
Synopsis
Synopsis
0
EU WC
0
KDMF
0
VMF
0
FDA Orange Book

0
Australia

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
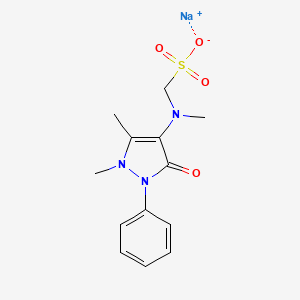

1. Algopyrin
2. Analgin
3. Biopyrin
4. Dipyronium
5. Metamizol
6. Metamizole
7. Metamizole Sodium
8. Methamizole
9. Methampyrone
10. Narone
11. Noramidopyrine Methanesulfonate
12. Noramidopyrine Methanesulfonate Sodium
13. Normelubrine
14. Novalgetol
15. Novalgin
16. Novamidazophen
17. Novaminsulfone
18. Optalgin
19. Pyralgin
20. Sulpyrin
21. Sulpyrine
1. Analgin
2. Metamizole Sodium
3. 68-89-3
4. Methampyrone
5. Novalgin
6. Methylmelubrin
7. Neomelurbrin
8. Algocalmin
9. Novalgetol
10. Optalgin
11. Pyralgin
12. Sulpyrin
13. Fevonil
14. Narone
15. Algopyrine
16. Farmolisina
17. Nevralgina
18. Novamidazophen
19. Novaminsulfone
20. Pharmalgine
21. Alginodia
22. Analgine
23. Bonpyrin
24. Dimethone
25. Feverall
26. Gifaril
27. Keypyrone
28. Metapyrin
29. Metilon
30. Noveltex
31. Paralgin
32. Pyretin
33. Pyrojec
34. Barone
35. Conmel
36. Sulpin
37. Neo-melubrine
38. Arpf
39. Di-podil
40. Aminopyrine Sodium Sulfonate
41. Metamizolum Natricum
42. Novaminsulfon
43. Novaminsulfonum
44. Diprofarn
45. Novaminsulfonium
46. Novaminophenazone
47. Metamizole Sodique
48. Novaminsulfone Sodium
49. Metamizolo
50. Noraminophenazone Sodium Mesylate
51. Meamizol Sodico
52. Sodium Novaminsulfonate
53. Noramidopyrine Methanesulfonate Sodium
54. Pyretin (pharmaceutical)
55. Sodium Noramidopyrine Methanesulfonate
56. Dipyrone [ban]
57. Sodium Methylaminoantipyrine Methanesulfonate
58. Noraminophenazone Methanesulfonate Sodium Salt
59. Dipyrone Anhydrous
60. Vetalgin
61. Sodium (antipyrinylmethylamino)methanesulfonate
62. Mexican Aspirin
63. Metamizol Sodium
64. Noramidopyrine Methanesulfonate Sodium Salt
65. Methylaminoantipyrine Sodium Methanesulfonate
66. Noraminopyrine Methanesulfonate Sodium
67. Metamizole (sodium)
68. Dipyrone [anhydrous]
69. Chebi:59033
70. Metamizole Sodium Salt
71. Analgin (sodium Salt)
72. Methylaminophenyldimethylpyrazolone Methanesulfonate Sodium
73. Metamizole Sodium [inn]
74. Methanesulfonic Acid, (antipyrinylmethylamino)-, Sodium Salt
75. 4-sodium Methanesulfonate Methylamine-antipyrine
76. Noramidopyrinium-methanesulphate Natrium
77. 5907-38-0
78. Sodium 1-phenyl-2,3-dimethyl-4-methylaminopyrazolon-n-methanesulfonate
79. Vsu62z74on
80. 68-89-3 (sodium)
81. (antipyrinylmethylamino)methanesulfonic Acid Sodium Salt
82. Antipyrine, 4-(methylamino)-, Monomethosulfate, Sodium Salt
83. Methanesulfonic Acid, ((2,3-dihydro-1,5-dimethyl-3-oxo-2-phenyl-1h-pyrazol-4-yl)methylamino)-, Sodium Salt
84. Sodium Phenyldimethylpyrazolon-methylamino-methane Sulfonate
85. Sulpyrinum
86. Analginum
87. Phenyl Dimethyl Pyrazolon Methyl Aminomethane Sodium Sulfonate
88. Metamizolnatrium
89. 1-phenyl-2,3-dimethyl-5-pyrazolone-4-methylaminomethanesulfonate Sodium
90. 4-methylamino-1,5-dimethyl-2-phenyl-3-pyrazolone Sodium Methanesulfonate
91. Dipyrone (ban)
92. Sodium 1-phenyl-2,3-dimethyl-5-pyrazolone-4-methylamino Methanesulfonate
93. 1-phenyl-2,3-dimethylpyrazolone-(5)-4-methylaminomethanesulfonic Acid Sodium
94. Sodium 4-methylamino-1,5-dimethyl-2-phenyl-3-pyrazolone 4-methanesulfonate
95. Fenildimetil-pirazolon-metilaminometansolfonato Sodico
96. Sodium [(1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)(methyl)amino]methanesulfonate
97. Noramidopyrine Methanesulphonate Sodium
98. Ncgc00017049-01
99. Metamizolo [italian]
100. Dipyron Monohydrate
101. Methanesulfonic Acid, (antipyrinylmethylamino)-, Monosodium Salt
102. Novamidazophenum
103. Dsstox_cid_543
104. Noraminophenazone
105. 1-phenyl-2,3-dimethyl-4-methylamino-5-pyrazolon-n-methanesulfonsaeuren Natrium
106. Sodium;[(1,5-dimethyl-3-oxo-2-phenylpyrazol-4-yl)-methylamino]methanesulfonate
107. Dsstox_rid_75651
108. Dsstox_gsid_20543
109. Metamizolo Sodico [dcit]
110. Metamizol Sodico [spanish]
111. Metamizolum Natricum [latin]
112. Prodolina
113. Natrium Novaminsulfonicum
114. Metamizol Sodico
115. Metamizole Sodique [inn-french]
116. Metamizole Sodium (anhydrous)
117. Metamizolum Natricum [inn-latin]
118. Metamizolo Sodico
119. Metamizolum Natricum Anhydricum
120. Methamizole Sodium
121. Cas-68-89-3
122. Metamizole Sodique [french]
123. Methanesulfonic Acid, [(2,3-dihydro-1,5-dimethyl-3-oxo-2-phenyl-1h-pyrazol-4-yl)methylamino]-, Sodium Salt
124. Sodium ((1,5-dimethyl-3-oxo-2-phenyl-2,3-dihydro-1h-pyrazol-4-yl)(methyl)amino)methanesulfonate
125. Ccris 4443
126. Dipyrone [usan:jan]
127. Einecs 200-694-7
128. Unii-vsu62z74on
129. Methylmelubrine
130. Dipyrone Sodium
131. Noramidopyrinium-methanesulphate Natrium [german]
132. Dipyrone,(s)
133. Vetalgin (tn)
134. Prestwick_987
135. Fenildimetil-pirazolon-metilaminometansolfonato Sodico [italian]
136. Epitope Id:124939
137. 1-phenyl-2,3-dimethyl-4-methylamino-5-pyrazolon-n-methanesulfonsaeuren Natrium [german]
138. Schembl24996
139. Dipyrone [usan:ban:jan]
140. Spectrum1503298
141. Chembl487894
142. Dtxsid8020543
143. Metamizol Sodico [inn-spanish]
144. Hms501p16
145. Hy-b1279a
146. Hms1570o07
147. Hms1922a06
148. Hms2093c17
149. Hms2097o07
150. Hms3714o07
151. Metamizole Sodium [who-dd]
152. Tox21_110759
153. Tox21_200497
154. Ccg-39331
155. Mfcd00020783
156. Akos015904648
157. Tox21_110759_1
158. Metamizole Sodium [ep Impurity]
159. Ncgc00017049-02
160. Ncgc00017049-03
161. Ncgc00095040-01
162. Ncgc00095040-02
163. Ncgc00178220-05
164. Ncgc00258051-01
165. As-81983
166. Sodium ((2,3-dihydro-1,5-dimethyl-3-oxo-2-phenyl-1h-pyrazol-4-yl)methylamino)methanesulphonate
167. Cas-5907-38-0
168. Cs-0013577
169. Ft-0602853
170. M3060
171. D08190
172. F20711
173. Q422761
174. Sodium 1-phenyl-2,3-dimethyl-4-methylaminopyrazolon-n-methanesulphonate
175. Methanesulfonic Acid, 1-[(2,3-dihydro-1,5-dimethyl-3-oxo-2-phenyl-1h-pyrazol-4-yl)methylamino]-, Sodium Salt (1:1)
| Molecular Weight | 333.34 g/mol |
|---|---|
| Molecular Formula | C13H16N3NaO4S |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Exact Mass | 333.07592146 g/mol |
| Monoisotopic Mass | 333.07592146 g/mol |
| Topological Polar Surface Area | 92.4 Ų |
| Heavy Atom Count | 22 |
| Formal Charge | 0 |
| Complexity | 552 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Antipyretics
Drugs that are used to reduce body temperature in fever. (See all compounds classified as Antipyretics.)
Anti-Inflammatory Agents, Non-Steroidal
Anti-inflammatory agents that are non-steroidal in nature. In addition to anti-inflammatory actions, they have analgesic, antipyretic, and platelet-inhibitory actions. They act by blocking the synthesis of prostaglandins by inhibiting cyclooxygenase, which converts arachidonic acid to cyclic endoperoxides, precursors of prostaglandins. Inhibition of prostaglandin synthesis accounts for their analgesic, antipyretic, and platelet-inhibitory actions; other mechanisms may contribute to their anti-inflammatory effects. (See all compounds classified as Anti-Inflammatory Agents, Non-Steroidal.)
N02BB02
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
N - Nervous system
N02 - Analgesics
N02B - Other analgesics and antipyretics
N02BB - Pyrazolones
N02BB02 - Metamizole sodium

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
56
PharmaCompass offers a list of Metamizole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Metamizole manufacturer or Metamizole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Metamizole manufacturer or Metamizole supplier.
PharmaCompass also assists you with knowing the Metamizole API Price utilized in the formulation of products. Metamizole API Price is not always fixed or binding as the Metamizole Price is obtained through a variety of data sources. The Metamizole Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Dipyrone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dipyrone, including repackagers and relabelers. The FDA regulates Dipyrone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dipyrone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dipyrone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Dipyrone supplier is an individual or a company that provides Dipyrone active pharmaceutical ingredient (API) or Dipyrone finished formulations upon request. The Dipyrone suppliers may include Dipyrone API manufacturers, exporters, distributors and traders.
click here to find a list of Dipyrone suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Dipyrone DMF (Drug Master File) is a document detailing the whole manufacturing process of Dipyrone active pharmaceutical ingredient (API) in detail. Different forms of Dipyrone DMFs exist exist since differing nations have different regulations, such as Dipyrone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Dipyrone DMF submitted to regulatory agencies in the US is known as a USDMF. Dipyrone USDMF includes data on Dipyrone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Dipyrone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Dipyrone suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Dipyrone Drug Master File in Japan (Dipyrone JDMF) empowers Dipyrone API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Dipyrone JDMF during the approval evaluation for pharmaceutical products. At the time of Dipyrone JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Dipyrone suppliers with JDMF on PharmaCompass.
A Dipyrone CEP of the European Pharmacopoeia monograph is often referred to as a Dipyrone Certificate of Suitability (COS). The purpose of a Dipyrone CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Dipyrone EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Dipyrone to their clients by showing that a Dipyrone CEP has been issued for it. The manufacturer submits a Dipyrone CEP (COS) as part of the market authorization procedure, and it takes on the role of a Dipyrone CEP holder for the record. Additionally, the data presented in the Dipyrone CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Dipyrone DMF.
A Dipyrone CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Dipyrone CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Dipyrone suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Dipyrone as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Dipyrone API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Dipyrone as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Dipyrone and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Dipyrone NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Dipyrone suppliers with NDC on PharmaCompass.
Dipyrone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dipyrone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Dipyrone GMP manufacturer or Dipyrone GMP API supplier for your needs.
A Dipyrone CoA (Certificate of Analysis) is a formal document that attests to Dipyrone's compliance with Dipyrone specifications and serves as a tool for batch-level quality control.
Dipyrone CoA mostly includes findings from lab analyses of a specific batch. For each Dipyrone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dipyrone may be tested according to a variety of international standards, such as European Pharmacopoeia (Dipyrone EP), Dipyrone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dipyrone USP).
