
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
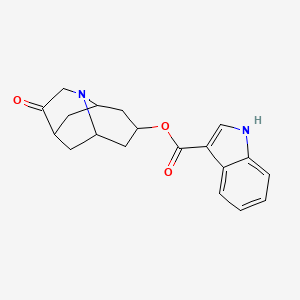

1. 1h-indole-3-carboxylic Acid, (6r,9as)-octahydro-3-oxo-2,6-methano-2h-quinolizin-8-yl Ester, Rel-, Methanesulfonate, Hydrate (1:1:1)
2. 1h-indole-3-carboxylic Acid, Octahydro-3-oxo-2,6-methano-2h-quinolizin-8-yl Ester, (2-alpha,6-alpha,8-alpha,9a-beta)-
3. 1h-indole-3-carboxylic Acid-trans-octahydro-3-oxo-2,6-methano-2h-quinolizin-8-yl Ester Methanesulfonate
4. Dolasetron
5. Dolasetron Mesilate Monohydrate
6. Dolasetron Mesylate
7. Dolasetron Mesylate Monohydrate
8. Indole-3-carboxylic Acid, Ester With (8r)-hexahydro-8-hydroxy-2,6-methano-2h-quinolizin-3(4h)-one
9. Mdl 73,147ef
10. Mdl 73147ef
11. Mdl-73147ef
12. Octahydro-3-oxo-2,6-methano-2h-quinolizin-8-yl 1h-indole-3-carboxylate
1. Dolasetron
2. 115956-12-2
3. Chebi:4682
4. Dolasetron (inn)
5. (2alpha,6alpha,8alpha,9ass)-octahydro-3-oxo-2,6-methano-2h-quinolizin-8-yl-1hindole-3-carboxylate Monomethanesulfonate, Monohydrate
6. 3-oxooctahydro-2h-2,6-methanoquinolizin-8-yl 1h-indole-3-carboxylate
7. Schembl42065
8. Chembl124754
9. Schembl14014817
10. Dtxsid20861231
11. (3r)-10-oxo-8-azatricyclo[5.3.1.03,8]undec-5-yl 1h-indole-3-carboxylate
12. 139014-62-3
13. Ft-0631080
14. C07866
15. D07867
16. 1h-indole-3-carboxylic Acid 10-oxo-8-aza-tricyclo[5.3.1.03,8]undec-5-yl Ester
| Molecular Weight | 324.4 g/mol |
|---|---|
| Molecular Formula | C19H20N2O3 |
| XLogP3 | 3.4 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 3 |
| Exact Mass | 324.14739250 g/mol |
| Monoisotopic Mass | 324.14739250 g/mol |
| Topological Polar Surface Area | 62.4 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 535 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 2 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antiemetic
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Dolasetron injection is indicated for the prevention of nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy, including high-dose cisplatin. Dolasetron tablets are indicated for the prevention of nausea and vomiting associated with moderate-emetogenic cancer chemotherapy, including initial and repeat courses. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1147
Dolasetron injection and tablets are indicated for the prevention of postoperative nausea and/or vomiting. Routine prophylaxis is not recommended when there is little risk of nausea and/or vomiting developing postoperatively, except in patients in whom nausea and/or vomiting must be avoided. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1147
Dolasetron injection is indicated for the treatment of postoperative nausea and/or vomiting. /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1147
/Administration is contraindicated in patients with/ known hypersensitivity to dolasetron mesylate.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2941
Acute, usually reversible ECG alterations and/or risk of altered cardiac conduction. Prolongation of PR, QTC, and JT intervals and widening of the QRS complex have been observed in patients receiving dolasetron therapy. These alterations, which are caused by prolongation of cardiac depolarization and repolarization, appear to be related to plasma concentrations of the active metabolite hydrodolasetron and generally are self-limiting as these concentrations decline. ECG interval prolongation rarely has resulted in heart block or cardiac arrhythmias. Sudden death has occurred in at least one patient 6 hours after receiving IV dolasetron (1.8 mg/kg), although the patient had other potential risk factors such as prior therapy with doxorubicin and concomitant cyclophosphamide.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2941
Dolasetron should be used with caution in patients who have or may develop prolongation of cardiac conduction intervals, particularly QTC, including those with congenital QT syndrome, those with uncorrected hypokalemia or hypomagnesemia, patients receiving diuretics that may induce electrolyte abnormalities, patients receiving antiarrhythmic agents or other drugs that alter cardiac conduction (e.g., prolong QT interval), and those receiving cumulative high-dose anthracycline therapy.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2941
Sensitivity reactions, including anaphylactic reaction, facial edema, and urticaria, have been reported rarely. Cross-sensitivity reactions have been reported in patients receiving other selective 5-HT3 receptor antagonists but have not been reported to date with dolasetron.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2941
For more Drug Warnings (Complete) data for DOLASETRON (7 total), please visit the HSDB record page.
Serotonin 5-HT3 Receptor Antagonists
Drugs that bind to but do not activate SEROTONIN 5-HT3 RECEPTORS, thereby blocking the actions of SEROTONIN or SEROTONIN 5-HT3 RECEPTOR AGONISTS. (See all compounds classified as Serotonin 5-HT3 Receptor Antagonists.)
Antiemetics
Drugs used to prevent NAUSEA or VOMITING. (See all compounds classified as Antiemetics.)
Orally-administered dolasetron is well absorbed, but the parent drug is rarely detected in plasma due to rapid and complete metabolism to hydrodolasetron.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
Orally-administered dolasetron intravenous solution and tablets are bioequivalent.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
The apparent absolute bioavailability of oral dolasetron is approximately 75%. Food does not affect the bioavailability of dolasetron taken by mouth.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
Time to peak plasma concentration /for hydrodolasetron/ following oral administration /was/ approximately 1 hour and following intravenous injection /was/ 0.6 hours.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
For more Absorption, Distribution and Excretion (Complete) data for DOLASETRON (15 total), please visit the HSDB record page.
Biotransformation /is/ hepatic and complete, mainly to the active metabolite hydrodolasetron (by means of the ubiquitous enzyme, carbonyl reductase). Further hydroxylation is mediated by cytochrome P450 CYP2D6 and further N-oxidation by both CYP3A and flavin monooxygenase.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
The metabolism of dolasetron mesylate was studied in six healthy male volunteers who were given a single 300 mg oral dose of [14C]dolasetron mesylate. An average of 59% of the total radioactivity was recovered in the urine and 25% in the feces. Metabolites were quantitated in urine samples taken up to 36 hr post-dose. Reduced dolasetron (RD) accounted for 17-54% of the dose in urine. Hydroxylated metabolites of RD made up no more than 9% of the dose in urine. Most of the remaining urinary radioactivity consisted of conjugated metabolites of RD and hydroxy RD. Hydrolysis of selected urine samples showed that the glucuronide of RD was the most abundant conjugate in urine. A small percentage of the dose (< 1%) in urine was identified as the N-oxide of RD. Analysis of urine samples by chiral HPLC indicated that the R(+):S(-) ratio of RD was approximately 9:1.
PMID:7493546 Reith MK, Sproles G et al; Drug Metab Dispos 23 (8): 806-12 (1995)
The initial step in the metabolism of dolasetron or MDL 73,147EF [(2 alpha, 6 alpha, 8 alpha, 9a beta)-octahydro-3-oxo-2,6-methano-2H- quinolizin-8-yl 1H-indol-3-carboxylate, monomethanesulfonate] is the reduction of the prochiral carbonyl group to give a chiral secondary alcohol "reduced dolasetron." An HPLC method, using a chiral column to separate reduced dolasetron enantiomers, has been developed and used to measure enantiomers in urine of rats, dogs, and humans after dolasetron administration. In all cases, the reduction was enantioselective for the (+)-(R)-enantiomer, although the dog showed lower stereoselectivity, especially after iv administration. An approximate enantiomeric ratio (+/-) of 90:10 was found in rat and human urine. The contribution of further metabolism to this enantiomeric ratio was considered small as preliminary studies showed that oxidation of the enantiomeric alcohols by human liver microsomes demonstrated only minor stereoselectivity. Further evidence for the role of stereoselective reduction in man was obtained from in vitro studies, where dolasetron was incubated with human whole blood. The enantiomeric composition of reduced dolasetron formed in human whole blood was the same as that found in human urine after administration of dolasetron. Enantioselectivity was not due to differences in the absorption, distribution, metabolism, or excretion of enantiomers, as iv or oral administration of rac-reduced dolasetron to rats and dogs lead to the recovery, in urine, of essentially the same enantiomeric composition as the dose administered. It is fortuitous that the (+)-(R)-enantiomer is predominantly formed by carbonyl reductase, as it is the more active compound.
PMID:7495640 Dow J, Berg C; Chirality 7 (5): 342-8 (1995)
Following oral administration hydrodolasetron has a elimination half-life of 8.1 hours (mean). Following intravenous injection dolasetron /has a elimination half-life of/ less than 10 minutes. Hydrodolasetron /has a elimination half-life of/ 7.3 hours after intravenous injection of dolasetron.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1148
Following intravenous administration to healthy male subjects of doses ranging from 0.6 to 5 mg/kg, dolasetron disappeared extremely rapidly from plasma; concentrations were generally measurable for only 2-4 hr. Less than 1 percent of the dose was excreted intact in urine. A major plasma metabolite, reduced dolasetron, peaked rapidly at approximately 0.625 hr (median). Its median terminal disposition half-life was 7.56 hr ...
PMID:1467456 Boxenbaum H, Gillespie T et al; Biopharm Drug Dispos 13 (9): 693-701 (1992)
Dolasetron, and its active metabolite hydrodolasetron, are highly specific and selective antagonist of serotonin subtype 3 (5-HT3) receptors. 5-HT3 receptors are present peripherally on vagal nerve terminals and centrally in the area postrema of the brain. Chemotherapeutic medications appear to precipitate release of serotonin from the enterochromaffin cells of the small intestine, which activates 5-HT3 receptors on vagal efferents to initiate the vomiting reflex. Dolasetron has not been shown to have activity at other known serotonin receptors, and has low affinity for dopamine receptors.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1147
Dolasetron causes dose-related acute, and usually reversible, electrocardiogram (ECG) changes including QRS widening and PR, QTc, and JT prolongation; QTc prolongation is caused primarily by QRS widening. Dolasetron seems to prolong both depolarization and,to a lesser extent, repolarization time, and its active metabolites may block sodium channels.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2007., p. 1147
The active metabolite of dolasetron (i.e., hydrodolasetron) may block sodium channels and prolong cardiac depolarization and, to a lesser extent, repolarization time.
McEvoy, G.K. (ed.). American Hospital Formulary Service. AHFS Drug Information. American Society of Health-System Pharmacists, Bethesda, MD. 2007., p. 2941
ABOUT THIS PAGE
87
PharmaCompass offers a list of Dolasteron API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Dolasteron manufacturer or Dolasteron supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Dolasteron manufacturer or Dolasteron supplier.
PharmaCompass also assists you with knowing the Dolasteron API Price utilized in the formulation of products. Dolasteron API Price is not always fixed or binding as the Dolasteron Price is obtained through a variety of data sources. The Dolasteron Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Dolasteron manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Dolasteron, including repackagers and relabelers. The FDA regulates Dolasteron manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Dolasteron API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Dolasteron manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Dolasteron supplier is an individual or a company that provides Dolasteron active pharmaceutical ingredient (API) or Dolasteron finished formulations upon request. The Dolasteron suppliers may include Dolasteron API manufacturers, exporters, distributors and traders.
click here to find a list of Dolasteron suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Dolasteron Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Dolasteron GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Dolasteron GMP manufacturer or Dolasteron GMP API supplier for your needs.
A Dolasteron CoA (Certificate of Analysis) is a formal document that attests to Dolasteron's compliance with Dolasteron specifications and serves as a tool for batch-level quality control.
Dolasteron CoA mostly includes findings from lab analyses of a specific batch. For each Dolasteron CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Dolasteron may be tested according to a variety of international standards, such as European Pharmacopoeia (Dolasteron EP), Dolasteron JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Dolasteron USP).
