey

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Switzerland
Brand Name : Cinacalcet Devatis
Dosage Form : Filmtabl
Dosage Strength : 30mg
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Switzerland

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Brand Name : Cinacalcet Mylan
Dosage Form : Antic-calc Tablet, Film Coated
Dosage Strength : 90 mg
Packaging : Blister
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Dosage Form : Orodispersible Tablet, Tablet
Grade : Oral
Category : Chewable & Orodispersible Aids, Direct Compression, Taste Masking
Dosage Form : Orodispersible Tablet, Tablet
Grade : Oral
Category : Chewable & Orodispersible Aids, Direct Compression, Taste Masking
Application : Chewable & Orodispersible Aids, Direct Compression, Taste Masking
Excipient Details : MS90 is a directly compressible magnesium hydroxide with starch used for chewable tablets due to its smooth mouthfeel and creamy texture.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 150-170, Taped Density: 0.80
Ingredient(s) : Starch
Dosage Form : Orodispersible Tablet, Tablet
Grade : Oral
Category : Chewable & Orodispersible Aids, Direct Compression, Taste Masking
Application : Chewable & Orodispersible Aids, Direct Compression, Taste Masking
Excipient Details : CS90 is a directly compressible calcium carbonate with starch used for chewable tablets due to its smooth mouthfeel and creamy texture.
Pharmacopoeia Ref : NA
Technical Specs : PSD D50: 150-175, Tapped Density: 0.85
Ingredient(s) : Calcium Carbonate Excipient
Dosage Form : Orodispersible Tablet, Tablet
Grade : Oral
Category : Chewable & Orodispersible Aids, Taste Masking
Application : Chewable & Orodispersible Aids, Taste Masking
Excipient Details : HiCel CE15 offers a superior mouthfeel with less chalkiness and gritness in chewable tablets and orally disintegrating tablets (ODTs).
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Guar Gum Excipient
Dosage Form : Orodispersible Tablet, Tablet
Grade : Oral
Category : Disintegrants & Superdisintegrants, Fillers, Diluents & Binders, Taste Masking
Application : Disintegrants & Superdisintegrants, Fillers, Diluents & Binders, Taste Masking
Excipient Details : F-Melt Type C is a pharmaceutical excipient used in oral dosage forms like orally disintegrating tablets, sachets, dispersible tablets, chewable tablets and sublingual tablets.
Pharmacopoeia Ref : Conforms to Japanese Pharmaceu...
Technical Specs : Not Available
Ingredient(s) : Crospovidone
Global Sales Information

Market Place

Reply
06 Jul 2020
Reply
17 Dec 2019
Reply
28 Sep 2019
Reply
07 Aug 2018

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 43 companies offering Cinacalcet Hydrochloride
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
