
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
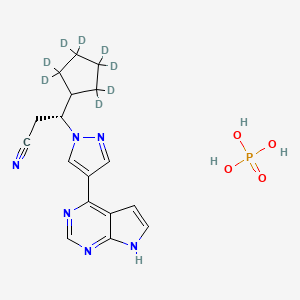

1. Ctp-543 Phosphate
2. C-21543 Phosphate
3. D8-ruxolitinib Phosphate
4. D8- Ruxolitinib Phosphate
5. 8vj43s4lcm
6. Deuruxolitinib Phosphate [usan]
7. 2147706-60-1
8. Deuruxolitinib Phosphate (usan)
9. (3r)-3-(2,2,3,3,4,4,5,5-d8)cyclopentyl-3-(4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-1h-pyrazol-1-yl)propanenitrile Phosphate (1:1)
10. Leqselvi (tn)
11. Unii-8vj43s4lcm
12. Chembl4594382
13. D11867
14. 1h-pyrazole-1-propanenitrile, .beta.-(cyclopentyl-2,2,3,3,4,4,5,5-d8)-4-(7h-pyrrolo(2,3-d)pyrimidin-4-yl)-, (.beta.r)-, Phosphate
15. 1h-pyrazole-1-propanenitrile, Beta-(cyclopentyl-2,2,3,3,4,4,5,5-d8)-4-(7hpyrrolo(2,3-d)pyrimidin-4-yl)-, (betar)-, Phosphate (1:1)
| Molecular Weight | 412.4 g/mol |
|---|---|
| Molecular Formula | C17H21N6O4P |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Exact Mass | g/mol |
| Monoisotopic Mass | g/mol |
| Topological Polar Surface Area | 161 |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 503 |
| Isotope Atom Count | 8 |
| Defined Atom Stereocenter Count | 1 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Patents & EXCLUSIVITIES
ABOUT THIS PAGE
