
Synopsis
Synopsis
0
KDMF
0
VMF
0
Australia

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
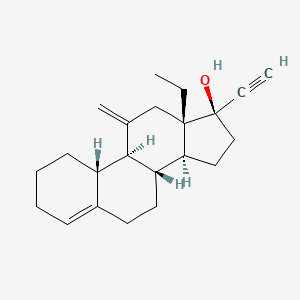

1. 13 Ethyl 11 Methylene 18,19 Dinor 17 Alpha Pregn 4 En 20 Yn 17 Ol
2. 13-ethyl-11-methylene-18,19-dinor-17 Alpha-pregn-4-en-20-yn-17-ol
3. 18,19-dinorpregn-4-en-20-yn-17-ol, 13-ethyl-11-methylene-, (17alpha)-
4. Alpha-pregn-4-en-20-yn-17-ol, 13-ethyl-11-methylene-18,19-dinor-17
5. Cerazette
6. Marvelon
7. Org 2969
8. Org-2969
9. Org2969
1. 54024-22-5
2. Cerazette
3. Desogen
4. Desogestrelum
5. Org-2969
6. Mircette
7. Org 2969
8. Desogestrelum [inn-latin]
9. 13-ethyl-11-methylene-18,19-dinor-17alpha-pregn-4-en-20-yn-17-ol
10. (8s,9s,10r,13s,14s,17r)-13-ethyl-17-ethynyl-11-methylidene-1,2,3,6,7,8,9,10,12,14,15,16-dodecahydrocyclopenta[a]phenanthren-17-ol
11. Chebi:4453
12. 81k9v7m3a3
13. Ncgc00167449-01
14. 13-ethyl-11-methylene-18,19-dinorpregn-4-en-20-yn-17alpha-ol
15. Hsdb 3593
16. Sr-01000883959
17. Einecs 258-929-4
18. Desogestrel (usan/inn)
19. Unii-81k9v7m3a3
20. Desogestrel [usan:inn:ban]
21. 18,19-dinorpregn-4-en-20-yn-17-ol, 13-ethyl-11-methylene-, (17.alpha.)-
22. Mfcd00869346
23. Desogestrel [mi]
24. Desogestrel [inn]
25. Desogestrel [jan]
26. Dsstox_cid_2898
27. Desogestrel [hsdb]
28. Desogestrel [usan]
29. 18,19-dinorpregn-4-en-20-yn-17-ol, 13-ethyl-11-methylene-, (17alpha)-
30. Desogestrel [vandf]
31. Chembl1533
32. Desogestrel [mart.]
33. Dsstox_rid_76779
34. Dsstox_gsid_22898
35. Schembl41341
36. Desogestrel [usp-rs]
37. Desogestrel [who-dd]
38. Gtpl7065
39. Dtxsid6022898
40. Desogestrel [orange Book]
41. 17alpha-ethynyl-11-methylidene-18a-homo-estr-4-en-17beta-ol
42. Desogestrel [ep Monograph]
43. Desogestrel [usp Monograph]
44. Kariva Component Desogestrel
45. Volnea Component Desogestrel
46. Bcp18794
47. Bekyree Component Desogestrel
48. Desogen Component Desogestrel
49. Enskyce Component Desogestrel
50. Pimtrea Component Desogestrel
51. Velivet Component Desogestrel
52. Viorele Component Desogestrel
53. Zinc4097416
54. Tox21_112453
55. 13-ethyl-11-methylene-18,19-dinor-17a-pregn-4-en-20-yn-17-ol
56. Ac-308
57. Bdbm50423510
58. Cyclessa Component Desogestrel
59. Isibloom Component Desogestrel
60. Lmst02030104
61. Mircette Component Desogestrel
62. S4638
63. (17alpha)-13-ethyl-11-methylene-18,19-dinorpregn-4-en-20-yn-17-ol
64. Akos015963198
65. Desogestrel Component Of Kariva
66. Ccg-267585
67. Cs-3686
68. Db00304
69. Desogestrel Component Of Desogen
70. Desogestrel Component Of Velivet
71. Ortho-cept Component Desogestrel
72. Desogestrel Component Of Cyclessa
73. Desogestrel Component Of Mircette
74. As-13022
75. Hy-12516
76. Desogestrel 100 Microg/ml In Acetonitrile
77. Desogestrel Component Of Ortho-cept
78. Cas-54024-22-5
79. B4976
80. D4163
81. C07629
82. C15951
83. D02367
84. Desogestrel, Vetranal(tm), Analytical Standard
85. 024d225
86. Q415304
87. J-520217
88. Sr-01000883959-1
89. Sr-01000883959-2
90. 17alpha-ethynyl-13beta-ethyl-11-methylene-4-gonen-17-ol
91. Desogestrel, British Pharmacopoeia (bp) Reference Standard
92. Desogestrel, European Pharmacopoeia (ep) Reference Standard
93. Desogestrel, United States Pharmacopeia (usp) Reference Standard
94. Desogestrel, Pharmaceutical Secondary Standard; Certified Reference Material
95. Desogestrel For System Suitability, European Pharmacopoeia (ep) Reference Standard
| Molecular Weight | 310.5 g/mol |
|---|---|
| Molecular Formula | C22H30O |
| XLogP3 | 4.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Exact Mass | 310.229665576 g/mol |
| Monoisotopic Mass | 310.229665576 g/mol |
| Topological Polar Surface Area | 20.2 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 605 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 6 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Desogen |
| Active Ingredient | Desogestrel; ethinyl estradiol |
| Dosage Form | Tablet |
| Route | Oral-28 |
| Strength | 0.03mg; 0.15mg |
| Market Status | Prescription |
| Company | Organon Usa |
| 2 of 2 | |
|---|---|
| Drug Name | Desogen |
| Active Ingredient | Desogestrel; ethinyl estradiol |
| Dosage Form | Tablet |
| Route | Oral-28 |
| Strength | 0.03mg; 0.15mg |
| Market Status | Prescription |
| Company | Organon Usa |
Contraceptives, Oral, Synthetic; Progestational Hormones, Synthetic
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
Apri (desogestrel and ethinyl estradiol) Tablets are indicated for the prevention of pregnancy in women who elect to use oral contraceptives as a method of contraception. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Cerazette /is indicated for/ contraception.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
Desogestrel is an synthetic progestogen structurally related to levonorgestrel, with actions and uses similar to those of progestogens in general. It is reported to have potent progestogenic activity and little or no androgenic activity. It is used as the progestogenic component of combined mono- and multiphasic oral contraceptive preparations and as a subdermal implantable 'progestogen-only' contraceptive.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V72 587 (1999)
Cigarette smoking increases the risk of serious cardiovascular side effects from oral contraceptive use. This risk increases with age and with heavy smoking (15 or more cigarettes per day) and is quite marked in women over 35 years of age. Women who use oral contraceptives should be strongly advised not to smoke.
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Oral contraceptives like Apri (desogestrel and ethinyl estradiol) tablets should not be used in women who currently have the following conditions: thrombophlebitis or thromboembolic disorders; a past history of deep vein thrombophlebitis or thromboembolic disorders; cerebral vascular or coronary artery disease; known or suspected carcinoma of the breast; carcinoma of the endometrium or other known or suspected estrogen-dependent neoplasia; undiagnosed abnormal genital bleeding; cholestatic jaundice of pregnancy or jaundice with prior pill use; hepatic adenomas or carcinomas...
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Cerazette does not influence the production or the quality (protein, lactose, or fat concentrations) of breast milk. However, small amounts of etonogestrel are excreted in the breast milk. As a result, 0.01 - 0.05 ug etonogestrel per kg body weight per day may be ingested by the child (based on an estimated milk ingestion of 150 mL/kg/day).
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
Epidemiological investigations have associated the use of combined oral contraceptives (COC) with an increased incidence of venous thromboembolism (VTE, deep venous thrombosis and pulmonary embolism). Although the clinical relevance of this finding for desogestrel used as a contraceptive in the absence of an estrogenic component is unknown, Cerazette should be discontinued in the event of a thrombosis. Discontinuation of Cerazette should also be considered in case of long-term immobilization due to surgery or illness. Women with a history of thrombo-embolic disorders should be made aware of the possibility of a recurrence.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
For more Drug Warnings (Complete) data for DESOGESTREL (48 total), please visit the HSDB record page.
Oral desogestrel is used in combination with [ethinylestradiol] as a contraceptive agent for the prevention of pregnancy. Desogestrel is part of the combined oral contraceptives that contain a mix of estrogen and progestin which inhibit ovulation.
FDA Label
The effects of desogestrel are divided on reproductive including modification of luteinizing hormone and follicle stimulating hormone, declines on the onset of menstruation, and increases the viscosity of the vaginal fluid; and on metabolic that includes increase insulin secretion and resistance, increased lipase activity, and increased fat deposition. The effect of desogestrel on the lipids has been studied extensively and the results are contradictory. Desogestrel main therapeutic effect due to its mechanism of action is known to be related to the inhibition of the ovulation in 97% of the cycles. This effect was proven in clinical trials in non-breastfeeding women from which the Pearl failure rate was reported to be of 0.17 per 100 women-years. This result indicated that desogestrel is more efficient when compared to other progestogen-only pills. All the therapeutic effect is produced by a transformation of the endometrium followed by an inhibition of the ovulation due to the suppression of other hormones. Desogestrel has been widely confirmed to be related to an increase in the risk of venous thromboembolism due to the driven increased in blood coagulation factors, leading to a pronounced prothrombotic state. However, the effects of desogestrel are known to not impact significantly the level of total cholesterol remaining in the range of change of 10% which allows it to be a molecule that presents a favorable lipid profile.
Contraceptives, Oral, Hormonal
Oral contraceptives which owe their effectiveness to hormonal preparations. (See all compounds classified as Contraceptives, Oral, Hormonal.)
Progestins
Compounds that interact with PROGESTERONE RECEPTORS in target tissues to bring about the effects similar to those of PROGESTERONE. Primary actions of progestins, including natural and synthetic steroids, are on the UTERUS and the MAMMARY GLAND in preparation for and in maintenance of PREGNANCY. (See all compounds classified as Progestins.)
Contraceptives, Oral, Synthetic
Oral contraceptives which owe their effectiveness to synthetic preparations. (See all compounds classified as Contraceptives, Oral, Synthetic.)
G03AC09
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
G - Genito urinary system and sex hormones
G03 - Sex hormones and modulators of the genital system
G03A - Hormonal contraceptives for systemic use
G03AC - Progestogens
G03AC09 - Desogestrel
Absorption
After oral administration, desogestrel is rapidly absorbed and it reaches a peak concentration of 2 ng/ml after 1.5 hours. The bioavailability of desogestrel is reported to be in the range of 60-80% and the reported AUC is of 3000 ng.h/ml. Almost all the administered dose is modified to the active metabolite, [etonogestrel].
Route of Elimination
The elimination of desogestrel is found to be mainly renal corresponding to about 6 times the dose eliminated in the bile. The elimination of desogestrel is only done as the metabolites and not as the unchanged drug and about 85% of the administered dose can be excreted as metabolites after 6-8 days.
Volume of Distribution
The apparent volume of distribution of desogestrel is of 1.5 L/kg.
Clearance
The metabolic clearance rate of desogestrel is reported to be of about 2 ml/min/kg.
After oral dosing of Cerazette desogestrel (DSG) is rapidly absorbed and converted into etonogestrel (ENG). Under steady-state conditions, peak serum levels are reached 1.8 hours after tablet-intake and the absolute bioavailability of ENG is approximately 70%.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
In the third cycle of use after a single desogestrel and ethinyl estradiol tablet, maximum concentrations of 3-keto-desogestrel of 2,805 +/- 1,203 pg/mL (mean+/-SD) are reached at 1.4+/-0.8 hours. The area under the curve (AUC) is 33,858+/-11,043 pg/mL (hr) after a single dose.
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
At steady state, attained from at least day 19 onwards, maximum concentrations of 5,840 +/-1,667 pg/mL are reached at 1.4+/-0.9 hours. The minimum plasma levels of 3-keto-desogestrel at steady state are 1,400+/-560 pg/mL. The AUC0-24 at steady state is 52,299+/-17,878 pg/mL (hr). The mean AUC0 for 3-keto-desogestrel at single dose is significantly lower than the mean AUC0-24 at steady state. This indicates that the kinetics of 3-keto-desogestrel are non-linear due to an increase in binding of 3-keto-desogestrel to sex hormone-binding globulin in the cycle, attributed to increased sex hormone-binding globulin levels which are induced by the daily administration of ethinyl estradiol. Sex hormone-binding globulin levels increased significantly in the third treatment cycle from day 1 (150+/-64 nmol/L) to day 21 (230+/-59 nmol/L).
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Etonogestrel is 95.5-99% bound to serum proteins, predominantly to albumin and to a lesser extent to sex hormone-binding globulin (SHBG).
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
For more Absorption, Distribution and Excretion (Complete) data for DESOGESTREL (8 total), please visit the HSDB record page.
Desogestrel is rapidly metabolized in the intestinal mucosa and by first-pass hepatic metabolism to form the major metabolite of desogestrel is [etonogestrel] which is the biologically active metabolite. This modification is described by the hydroxylation in C3 of the desogestrel molecule. Later, etonogestrel is metabolized following the normal pathways of steroid metabolism. On the other hand, due to the 11-methylene side chain, desogestrel cannot be metabolized to other progestins.
In addition to 3-keto-desogestrel, other phase I metabolites are 3alpha-OH-desogestrel, 3beta-OH-desogestrel, and 3alpha-OH-5alpha-H-desogestrel. These other metabolites are not known to have any pharmacologic effects, and are further converted in part by conjugation (phase II metabolism) into polar metabolites, mainly sulfates and glucuronides.
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Desogestrel is metabolized via hydroxylation and dehydrogenation to the active metabolite etonogestrel. Etonogestrel is metabolised via sulphate and glucuronide conjugation.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
Desogestrel is metabolized rapidly and completely in the liver and gut wall. It is metabolized to 3-keto-desogestrel, which mediates its progestogenic effects, and it is not metabolized further to another progestogen. The serum concentrations of 3-keto-desogestrel reached maximum levels within 2-3 hours after oral administration of desogestrel and were subsequently cleared with a half-life of 12-24 hours.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V72 227 (1999)
The metabolism of desogestrel in microsomes from six hours livers in vitro /were studied/. The main metabolite formed was 3-keto-desogestrel; 3alpha-hydroxydesogestrel and 3beta-hydroxydesogestrel were also detected. The metabolism of desogestrel was inhibited by 50% by primaquine at a concentration of 30 umol/L, but not by levonorgestrel at 250 umol/L.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V72 227 (1999)
For more Metabolism/Metabolites (Complete) data for DESOGESTREL (7 total), please visit the HSDB record page.
Desogestrel has known human metabolites that include 3-alpha-hydroxydesogestrel, 3-beta-hydroxy-desogestrel, and Desogestrel 17-O-glucuronide.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
The terminal half-life of desogestrel is determined to be of 30 hours.
Etonogestrel is eliminated with a mean half-life of approximately 30 hours, with no difference between single and multiple dosing.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
The elimination half-life for 3-keto-desogestrel is approximately 38+/-20 hours at steady state. /3-Keto-desogestrel/
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
Desogestrel enters the cell passively and acts by binding selectively to the progesterone receptor and generating low androgenic activity. Its binding produces an effect like a transcription factor and thus, it produces modifications in the mRNA synthesis. The active metabolite of desogestrel, [etonogestrel], presents a combination of high progestational activity with minimal intrinsic androgenicity.
Combination oral contraceptives act by suppression of gonadotropins. Although the primary mechanism of this action is inhibition of ovulation, other alterations include changes in the cervical mucus, which increase the difficulty of sperm entry into the uterus, and changes in the endometrium which reduce the likelihood of implantation. Receptor binding studies, as well as studies in animals, have shown that 3-keto-desogestrel, the biologically active metabolite of desogestrel, combines high progestational activity with minimal intrinsic androgenicity. The relevance of this latter finding in humans is unknown.
US Natl Inst Health; DailyMed. Current Medication Information APRI (desogestrel and ethinyl estradiol) kit (January 2011). Available from, as of March 22, 2010: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?id=37841
In contrast to traditional progestogen-only pills, the contraceptive effect of Cerazette is achieved primarily by inhibition of ovulation. Other effects include increased viscosity of the cervical mucus.
Datapharm Communications Ltd; Electronic Medicines Compendium (eMC), Summary of Product Characteristics (SPC) for A Cerazette film-coated tablet (Last updated December 2007). Available from, as of March 23, 2011: https://www.medicines.org.uk/EMC/medicine/10098/SPC/Cerazette+75+microgram+film-coated+tablet/
Recent studies have demonstrated that desogestrel activates the estrogen receptor-alpha at an activity of about 50% of that of 17beta-estradiol but activates the estrogen receptor-beta at an activity of only 20%. Desogestrel and/or its metabolite 3-keto-desogestrel (etonogestrel) were strongly progestogenic (approximately twofold over progesterone), weakly or not androgenic in animal studies in vivo and in-vitro binding assays and weakly or not active on the glucocorticoid receptor. The active metabolite of desogestrel, 3-ketodesogestrel, strongly bound to and activated progesterone receptor-A and, to a slightly lesser extent, progesterone receptor-B
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V91 163 (2007)
Improvement in oral contraceptive formulations was originally achieved through dose reduction of the estrogen and progestogen components. Recently, further improvement was achieved by increasing the selectivity of contraceptive progestins. The ratio between the affinity for the progesterone receptor and the affinity for the androgen receptor is an indicator of progesterone (or androgen) selectivity of a progestin. This ratio (selectivity index) reflects the relative amount of androgenic or progestational effect at a given dose. Relative selectivity can be characterized with in vitro receptor-binding studies and animal pharmacologic experiments. In comparison with levonorgestrel, desogestrel displays markedly lower androgenicity and slightly increased relative progestational activity. In receptor-binding experiments and animal pharmacologic studies, 3-keto-desogestrel, the active metabolite of desogestrel, shows the highest selectivity index. The favorable effect of desogestrel-containing oral contraceptives on lipoprotein metabolism and preexisting androgen-dependent skin disorders and the absence of adverse effects on blood pressure and body weight are attributed to the increased progestin selectivity of desogestrel.
PMID:8447353 Collins D; Am J Obstet Gynecol 168 (3 Pt 2): 1010-6 (1993)
For more Mechanism of Action (Complete) data for DESOGESTREL (6 total), please visit the HSDB record page.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
99
PharmaCompass offers a list of Desogestrel API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Desogestrel manufacturer or Desogestrel supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Desogestrel manufacturer or Desogestrel supplier.
PharmaCompass also assists you with knowing the Desogestrel API Price utilized in the formulation of products. Desogestrel API Price is not always fixed or binding as the Desogestrel Price is obtained through a variety of data sources. The Desogestrel Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Desogestrel manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Desogestrel, including repackagers and relabelers. The FDA regulates Desogestrel manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Desogestrel API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Desogestrel manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Desogestrel supplier is an individual or a company that provides Desogestrel active pharmaceutical ingredient (API) or Desogestrel finished formulations upon request. The Desogestrel suppliers may include Desogestrel API manufacturers, exporters, distributors and traders.
click here to find a list of Desogestrel suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Desogestrel DMF (Drug Master File) is a document detailing the whole manufacturing process of Desogestrel active pharmaceutical ingredient (API) in detail. Different forms of Desogestrel DMFs exist exist since differing nations have different regulations, such as Desogestrel USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Desogestrel DMF submitted to regulatory agencies in the US is known as a USDMF. Desogestrel USDMF includes data on Desogestrel's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Desogestrel USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Desogestrel suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Desogestrel Drug Master File in Japan (Desogestrel JDMF) empowers Desogestrel API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Desogestrel JDMF during the approval evaluation for pharmaceutical products. At the time of Desogestrel JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Desogestrel suppliers with JDMF on PharmaCompass.
A Desogestrel CEP of the European Pharmacopoeia monograph is often referred to as a Desogestrel Certificate of Suitability (COS). The purpose of a Desogestrel CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Desogestrel EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Desogestrel to their clients by showing that a Desogestrel CEP has been issued for it. The manufacturer submits a Desogestrel CEP (COS) as part of the market authorization procedure, and it takes on the role of a Desogestrel CEP holder for the record. Additionally, the data presented in the Desogestrel CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Desogestrel DMF.
A Desogestrel CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Desogestrel CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Desogestrel suppliers with CEP (COS) on PharmaCompass.
A Desogestrel written confirmation (Desogestrel WC) is an official document issued by a regulatory agency to a Desogestrel manufacturer, verifying that the manufacturing facility of a Desogestrel active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Desogestrel APIs or Desogestrel finished pharmaceutical products to another nation, regulatory agencies frequently require a Desogestrel WC (written confirmation) as part of the regulatory process.
click here to find a list of Desogestrel suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Desogestrel as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Desogestrel API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Desogestrel as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Desogestrel and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Desogestrel NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Desogestrel suppliers with NDC on PharmaCompass.
Desogestrel Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Desogestrel GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Desogestrel GMP manufacturer or Desogestrel GMP API supplier for your needs.
A Desogestrel CoA (Certificate of Analysis) is a formal document that attests to Desogestrel's compliance with Desogestrel specifications and serves as a tool for batch-level quality control.
Desogestrel CoA mostly includes findings from lab analyses of a specific batch. For each Desogestrel CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Desogestrel may be tested according to a variety of international standards, such as European Pharmacopoeia (Desogestrel EP), Desogestrel JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Desogestrel USP).
