
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
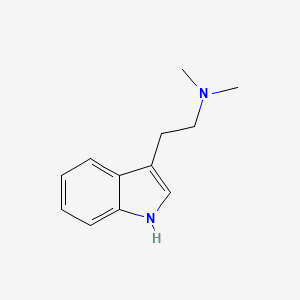

1. Dimethyltryptamine
2. N,n Dimethyltryptamine
1. Dimethyltryptamine
2. 61-50-7
3. 2-(3-indolyl)ethyldimethylamine
4. 3-(2-dimethylaminoethyl)indole
5. N,n-dimethyl-1h-indole-3-ethylamine
6. 2-(1h-indol-3-yl)-n,n-dimethylethanamine
7. 1h-indole-3-ethanamine, N,n-dimethyl-
8. Dmt
9. N,n-dimethyl-1h-indole-3-ethanamine
10. 3-[2-(dimethylamino)ethyl]indole
11. Dea No. 7435
12. Wub601bhaa
13. Chembl12420
14. Chebi:28969
15. (2-indol-3-ylethyl)dimethylamine
16. Nsc-63795
17. Indole, 3-(2-(dimethylamino)ethyl)-
18. Indole, 3-[2-(dimethylamino)ethyl]-
19. [2-(1h-indol-3-yl)ethyl]dimethylamine
20. Dmt (psychogenic)
21. [2-(1h-indol-3-yl)-ethyl]-dimethyl-amine
22. (psychogenic)
23. 3-(2-(dimethylamino)ethyl)indole
24. Einecs 200-508-4
25. Unii-wub601bhaa
26. Nsc 63795
27. Brn 0138259
28. Hsdb 8017
29. Indolalkylamine Der
30. 2-(1h-indol-3-yl)-n,n-dimethyl-ethanamine
31. Nn-dimethyltryptamine
32. 1h-indole-3-ethanamine,n,n-dimethyl-
33. Dimethyltryptamine(dmt)
34. (n,n)-dimethyltryptamine
35. N-dimethyltryptamine
36. Gtpl141
37. 3-(dimethylaminoethyl)-indole
38. Schembl335710
39. Dtxsid60110053
40. Dimethyltryptamine [mart.]
41. N,n-dimethyltryptamine, Free Base
42. Zinc897457
43. Dimethyltryptamine [who-dd]
44. N,n-dimethyltryptamine [mi]
45. Nsc63795
46. Wln: T56 Bmj D2n1&1
47. 3-(2-(dimethylamino)ethyl)-indole
48. Bdbm50026868
49. Mfcd00055989
50. Stk370594
51. 3-[2- (dimethylamino)ethyl]-indole
52. Akos005446117
53. Db01488
54. Mb00483
55. 3-(2-dimethylaminoethyl) Indole
56. 2-(1h-indol-3-yl)-ethyl-dimethyl-amine
57. As-47089
58. N,n-dimethyltryptamine, >=97% (hplc)
59. 2-(1h-indol-3-yl)-n,n-dimethylethylamine
60. 1h-indole-3-ethanamine,n,n-dimethyl
61. N,n-dimethyl-1h-indole-3-ethanamine (9ci)
62. 2-(1h-indol-3-yl)-n,n-dimethylethanamine #
63. C08302
64. D-5500
65. 061d507
66. L001288
67. Q407217
68. 2-(1h-indol-3-yl)-n,n-dimethylethanamine (acd/name 4.0)
69. N-(2-(1h-indol-3-yl)ethyl)-n,n-dimethylamine (acd/name 4.0)
70. N,n-dimethyltryptamine (dmt) Solution, 1.0 Mg/ml In Methanol, Certified Reference Material
| Molecular Weight | 188.27 g/mol |
|---|---|
| Molecular Formula | C12H16N2 |
| XLogP3 | 2.5 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 3 |
| Exact Mass | 188.131348519 g/mol |
| Monoisotopic Mass | 188.131348519 g/mol |
| Topological Polar Surface Area | 19 Ų |
| Heavy Atom Count | 14 |
| Formal Charge | 0 |
| Complexity | 179 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Some people use this compound as a psychedelic inducing agent.
Hallucinogens
Drugs capable of inducing illusions, hallucinations, delusions, paranoid ideations, and other alterations of mood and thinking. Despite the name, the feature that distinguishes these agents from other classes of drugs is their capacity to induce states of altered perception, thought, and feeling that are not experienced otherwise. (See all compounds classified as Hallucinogens.)
Serotonin Antagonists
Drugs that bind to but do not activate serotonin receptors, thereby blocking the actions of serotonin or SEROTONIN RECEPTOR AGONISTS. (See all compounds classified as Serotonin Antagonists.)
Serotonin Receptor Agonists
Endogenous compounds and drugs that bind to and activate SEROTONIN RECEPTORS. Many serotonin receptor agonists are used as ANTIDEPRESSANTS; ANXIOLYTICS; and in the treatment of MIGRAINE DISORDERS. (See all compounds classified as Serotonin Receptor Agonists.)
(14)C-DMT accumulates in rat brain cortical slices incubated at 37 C. ... Most of the accumulated (14)C-DMT was associated with the cytoplasmic fraction. Of the portion associated with the crude mitochondrial fraction, 54.4% was associated with nerve-ending fraction.
PMID:41604 Sangiah et al; Biol Psychiatry 14 (6): 925-36 (1979)
The hallucinogenic substance N',N'-dimethyltryptamine and its precursor N-methyltryptamine were found in 24-hr specimens of urine from 19 normal human subjects; the mean excretion rates were 386 ng 24 hr(-1) and 856 ng 24 hr(-1) respectively. The urinary excretion of both compounds was unrelated to age, sex, urinary volume, or creatinine, nor was any consistent diurnal pattern observed. Rates for the mono and dimethylated compounds were not correlated. Diet and the intestinal flora were excluded as a source of urinary dimethyltryptamine. Administration to 4 subjects of sufficient ammonium chloride to /decrease/ the /pH/ of the urine caused a transient increase in dimethyltryptamine excretion but no consistent increase in the rate for N-methyltryptamine. Acidification of the urine did not appear to be the determining factor in this result since in one subject the same drop in urinary pH was achieved by feeding methionine without any increase in dimethyltryptamine excretion.
PMID:22091 Oon MC et al; Psychopharmacology (Berl) 54 (2): 171-5 (1977)
... Eighteen volunteers with prior experience in the use of psychedelics received single oral doses of encapsulated freeze-dried ayahuasca (0.6 and 0.85 mg of DMT/kg of body weight) and placebo. Ayahuasca produced significant subjective effects, peaking between 1.5 and 2 hr, involving perceptual modifications and increases in ratings of positive mood and activation. ...Cmax values for DMT after the low and high ayahuasca doses were 12.14 ng/mL and 17.44 ng/mL, respectively. Tmax (median) was observed at 1.5 hr after both doses. The Tmax for DMT coincided with the peak of subjective effects. ...
PMID:12660312 Riba J et al; J Pharmacol Exp Ther 306 (1): 73-83 (2003)
The endogenous hallucinogen, N,N-dimethyltryptamine (DMT), was labeled with (11)C and its regional distribution in rat brain studied. (11)C-DMT showed higher accumulation in the cerebral cortex, caudate putamen, and amygdaloid nuclei. Studies of the subcellular distribution of (11)C-DMT revealed the specific localization in the fractions enriched with serotonin receptors only when a very low dose was injected into rats. ...
PMID:3489620 Yanai K et al; Eur J Nucl Med 12 (3): 141-6 (1986)
For more Absorption, Distribution and Excretion (Complete) data for N,N-Dimethyltryptamine (8 total), please visit the HSDB record page.
Following intraperitoneal administration, 5-methoxy-N,N-dimethyltryptamine and N,N-dimethyltryptamine are subject to both a very rapid uptake into, and clearance from, all tissues examined. The current studies in vivo confirm previous in vitro observations that the routes involved in the metabolism of these compounds include oxidative deamination, N-demethylation, O-demethylation, and N-oxidation. The analysis of metabolic profiles in various tissues led to the identification of the N-oxides as major metabolites...
PMID:3472526 Sitaram BR et al; Biochem Pharmacol 36 (9): 1509-12 (1987)
... /From/ ... urine samples collected from three individuals that were administered ayahuasca ... /authors/ show that the major metabolite of the hallucinogenic component of ayahuasca, N,N-dimethyltryptamine (DMT), is the corresponding N-oxide... Further, very little DMT was detected in urine, despite the inhibition of monoamine oxidase afforded by the presence of the harmala alkaloids in ayahuasca. ...
PMID:21058415 McIlhenny EH et al; Biomed Chromatogr 25 (9): 970-84 (2011)
Behavioral aspects and metabolic fate of N,N-dimethyltryptamine (DMT) were studied in mice pretreated with beta-diethylaminoethyl-diphenylpropylacetate (SKF 525-A), iproniazid or chlorpromazine (CPZ.). ... Dose-dependent increases with rapid uptake and disappearance in the brain, plasma and hepatic levels of DMT were measured with doses of 10 and 25 mg/kg DMT. ... It is concluded that DMT is metabolized chiefly by monoamine oxidase rather than by drug-metabolizing hepatic microsomal enzymes and that DMT-induced behavioral effects are due to the parent compound rather than its metabolite.
PMID:276891 Shah NS, Hedden MP; Pharmacol Biochem Behav 8 (4): 351-6 (1978)
Studies were conducted using tritiated DMT and DMT-N-oxide (DMT-NO), and metabolites were identified and quantified using thin-layer chromatography and liquid scintillation counting techniques. Metabolite confirmation was obtained by incubation of alpha,alpha,beta,beta-tetradeutero-DMT (DDMT) with whole brain homogenate followed by combined gas chromatographic/mass spectrometric analyses. The metabolites of DMT were identified as indoleacetic acid (IAA), DMT-NO, N-methyltryptamine (NMT), 2-methyl-1,2,3,4-tetrahydro-beta-carboline (2-MTHBC), tryptamine (TA) and 1,2,3,4- tetrahydro-beta-carboline (THBC). DMT-NO was metabolized to give DMT, NMT, IAA and 2-MTHBC. Formation of these metabolites from DMT-NO was stimulated by anaerobic incubation. ...
PMID:6770869 Barker et al; Biochem Pharmacol 29 (7): 1049-57 (1980)
DMT acts as a non-selective agonist at most or all of the serotonin receptors.
N,N-dimethyltryptamine (DMT) is a hallucinogen found endogenously in human brain that is commonly recognized to target the 5-hydroxytryptamine 2A receptor or the trace amine-associated receptor to exert its psychedelic effect. DMT has been recently shown to bind sigma-1 receptors, which are ligand-regulated molecular chaperones whose function includes inhibiting various voltage-sensitive ion channels. Thus, it is possible that the psychedelic action of DMT might be mediated in part through sigma-1 receptors. ...
PMID:19278957 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3155724 Su TP et al; Sci Signal 2 (61): pe12 (2009)
The psychotomimetic agent dimethyltryptamine (DMT) has been identified as an endogenous compound in the central nervous system of rodents using a sensitive electron capture gas chromatographic technique. DMT along with its proposed precursor, tryptamine, were identified and quantitated as the heptafluorobutyryl derivatives. A specific high affinity binding site on synaptosomal membranes has been proposed for DMT. This proposal is based on equilibrium dialysis experiments which indicate that DMT at a concentration of 1X10-5M will displace d-LSD on isolated membranes but will not displace bound serotonin at the same concentration. When DMT interacts with the synaptosomal membranes at a concentration of 5X10-10M, the membrane-bound enzyme adenylate cyclase is stimulated such that adenosine3', 5'-monophosphate (cAMP) is produced at a rate of 100 pM/min/mg of protein (2.3 times the endogenous rate). It has also been shown that its presumed precursor, tryptamine, inhibits this process. ... From data obtained in this study it has been postulated that DMT may have in vivo activity similar to those proposed for neurotransmitters or other neuroregulatory agents. ...
PMID:20877 Christian ST et al; Biochem Med 18(2) : 164-83 (1977)
The interactions of the indolealkylamine N,N-dimethyltryptamine (DMT) with 5-hydroxytryptamine1A (5-HT1A) and 5-HT2 receptors in rat brain were analyzed using radioligand binding techniques and biochemical functional assays. The affinity of DMT for 5-HT1A sites labeled by (3)H-8-hydroxy-2-(di-n-propylamino)tetralin (3)H-8-OH-DPAT) was decreased in the presence of 1X10-4 M GTP, suggesting agonist activity of DMT at this receptor. Adenylate cyclase studies in rat hippocampi showed that DMT inhibited forskolin-stimulated cyclase activity, a 5-HT1A agonist effect. DMT displayed full agonist activity with an EC50 of 4X10-6 M in the cyclase assay. In contrast to the agonist actions of DMT at 5-HT1A receptors, DMT appeared to have antagonistic properties at 5-HT2 receptors. The ability of DMT to compete for (3)H-ketanserin-labeled 5-HT2 receptors was not affected by the presence of 1X10-4 M GTP, suggesting antagonist activity of DMT at 5-HT2 receptors. In addition, DMT antagonized 5-HT2-receptor-mediated phosphatidylinositol (PI) turnover in rat cortex at concentrations above 1X10-7 M, with 70% of the 5-HT-induced PI response inhibited at 1X10-4 M DMT. Micromolar concentrations of DMT produced a slight PI stimulation that was not blocked by the 5-HT2 antagonist ketanserin. These studies suggest that DMT has opposing actions on 5-HT receptor subtypes, displaying agonist activity at 5-HT1A receptors and antagonist activity at 5-HT2 receptors.
PMID:1828347 Deliganis AV et al; Biochem Pharmacol 41 (11): 1739-44 (1991)
... The sigma-1 receptor pharmacophore includes an alkylamine core, also found in the endogenous compound N,N-dimethyltryptamine (DMT). DMT acts as a hallucinogen, but its receptor target has been unclear. DMT bound to sigma-1 receptors and inhibited voltage-gated sodium ion (Na+) channels in both native cardiac myocytes and heterologous cells that express sigma-1 receptors. ... DMT induced hypermobility in wild-type mice but not in sigma-1 receptor knockout mice. These biochemical, physiological, and behavioral experiments indicate that DMT is an endogenous agonist for the sigma-1 receptor.
PMID:19213917 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2947205 Fontanilla D et al; Science 323 (5916): 934-7 (2009)
For more Mechanism of Action (Complete) data for N,N-Dimethyltryptamine (7 total), please visit the HSDB record page.
Market Place

ABOUT THIS PAGE
45
PharmaCompass offers a list of Dimethyltryptamine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Dimethyltryptamine manufacturer or Dimethyltryptamine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dimethyltryptamine manufacturer or Dimethyltryptamine supplier.
PharmaCompass also assists you with knowing the Dimethyltryptamine API Price utilized in the formulation of products. Dimethyltryptamine API Price is not always fixed or binding as the Dimethyltryptamine Price is obtained through a variety of data sources. The Dimethyltryptamine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Dimethyltryptamine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dimethyltryptamine, including repackagers and relabelers. The FDA regulates Dimethyltryptamine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dimethyltryptamine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dimethyltryptamine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Dimethyltryptamine supplier is an individual or a company that provides Dimethyltryptamine active pharmaceutical ingredient (API) or Dimethyltryptamine finished formulations upon request. The Dimethyltryptamine suppliers may include Dimethyltryptamine API manufacturers, exporters, distributors and traders.
click here to find a list of Dimethyltryptamine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Dimethyltryptamine DMF (Drug Master File) is a document detailing the whole manufacturing process of Dimethyltryptamine active pharmaceutical ingredient (API) in detail. Different forms of Dimethyltryptamine DMFs exist exist since differing nations have different regulations, such as Dimethyltryptamine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Dimethyltryptamine DMF submitted to regulatory agencies in the US is known as a USDMF. Dimethyltryptamine USDMF includes data on Dimethyltryptamine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Dimethyltryptamine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Dimethyltryptamine suppliers with USDMF on PharmaCompass.
Dimethyltryptamine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dimethyltryptamine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Dimethyltryptamine GMP manufacturer or Dimethyltryptamine GMP API supplier for your needs.
A Dimethyltryptamine CoA (Certificate of Analysis) is a formal document that attests to Dimethyltryptamine's compliance with Dimethyltryptamine specifications and serves as a tool for batch-level quality control.
Dimethyltryptamine CoA mostly includes findings from lab analyses of a specific batch. For each Dimethyltryptamine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dimethyltryptamine may be tested according to a variety of international standards, such as European Pharmacopoeia (Dimethyltryptamine EP), Dimethyltryptamine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dimethyltryptamine USP).
