
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
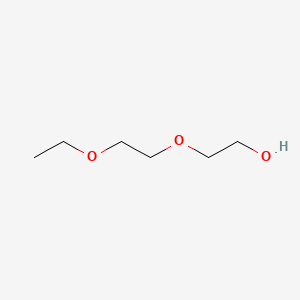

1. Carbitol
2. Ethyl Carbitol
3. Ethylcarbitol
4. Transcutol
5. Transcutol Hp
6. Transcutol P
1. 2-(2-ethoxyethoxy)ethanol
2. 111-90-0
3. Carbitol
4. Transcutol
5. 2(2-ethoxyethoxy)ethanol
6. Ethoxy Diglycol
7. Ethyl Carbitol
8. Ethoxydiglycol
9. Dioxitol
10. Ethanol, 2-(2-ethoxyethoxy)-
11. Ethyl Digol
12. Carbitol Solvent
13. Transcutol P
14. Solvolsol
15. Losungsmittel Apv
16. Dowanol De
17. Carbitol Cellosolve
18. Poly-solv De
19. Diglycol Monoethyl Ether
20. Diethylene Glycol Ethyl Ether
21. Degmee
22. Ektasolve De
23. Ethyl Diethylene Glycol
24. 3,6-dioxa-1-octanol
25. Dowanol 17
26. Karbitol
27. Diethyleneglycol Monoethyl Ether
28. Ethylene Diglycol Monoethyl Ether
29. 2-(2-ethoxyethoxy) Ethanol
30. Monoethyl Ether Of Diethylene Glycol
31. 3,6-dioxa-1-oktanol
32. Ethanol, 2,2'-oxybis-, Monoethyl Ether
33. 2-(ethoxyethoxy)ethanol
34. A1a1i8x02b
35. Chebi:40572
36. Nsc-408451
37. Diethylene Glycol Monoethyl Ether [nf]
38. Dsstox_cid_1941
39. Aqualine™ Complete 1
40. Aqualine™ Complete 2
41. Aqualine™ Complete 5
42. Dsstox_rid_76417
43. Karbitol [czech]
44. Dsstox_gsid_21941
45. 149818-01-9
46. 2-(2-ethoxyethoxy)-ethanol
47. Diethylene Glycol Monoethyl Ether (nf)
48. Acetamide, N-5-(1,2-dihydroxyethyl)-4-hydroxy-3-pyrrolidinyl-, Monohydrochloride, 3s-3.alpha.,4.beta
49. Ae3
50. Cas-111-90-0
51. Hsdb 51
52. Aethyldiaethylenglycol
53. Aethyldiaethylenglycol [german]
54. 3,6-dioxa-1-oktanol [czech]
55. Einecs 203-919-7
56. Nsc 408451
57. Pm 1799
58. Brn 1736441
59. Unii-a1a1i8x02b
60. Diethoxol
61. Ai3-01740
62. O-ethyldigol
63. Eastman De
64. Ethyl Di-icinol
65. Glycol Ether De
66. Mfcd00002872
67. (ethoxyethoxy)ethanol
68. 3,6-dioxaoctan-1-ol
69. 2-(2ethoxyethoxy)ethanol
70. Ec 203-919-7
71. Diethyleneglycolmonoethylether
72. 1-hydroxy-3,6-dioxaoctane
73. Schembl16399
74. 2-(2-ethoxyethoxy);ethanol
75. Aqualine™ Complete 5k
76. Ethoxydiglycol [inci]
77. Diethyleneglycol Monoethylether
78. Wln: Q2o2o2
79. 2-(2-ethoxy-ethoxy)-ethanol
80. 2-(.beta.-ethoxyethoxy)ethanol
81. 3,6-dioxaoctane-1-ol
82. Chembl1230841
83. Diethylene Glycol-monoethyl Ether
84. Dtxsid2021941
85. Zinc1600621
86. Tox21_200413
87. Tox21_300080
88. Ethanol,2'-oxybis-, Monoethyl Ether
89. Nsc408451
90. Stl453580
91. Akos009031390
92. Ehanol, 2,2'-oxybis-, Monoethyl Ether
93. Ncgc00247898-01
94. Ncgc00247898-02
95. Ncgc00254003-01
96. Ncgc00257967-01
97. Di(ethylene Glycol) Ethyl Ether, >=99%
98. Diethylene Glycol Monoethyl Ether, >=99%
99. Cs-0015134
100. E0048
101. Ft-0624897
102. Ft-0693130
103. Diethylene Glycol Monoethyl Ether [ii]
104. Diethylene Glycol Monoethyl Ether [mi]
105. Glycol Ether De (low Gravity) Reagent Grade
106. D08904
107. D72502
108. Diethylene Glycol Monoethyl Ether [hsdb]
109. A802441
110. Diethylene Glycol Monoethyl Ether [usp-rs]
111. Diethylene Glycol Monoethyl Ether [who-dd]
112. Q416399
113. J-505606
114. Diethylene Glycol Monoethyl Ether [ep Monograph]
115. Diethylene Glycol Monoethyl Ether, Reagentplus(r), 99%
116. Diethylene Glycol Monoethyl Ether, Saj First Grade, >=98.0%
117. Diethylene Glycol Monoethyl Ether, Vetec(tm) Reagent Grade, 99%
118. Diethylene Glycol Monoethyl Ether, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 134.17 g/mol |
|---|---|
| Molecular Formula | C6H14O3 |
| XLogP3 | -0.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 6 |
| Exact Mass | 134.094294304 g/mol |
| Monoisotopic Mass | 134.094294304 g/mol |
| Topological Polar Surface Area | 38.7 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 47.6 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
It has been estimated that the single oral dose /of diethylene glycol/ lethal for humans is approximately 1 ml/kg. /Diethylene glycols/
Amdur, M.O., J. Doull, C.D. Klaasen (eds). Casarett and Doull's Toxicology. 4th ed. New York, NY: Pergamon Press, 1991., p. 704
To assist evaluation of the hazards of skin contact with selected undiluted glycol ethers, their absorption across isolated human abdominal epidermis was measured in vitro. Epidermal membranes were set up in glass diffusion cells and, following an initial determination of permeability to tritiated water, excess undiluted glycol ether was applied to the outer surface for 8 hr. The appearance of glycol ether in an aqueous receptor phase bathing the underside of the epidermis was quantified by a gas chromatographic technique. A final determination of tritiated water permeability was compared with initial values to establish any irreversible alterations in epidermal barrier function induced by contact with the glycol ethers. 2-methoxyethanol (EM) was most readily absorbed (mean steady rate 2.82 mg/sq cm/hr), and a relatively high absorption rate (1.17 mg/sq cm/hr) was also apparent for 1-methoxypropan-2-ol (PM). There was a trend of reducing absorption rate with increasing molecular weight or reducing volatility for monoethylene glycol ethers (EM, 2.82 mg/sq cm/hr; 2-ethoxyethanol, EE, 0.796 mg/sq cm/hr; 2-butoxyethanol, EB, 0.198 mg/sq cm/hr) and also within the diethylene glycol series: 2-(2-methoxyethoxy) ethanol (DM, 0.206 mg/sq cm/hr); 2-(2-ethoxyethoxy) ethanol (DE, 0.125 mg/sq cm/hr) and 2-(2-butoxyethoxy) ethanol (DB, 0.035 mg/sq cm/hr). The rate of absorption of 2-ethoxyethyl acetate (EEAc) was similar to that of the parent alcohol, EE. Absorption rates of diethylene glycol ethers were slower than their corresponding monoethylene glycol equivalents. Combination of intrinsic toxicity and ability to pass across skin contribute to assessment of hazards of contact with undiluted glycol ethers.
PMID:6499804 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1568269 Dugard PH et al; Environ Health Perspect 57: 193-7 (1984)
When a single /oral/ dose of diethylene glycol monoethyl ether (11.2 mmol) was given to an adult human volunteer (sex and age not specified) about 68% of the dose was excreted in the urine as (2-ethoxyethoxy)acetic acid within 12 hr.
FAO/WHO Joint Expert Committee on Food Additives; WHO Food Additives Series 30 Diethylene Glycol Monoethyl Ether. Available from, as of February 20, 2007: https://www.inchem.org/pages/jecfa.html
The quantitative urinary excretion of diethylene glycol monoethyl ether was investigated in the rabbit after oral, intravenous, subcutaneous and percutaneous administration. After oral dosing of two animals at a level of 5 mL/kg bw, both animals died during the first day and total excretion was only 0.8% and 0.33%. After intravenous administration to 15 rabbits at dose levels of 0.5-3.4 mL/kg most of the dose was excreted during the first day (for 13/15 rabbits), the percentage excreted tending to increase with dose. After a single parenteral dose of 1.0-3.0 mL/kg, urinary excretion was monitored for up to 4 consecutive days. Excretion was high in the first 24 hr and the total percentage of the dose excreted in urine increased with dose. After repeated daily parenteral doses of 0.16, 0.32 or 0.63 mL/kg bw, total urinary excretion increased with dose and equalled 4.7, 5.0 and 11.6%, respectively.
FAO/WHO Joint Expert Committee on Food Additives; WHO Food Additives Series 30 Diethylene Glycol Monoethyl Ether. Available from, as of February 20, 2007: https://www.inchem.org/pages/jecfa.html
The oral administration fo 1503 mg Diethylene glycol monoethyl ether to a normal man resulted in the excretion of 1140 mg of 2-ethoxyethoxyacetic acid, 69 % of the total dose, within 12 hr.
Bingham, E.; Cohrssen, B.; Powell, C.H.; Patty's Toxicology Volumes 1-9 5th ed. John Wiley & Sons. New York, N.Y. (2001)., p. V7 197
...Carbitol is largely destroyed in body or conjugated with glucuronic acid and excreted as the glucuronate. ...This metabolic peculiarity may explain its lesser toxicity when compared with that of diethyl, methyl and butyl cellosolve.
Browning, E. Toxicity and Metabolism of Industrial Solvents. New York: American Elsevier, 1965., p. 630

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Market Place

ABOUT THIS PAGE
72
PharmaCompass offers a list of Diethylene Glycol Monoethyl Ether API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Diethylene Glycol Monoethyl Ether manufacturer or Diethylene Glycol Monoethyl Ether supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Diethylene Glycol Monoethyl Ether manufacturer or Diethylene Glycol Monoethyl Ether supplier.
PharmaCompass also assists you with knowing the Diethylene Glycol Monoethyl Ether API Price utilized in the formulation of products. Diethylene Glycol Monoethyl Ether API Price is not always fixed or binding as the Diethylene Glycol Monoethyl Ether Price is obtained through a variety of data sources. The Diethylene Glycol Monoethyl Ether Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Diethylene Glycol Monoethyl Ether manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Diethylene Glycol Monoethyl Ether, including repackagers and relabelers. The FDA regulates Diethylene Glycol Monoethyl Ether manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Diethylene Glycol Monoethyl Ether API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Diethylene Glycol Monoethyl Ether supplier is an individual or a company that provides Diethylene Glycol Monoethyl Ether active pharmaceutical ingredient (API) or Diethylene Glycol Monoethyl Ether finished formulations upon request. The Diethylene Glycol Monoethyl Ether suppliers may include Diethylene Glycol Monoethyl Ether API manufacturers, exporters, distributors and traders.
click here to find a list of Diethylene Glycol Monoethyl Ether suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Diethylene Glycol Monoethyl Ether DMF (Drug Master File) is a document detailing the whole manufacturing process of Diethylene Glycol Monoethyl Ether active pharmaceutical ingredient (API) in detail. Different forms of Diethylene Glycol Monoethyl Ether DMFs exist exist since differing nations have different regulations, such as Diethylene Glycol Monoethyl Ether USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Diethylene Glycol Monoethyl Ether DMF submitted to regulatory agencies in the US is known as a USDMF. Diethylene Glycol Monoethyl Ether USDMF includes data on Diethylene Glycol Monoethyl Ether's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Diethylene Glycol Monoethyl Ether USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Diethylene Glycol Monoethyl Ether suppliers with USDMF on PharmaCompass.
Diethylene Glycol Monoethyl Ether Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Diethylene Glycol Monoethyl Ether GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Diethylene Glycol Monoethyl Ether GMP manufacturer or Diethylene Glycol Monoethyl Ether GMP API supplier for your needs.
A Diethylene Glycol Monoethyl Ether CoA (Certificate of Analysis) is a formal document that attests to Diethylene Glycol Monoethyl Ether's compliance with Diethylene Glycol Monoethyl Ether specifications and serves as a tool for batch-level quality control.
Diethylene Glycol Monoethyl Ether CoA mostly includes findings from lab analyses of a specific batch. For each Diethylene Glycol Monoethyl Ether CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Diethylene Glycol Monoethyl Ether may be tested according to a variety of international standards, such as European Pharmacopoeia (Diethylene Glycol Monoethyl Ether EP), Diethylene Glycol Monoethyl Ether JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Diethylene Glycol Monoethyl Ether USP).

