
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
VMF
DRUG PRODUCT COMPOSITIONS
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
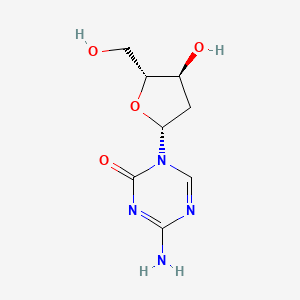

1. 2' Deoxy 5 Azacytidine
2. 2'-deoxy-5-azacytidine
3. 5 Aza 2' Deoxycytidine
4. 5 Azadeoxycytidine
5. 5 Deoxyazacytidine
6. 5-aza-2'-deoxycytidine
7. 5-azadc
8. 5-azadeoxycytidine
9. 5-deoxyazacytidine
10. 5azadc
11. Azadc Compound
12. Compound, Azadc
13. Dacogen
14. Decitabine Mesylate
15. Mesylate, Decitabine
16. Nsc 127716
17. Nsc-127716
18. Nsc127716
1. 5-aza-2'-deoxycytidine
2. 2353-33-5
3. Dacogen
4. 2'-deoxy-5-azacytidine
5. 5-azadeoxycytidine
6. Azadc
7. 5-aza-cdr
8. 5-aza-dc
9. Dezocitidine
10. Nsc 127716
11. Dac
12. 5-aza-deoxycytidine
13. Alpha-decitibine
14. 4-amino-1-((2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1,3,5-triazin-2(1h)-one
15. Nsc-127716
16. 4-amino-1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,3,5-triazin-2-one
17. 5a2dc
18. 5-aza-2-deoxycytidine
19. 4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-s-triazin-2(1h)-one
20. 4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-1,3,5-triazin-2(1h)-one
21. Mls001332587
22. Chebi:50131
23. 776b62cq27
24. Smr000857076
25. 4-amino-1-[(2s,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,3,5-triazin-2-one
26. Dsstox_cid_10432
27. Dsstox_rid_78849
28. Dsstox_gsid_30432
29. 4-amino-1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-1,2-dihydro-1,3,5-triazin-2-one
30. 5-azadc
31. Nsc127716
32. 2-deoxyazacytidine
33. 4-amino-1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-1,3,5-triazin-2-one
34. Dacogen (tn)
35. Cas-2353-33-5
36. 2-desoxy-5-azacytidine
37. Ncgc_5adoc
38. Decitabine (usan/inn)
39. Decitabine [usan:inn:ban]
40. Ccris 8227
41. Unii-776b62cq27
42. Ncgc00166088-01
43. 22432-95-7
44. Decitabine- Bio-x
45. Deoxy-5-azacytidine
46. Einecs 219-089-4
47. Mfcd00043011
48. Brn 0617982
49. Decitabine, Free Base
50. Jnj 30979754
51. Jnj-30979754
52. Decitabine [mi]
53. Decitabine [inn]
54. Decitabine [usan]
55. 4-amino-1-(2-deoxy-beta-d-ribofuranosyl)-1,3,5-triazin-2(1h)-one
56. Decitabine [vandf]
57. Molmap_000063
58. E-7373
59. Decitabine [mart.]
60. Schembl4006
61. Decitabine [who-dd]
62. Mls001332588
63. Mls006010136
64. Astx-727
65. Cid_451668
66. Gtpl6805
67. Chembl1201129
68. Decitabine [orange Book]
69. Dtxsid7030432
70. Bdbm96274
71. Ex-a961
72. Inqovi Component Decitabine
73. Hms2235o03
74. Hms3413l07
75. Hms3677l07
76. 105597-46-4
77. 4-amino-1-((2s,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-1,3,5-triazin-2(1h)-one
78. Act00147
79. Amy33354
80. Bcp02870
81. Hy-a0004
82. 5-aza-2'-deoxycytidine, >=97%
83. Tox21_112311
84. Astx727 Component Decitabine
85. S1200
86. Zinc16929327
87. Decitabine Component Of Inqovi
88. 4-amino-1-(2-deoxy-?-d-erythro-pentofuranosyl)-1,3,5-triazin-2(1h)-one
89. Akos015895047
90. Astx-727 Component Decitabine
91. Inqovi (decitabine + Cedazuridine)
92. Tox21_112311_1
93. Ac-1135
94. Bcp9000593
95. Ccg-208143
96. Cs-0372
97. Db01262
98. 1,3,5-triazin-2(1h)-one, 4-amino-1-(2-deoxy-.beta.-d-erythro-pentofuranosyl)-
99. Astx727 (decitabine + Cedazuridine)
100. Decitabine (nsc127716, 5aza-cdr)
101. Decitabine (nsc127716; 5aza-cdr)
102. Ncgc00166088-02
103. Ncgc00166088-05
104. As-17558
105. Ba164359
106. Bcp0726000271
107. Db-029060
108. Sw218076-2
109. D03665
110. Ab00918337-07
111. Ab00918337_08
112. 353a335
113. Sr-01000838879
114. J-700084
115. Q1181878
116. Sr-01000838879-4
117. Decitabine (2 Inverted Exclamation Marka-deoxy-5-azacytidine).cd
118. S-triazin-2(1h)-one, 4-amino-1-(2-deoxy-beta-d-erythro-pentofuranosyl)-
119. 1,3, 5-triazin-2(1h)-one, 4-amino-1-(2-deoxy--d-erythro-pentofuranosyl)-
120. 4-amino-1-(2-deoxy-.beta.-d-erythro-pentofuranosyl)-s-triazin-2(1h)-one
| Molecular Weight | 228.21 g/mol |
|---|---|
| Molecular Formula | C8H12N4O4 |
| XLogP3 | -1.2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Exact Mass | 228.08585488 g/mol |
| Monoisotopic Mass | 228.08585488 g/mol |
| Topological Polar Surface Area | 121 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 356 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Dacogen |
| PubMed Health | Decitabine (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Dacogen (decitabine) for Injection contains decitabine (5-aza-2-deoxycitidine), an analogue of the natural nucleoside 2-deoxycytidine. Decitabine is a fine, white to almost white powder with the molecular formula of C8H12N4O4 and a mo... |
| Active Ingredient | Decitabine |
| Dosage Form | Injectable |
| Route | Intravenous |
| Strength | 50mg/vial |
| Market Status | Prescription |
| Company | Eisai |
| 2 of 4 | |
|---|---|
| Drug Name | Decitabine |
| PubMed Health | Decitabine (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Decitabine for Injection contains decitabine (5-aza-2deoxycitidine), an analogue of the natural nucleoside 2-deoxycytidine. Decitabine is a fine, white to almost white powder with the molecular formula of C8H12N4O4 and a molecular weight of 228... |
| Active Ingredient | Decitabine |
| Dosage Form | Injectable; Powder |
| Route | Intravenous |
| Strength | 50mg/vial |
| Market Status | Prescription |
| Company | Dr Reddys Labs; Sandoz; Sun Pharma Global |
| 3 of 4 | |
|---|---|
| Drug Name | Dacogen |
| PubMed Health | Decitabine (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Dacogen (decitabine) for Injection contains decitabine (5-aza-2-deoxycitidine), an analogue of the natural nucleoside 2-deoxycytidine. Decitabine is a fine, white to almost white powder with the molecular formula of C8H12N4O4 and a mo... |
| Active Ingredient | Decitabine |
| Dosage Form | Injectable |
| Route | Intravenous |
| Strength | 50mg/vial |
| Market Status | Prescription |
| Company | Eisai |
| 4 of 4 | |
|---|---|
| Drug Name | Decitabine |
| PubMed Health | Decitabine (Injection) |
| Drug Classes | Antineoplastic Agent |
| Drug Label | Decitabine for Injection contains decitabine (5-aza-2deoxycitidine), an analogue of the natural nucleoside 2-deoxycytidine. Decitabine is a fine, white to almost white powder with the molecular formula of C8H12N4O4 and a molecular weight of 228... |
| Active Ingredient | Decitabine |
| Dosage Form | Injectable; Powder |
| Route | Intravenous |
| Strength | 50mg/vial |
| Market Status | Prescription |
| Company | Dr Reddys Labs; Sandoz; Sun Pharma Global |
Decitabine is indicated for the treatment of patients with myelodysplastic syndromes (MDS) including all French-American-British subtypes (refractory anemia, refractory anemia with ringed sideroblasts, refractory anemia with excess blasts, refractory anemia with excess blasts in transformation, and chronic myelomonocytic leukemia), as well as for MDS scored as belonging to the intermediate-1, intermediate-2, or high-risk group in the International Prognostic Scoring System.
FDA Label
Treatment of adult patients with newly diagnosed de novo or secondary acute myeloid leukaemia (AML), according to the World Health Organization (WHO) classification, who are not candidates for standard induction chemotherapy.
Decitabine is a prodrug analogue of the natural nucleotide 2-deoxycytidine, which, upon being phosphorylated intracellularly, is incorporated into DNA and exerts numerous effects on gene expression. The use of decitabine is associated with neutropenia and thrombocytopenia. In addition, decitabine can cause fetal harm in pregnant women; effective contraception and avoidance of pregnancy are recommended during treatment with decitabine.
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Antimetabolites, Antineoplastic
Antimetabolites that are useful in cancer chemotherapy. (See all compounds classified as Antimetabolites, Antineoplastic.)
L01BC08
L01BC08
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L01 - Antineoplastic agents
L01B - Antimetabolites
L01BC - Pyrimidine analogues
L01BC08 - Decitabine
Absorption
Decitabine administered intravenously at 15 mg/m2 for three hours every eight hours over three days resulted in a Cmax of 73.8 ng/mL (66% coefficient of variation, CV), an AUC0- of 163 ng\*h/mL (62% CV), and a cumulative AUC of 1332 ng\*h/mL (95% CI of 1010-1730). Similarly, decitabine at 20 mg/m2 for one hour once daily over five days resulted in a Cmax of 147 ng/mL (49% CV), an AUC0- of 115 ng\*h/mL (43% CV), and a cumulative AUC of 570 ng\*h/mL (95% CI of 470-700).
Route of Elimination
Less than 1% of administered decitabine is excreted in the urine.
Volume of Distribution
Decitabine as an apparent volume of distribution of 4.59 1.42 L/kg.
Clearance
Decitabine has a clearance of 125 L/hr/m2 (53% CV) when administered intravenously at 15 mg/m2 for three hours every eight hours over three days, and a clearance of 210 L/hr/m2 (47% CV) at 20 mg/m2 for one hour once daily over five days.
Decitabine is phosphorylated inside cells by the sequential action of deoxycytidine kinase, nucleotide monophosphate kinase, and nucleotide diphosphate kinase, prior to being incorporated into newly synthesized DNA by DNA polymerase. Decitabine not incorporated into cellular DNA undergoes deamination by cytidine deaminase followed by additional degradation prior to excretion.
Decitabine has a half-life of 0.62 hours (49% CV) when administered intravenously at 15 mg/m2 for three hours every eight hours over three days, and a half-life of 0.54 hours (43% CV) at 20 mg/m2 for one hour once daily over five days.
Myelodysplastic syndromes (MDS) are a group of hematopoietic neoplasms that manifest in peripheral cytopenias and may eventually progress to secondary acute myeloid leukemia (sAML). Included in the over 45 genes commonly mutated in MDS patients are those involved in DNA methylation and histone modification, and it is well-established that alteration of the epigenetic landscape is a feature of myeloid leukemias. Decitabine is considered a prodrug, as it requires transport into cells and subsequent phosphorylation by distinct kinases to generate the active molecule 5-aza-2'-deoxycytidine-triphosphate, which is incorporated by DNA polymerase during DNA replication. Once incorporated into DNA, decitabine is recognized as a substrate by DNA methyltransferase enzymes (DNMTs), specifically DNMT1, but due to the presence of an N5 rather than C5 atom, traps the DNMT through the irreversible formation of a covalent bond. At low concentrations, this mode of action depletes DNMTs and results in global DNA hypomethylation while at high concentrations, it additionally results in double-strand breaks and cell death. The general hypothesis regarding decitabine's therapeutic efficacy is that the global hypomethylation it induces results in the expression of previously silent tumour suppressor genes. However, there are other putative mechanisms also related to this change in DNA methylation, including indirect alteration of transcription through effects on transcription factors, indirectly altering histone modifications and chromatin structure, and activating pathways involved in DNA damage response. The overall effect of decitabine is a decrease in neoplastic cell proliferation and an increase in the expression of tumour suppressor genes.
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
54
PharmaCompass offers a list of Decitabine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Decitabine manufacturer or Decitabine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Decitabine manufacturer or Decitabine supplier.
A Decitabine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Decitabine, including repackagers and relabelers. The FDA regulates Decitabine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Decitabine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Decitabine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Decitabine supplier is an individual or a company that provides Decitabine active pharmaceutical ingredient (API) or Decitabine finished formulations upon request. The Decitabine suppliers may include Decitabine API manufacturers, exporters, distributors and traders.
click here to find a list of Decitabine suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Decitabine DMF (Drug Master File) is a document detailing the whole manufacturing process of Decitabine active pharmaceutical ingredient (API) in detail. Different forms of Decitabine DMFs exist exist since differing nations have different regulations, such as Decitabine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Decitabine DMF submitted to regulatory agencies in the US is known as a USDMF. Decitabine USDMF includes data on Decitabine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Decitabine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Decitabine suppliers with USDMF on PharmaCompass.
In Korea, the Ministry of Food and Drug Safety (MFDS) is in charge of regulating pharmaceutical products and services.
Pharmaceutical companies submit a Decitabine Drug Master File in Korea (Decitabine KDMF) to the MFDS, which includes comprehensive information about the production, processing, facilities, materials, packaging, and testing of Decitabine. The MFDS reviews the Decitabine KDMF as part of the drug registration process and uses the information provided in the Decitabine KDMF to evaluate the safety and efficacy of the drug.
After submitting a Decitabine KDMF to the MFDS, the registered manufacturer can provide importers or distributors with the registration number without revealing confidential information to Korean business partners. Applicants seeking to register their Decitabine API can apply through the Korea Drug Master File (KDMF).
click here to find a list of Decitabine suppliers with KDMF on PharmaCompass.
A Decitabine written confirmation (Decitabine WC) is an official document issued by a regulatory agency to a Decitabine manufacturer, verifying that the manufacturing facility of a Decitabine active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Decitabine APIs or Decitabine finished pharmaceutical products to another nation, regulatory agencies frequently require a Decitabine WC (written confirmation) as part of the regulatory process.
click here to find a list of Decitabine suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Decitabine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Decitabine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Decitabine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Decitabine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Decitabine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Decitabine suppliers with NDC on PharmaCompass.
Decitabine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Decitabine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Decitabine GMP manufacturer or Decitabine GMP API supplier for your needs.
A Decitabine CoA (Certificate of Analysis) is a formal document that attests to Decitabine's compliance with Decitabine specifications and serves as a tool for batch-level quality control.
Decitabine CoA mostly includes findings from lab analyses of a specific batch. For each Decitabine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Decitabine may be tested according to a variety of international standards, such as European Pharmacopoeia (Decitabine EP), Decitabine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Decitabine USP).

