
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
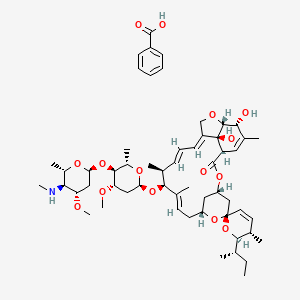

1. 4''-deoxy-4''-epi-n-methylaminoavermectin B1 Benzoate
2. Mk 244
3. Mk-244
1. 155569-91-8
2. Emamectin
3. 137512-74-4
4. 119791-41-2
5. Emamectin Benzoate, 90%
6. Schembl4291571
7. Chembl1883852
8. Mk-244mk-244
9. Akos025311598
10. As-72351
11. S4423
12. 569e918
13. (4/'/'r)-4/'/'-deoxy-4/'/'-(methylamino)avermectin B1 Benzoate
| Molecular Weight | 1008.2 g/mol |
|---|---|
| Molecular Formula | C56H81NO15 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 16 |
| Rotatable Bond Count | 10 |
| Exact Mass | 1007.56062088 g/mol |
| Monoisotopic Mass | 1007.56062088 g/mol |
| Topological Polar Surface Area | 199 Ų |
| Heavy Atom Count | 72 |
| Formal Charge | 0 |
| Complexity | 1850 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 20 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 3 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Therapeutic category (veterinary): antiparasitic
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 655
Antiparasitic Agents
Drugs used to treat or prevent parasitic infections. (See all compounds classified as Antiparasitic Agents.)
Insecticides
Pesticides designed to control insects that are harmful to man. The insects may be directly harmful, as those acting as disease vectors, or indirectly harmful, as destroyers of crops, food products, or textile fabrics. (See all compounds classified as Insecticides.)
There were 2 dosing groups, each consisting of 2 male beagles. Group 1 received 0.5 mg/kg of (3)H-MK-0243 benzoate (1 mL/kg in 5% ethanol ... 0.239 mCi/mg; 98.8% radiochemically pure) on day 1 and 0.5 mg/kg of (3)H-MK-0243 HCl (1 mL/kg in deionized water ... 0.229 mCi/mg; 98.7% radiochemically pure) on day 15. Dosing was reversed for Group 2. Body weights were determined before each dose. 2 mL of blood was withdrawn for drug level determinations following each dose at 0.5, 1, 2, 4, 6, 8, 24, 48, 96 and 168 hr. Urine and feces were collected for drug level analysis at 0 to 24 and 72 to 96 hr. There was no evidence of drug effects. The mean plasma half lives for the benzoate and HCl salts were 35.7 +/- 3.4 hr and 35.5 +/- 4.4 hr, respectively. The mean plasma approximate area under the curve (AUC) for the benzoate and HCl salts was 4479 +/- 1476 and 4574 +/- 1514 ng/g plasma/7days. The mean peak plasma MAB1a (the major component of MK-0243 at 90 to 95%) levels were ~100 ng equivalents/g plasma, occurring at ~6 hr for either salt. Combined fecal and urine recoveries during the 1st and 4th days were ~40% and 0.01% of the dose, respectively. It is concluded that the 2 salts are bioequivalent in male beagle dogs.
California Environmental Protection Agency/Department of Pesticide Regulation; Toxicology Data Review Summary for Emamectin Benzoate (119791-41-2) p.17 (October 2, 1999). Available from, as of March 21, 2014: https://www.cdpr.ca.gov/docs/risk/toxsums/toxsumlist.htm
The dermal absorption of the experimental avermectin insecticide emamectin benzoate was studied in the Rhesus monkey. Dermal absorption was calculated by comparing radioactivity levels in excreta following dermal application of the compound with those following administration of an equivalent intravenous dose. After iv administration of 300 ug (3)H-MAB1a (prepared as a 1:1 solution of propylene glycol:saline) to three monkeys, plasma levels decreased biphasically with a rapid decline in radioactivity during the first 15 min followed by a slower decline to background. By 7 days post-dose, approximately 90% and 5% of the administered radioactivity was recovered in the feces and urine, respectively. After a washout period, 300 micrograms [(3)H]MAB1a (dissolved in emulsifiable concentrate) was applied topically to the shaved forearm of the same monkeys. Following a 10-hr exposure period, approximately 90% of the radioactivity was recovered in a soap and water wash of the exposed forearms. Although plasma radioactivity levels generally remained below background levels, approximately 1.5% of the applied dose was recovered in the excreta. Dermal absorption of [()3H]emamectin benzoate was calculated as 1.6%. The low dermal penetration of emamectin benzoate indicates that minimal actual exposure of agricultural workers to this compound will occur.
PMID:9463543 Wrzesinski CL et al; Food Chem Toxicol 35 (10-11): 1085-9 (1997)
The aims of this study were to investigate the content of emamectin in blood, mucus and muscle following field administration of the recommended dose, and correlation with sea lice infection on the same fish (elimination study). The tissue distribution of tritiated emamectin benzoate after a single oral dose in Atlantic salmon was also investigated by means of whole-body autoradiography and scintillation counting (distribution study). In the elimination study, concentrations of emamectin benzoate reached maximum levels of 128, 105 and 68 ng/g (p.p.b.) for blood, mucus and muscle respectively, on day 7, the last day of administration. From day 7, the concentration in the blood declined until concentration was less than the limit of detection on day 77. The concentration was higher in mucus compared with plasma (P < 0.05) except on days 7 and 21. The concentration of emamectin benzoate decreased gradually from the end of treatment (day 7) to day 70 with half-lives of 9.2, 10.0 and 11.3 days in muscle, plasma and mucus respectively. The distribution study demonstrated a high quantity of radioactivity in mucous membranes (gastrointestinal tract, gills) throughout the observation period (56 days). Activity was high in the epiphysis, hypophysis and olfactory rosette throughout the study. The highest activity was observed in the bile, indicating this to be an important route for excretion. The distribution study confirmed the results from the elimination study with respect to concentrations in blood, skin mucous and muscle.
PMID:15720522 Sevatdal S et al; J Vet Pharmacol Ther. 28(1):101-7 (2005).
Atlantic salmon (approximately 1.3 kg) maintained in tanks of seawater at 5 +/- 1 degrees C were dosed with 3H-emamectin B1 benzoate in feed at a nominal rate of 50 ug of emamectin benzoate/kg/day for 7 consecutive days. Tissues, blood, and bile were collected from 10 fish each at 3 and 12 hr and at 1, 3, 7, 15, 30, 45, 60, and 90 days post final dose. Feces were collected daily from the tanks beginning just prior to dosing to 90 days post final dose. The total radioactive residues (TRR) of the daily feces samples during dosing were 0.25 ppm maximal, and >97% of the TRR in pooled feces covering the dosing period was emamectin B1a. Feces TRR then rapidly declined to approximately 0.05 ppm by 1 day post final dose. The ranges of mean TRR for tissues over the 90 days post dose period were as follows: kidney, 1.4-3 ppm; liver, 1.0-2.3 ppm; skin, 0.04-0.09 ppm; muscle, 0.02-0.06 ppm; and bone, <0.01 ppm. The residue components of liver, kidney, muscle, and skin samples pooled by post dose interval were emamectin B1a (81-100% TRR) and desmethylemamectin B1a (0-17% TRR) with N-formylemamectin B1a seen in trace amounts (<2%) in some muscle samples. The marker residue selected for regulatory surveillance of emamectin residues was emamectin B1a. The emamectin B1a level was quantified in individual samples of skin and muscle using HPLC-fluorometry and was below 85 ppb in all samples analyzed (3 hr to 30 days post dose).
PMID:15053560 Kim-Kang H et al; J Agric Food Chem 52 (7): 2108-18 (2004)
For more Absorption, Distribution and Excretion (Complete) data for EMAMECTIN (8 total), please visit the HSDB record page.
Atlantic salmon (approximately 1.3 kg) maintained in tanks of seawater at 5 +/- 1 degrees C were dosed with [(3)H]emamectin B1 benzoate in feed at a nominal rate of 50 ug of emamectin benzoate/kg/day for 7 consecutive days. Tissues, blood, and bile were collected from 10 fish each at 3 and 12 hr and at 1, 3, 7, 15, 30, 45, 60, and 90 days post final dose. Feces were collected daily from the tanks beginning just prior to dosing to 90 days post final dose. The total radioactive residues (TRR) of the daily feces samples during dosing were 0.25 ppm maximal, and >97% of the TRR in pooled feces covering the dosing period was emamectin B1a. Feces TRR then rapidly declined to approximately 0.05 ppm by 1 day post final dose. The ranges of mean TRR for tissues over the 90 days post dose period were as follows: kidney, 1.4-3 ppm; liver, 1.0-2.3 ppm; skin, 0.04-0.09 ppm; muscle, 0.02-0.06 ppm; and bone, <0.01 ppm. The residue components of liver, kidney, muscle, and skin samples pooled by post dose interval were emamectin B1a (81-100% TRR) and desmethylemamectin B1a (0-17% TRR) with N-formylemamectin B1a seen in trace amounts (<2%) in some muscle samples. The marker residue selected for regulatory surveillance of emamectin residues was emamectin B1a. The emamectin B1a level was quantified in individual samples of skin and muscle using HPLC-fluorometry and was below 85 ppb in all samples analyzed (3 hr to 30 days post dose).
PMID:15053560 Kim-Kang H et al; J Agric Food Chem. 52(7):2108-18 (2004).
...a single mammalian metabolite has been identified. This metabolite is characterized as an N-demethylation byproduct of emamectin.
USDA; Emamectin benzoate Human Health and Ecological Risk Assessment FINAL REPORT, October 28, 2010; Available from, as of March 13, 2014: https://www.fs.fed.us/foresthealth/pesticide/pdfs/052-23-03b_Emamectin-benzoate.pdf
The metabolism of (3)H/(14)C-labeled 4"-deoxy-4"-epimethylaminoavermectin B1a (MAB1a) benzoate, the major homologue (>/=90%) of the avermectin insecticide emamectin benzoate, was studied in laying chickens. Ten Leghorn hens (Gallus domesticus) were orally dosed once daily for 7 days (1 mg/kg of body weight/day). Eggs and excreta were collected daily, and eggs were subsequently separated into whites and yolks. Chickens were euthanized within 20 hr after the last dose, and liver, kidney, heart, muscle, fat, ovaries, gizzard, gastrointestinal tract and contents, and carcass were collected. Approximately 70 and 6% of the total administered dose were recovered in the excreta plus gastrointestinal tract and contents and in the tissues plus eggs, respectively. Two novel metabolites, i.e. the 24-hydroxymethyl derivative of the parent compound (24-hydroxymethyl-4"-deoxy-4"-epimethylaminoavermectin B1a) and the N-demethylated derivative of 24-hydroxymethyl-4"-deoxy-4"-epimethylaminoavermectin B1a (24-hydroxymethyl-4"-deoxy-4"-epiaminoavermectin B1a), were identified. In addition, eight fatty acid conjugates of each of these two metabolites, comprising 8-75% of total radioactive residues in tissues and eggs, were isolated and identified. Although this represents some of the most extensive in vivo fatty acid conjugation to a xenobiotic reported to date, potential human exposure to MAB1a residues from consumption of chicken would be extremely low, because the dosage level in this study was approximately 1000-fold greater than the MAB1a residue levels seen in crops and because the majority of the applied dose was recovered in the excreta. Based on these findings, the avian biotransformation of MAB1a differs substantially from the mammalian biotransformation.
PMID:9698294 Wrzesinski CL et al; Drug Metab Dispos 26 (8): 786-94 (1998)
While emamectin benzoate is not extensively metabolized in mammals, the limited information on the metabolites of emamectin benzoate suggests that metabolism does not result in the detoxification of emamectin benzoate. One plant metabolite of emamectin benzoate is somewhat more toxic than emamectin benzoate itself.
USDA; Emamectin benzoate Human Health and Ecological Risk Assessment FINAL REPORT, October 28, 2010; Available from, as of March 13, 2014: https://www.fs.fed.us/foresthealth/pesticide/pdfs/052-23-03b_Emamectin-benzoate.pdf
Emamectin benzoate was rapidly cleared from plasma /of rats/ with half-lives ranging from about 15 to 28 hours after oral or intravenous dosing.
USDA; Emamectin benzoate Human Health and Ecological Risk Assessment FINAL REPORT, October 28, 2010; Available from, as of March 13, 2014: https://www.fs.fed.us/foresthealth/pesticide/pdfs/052-23-03b_Emamectin-benzoate.pdf
Emamectin benzoate is a relatively large molecule (actually a mixture of four closely related molecules) which is not completely absorbed on oral administration, is poorly absorbed by the dermal administration, and rapidly eliminated in the feces with whole-body half-lives of about 1.5 days.
USDA; Emamectin benzoate Human Health and Ecological Risk Assessment FINAL REPORT, October 28, 2010; Available from, as of March 13, 2014: https://www.fs.fed.us/foresthealth/pesticide/pdfs/052-23-03b_Emamectin-benzoate.pdf
The tissue distribution of tritiated emamectin benzoate after a single oral dose in Atlantic salmon was also investigated by means of whole-body autoradiography and scintillation counting (distribution study). The concentration of emamectin benzoate decreased gradually from the end of treatment (day 7) to day 70 with half-lives of 9.2, 10.0 and 11.3 days in muscle, plasma and mucus respectively.
PMID:15720522 Sevatdal S et al; J Vet Pharmacol Ther. 28(1):101-7 (2005).
Emamectin benzoate is the 4'-deoxy-4'-epi-methyl-amino benzoate salt of avermectin B1 (abamectin), which is similar structurally to natural fermentation products of Streptomyces avermitilis. Emamectin benzoate is being developed as a newer broad-spectrum insecticide for vegetables and has a very low application rate. The mechanism of action involves stimulation of high-affinity GABA receptors and a consequent increase in membrane chloride ion permeability.
PMID:15462160 Yen TH, Lin JL; J Toxicol Clin Toxicol 42 (5): 657-61 (2004)
Overexpression of P-glycoproteins (Pgps) is assumed to be a principal mechanism of resistance of nematodes and arthropods to macrocyclic lactones. Quantitative RT-PCR (Q-RT-PCR) was used to demonstrate changes in transcription levels of two putative P-glycoprotein genes, designated here as SL0525 and SL-Pgp1, in sea lice (Lepeophtheirus salmonis) following exposure to emamectin benzoate (EMB). Pre-adult L. salmonis were challenged in an EMB bioassay for 24 hr and gene expression was studied from lice surviving EMB concentrations of 0, 10, and 30 ppb. Gene expression was measured using Q-RT-PCR with elongation factor 1 (eEF1alpha) as an internal reference gene. The results show that both target genes, SL0525 and SL-Pgp1, had significantly increased levels of expression with exposure to 10ppb EMB (p=0.11 and p=0.17, respectively) whereas the group exposed to 30 ppb was on the verge of being significant (p=0.053) only in the expression of SL-Pgp1. Gene expression for SL0525 and SL-Pgp1 were increased over five-fold at 10 ppb EMB. Therefore, the upregulation of these target genes may offer protection by increasing Pgp expression when lice are exposed to EMB. Optimized Q-RT-PCR can be used to determine if over-expression of these genes could be the basis for development of resistance in sea lice and thus allow suitable alternative chemotherapeutic options to be assessed.
PMID:17350696 Tribble ND et al; Mol Biochem Parasitol. 153(1):59-65 (2007).
Macrocyclic lactones, including avermectins and milbemycins, are novel parasiticides and insecticides that are produced through fermentation by soil-dwelling microorganisms. Although various macrocyclic lactones may differ in their potency and safety, all of them are believed to share common pharmacologic/toxicologic mechanisms, i.e. leading to paralysis and death of parasites and other target organisms via the activation of a glutamate-gated chloride channel in the invertebrate nerve and muscle cells and/or through the effect on gamma-aminobutyric acid (GABA) receptors. Ivermectin is the first macrocyclic lactone that was released for use in both animals and humans, and has demonstrated both excellent efficacy and high tolerability in the treatment of parasite infestations. Other macrocyclic lactones, such as abamectin, emamectin, and moxidectin were subsequently commercialized and have been used as insecticides and acaricides for crop protection or parasiticides for animal health.
PMID:22039794 Yang CC; Curr Pharm Biotechnol 13 (6):999-1003 (2012)
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
92
PharmaCompass offers a list of Emamectin Benzoate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Emamectin Benzoate manufacturer or Emamectin Benzoate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Emamectin Benzoate manufacturer or Emamectin Benzoate supplier.
A Emamectin Benzoate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Emamectin Benzoate, including repackagers and relabelers. The FDA regulates Emamectin Benzoate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Emamectin Benzoate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Emamectin Benzoate supplier is an individual or a company that provides Emamectin Benzoate active pharmaceutical ingredient (API) or Emamectin Benzoate finished formulations upon request. The Emamectin Benzoate suppliers may include Emamectin Benzoate API manufacturers, exporters, distributors and traders.
click here to find a list of Emamectin Benzoate suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
Emamectin Benzoate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Emamectin Benzoate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Emamectin Benzoate GMP manufacturer or Emamectin Benzoate GMP API supplier for your needs.
A Emamectin Benzoate CoA (Certificate of Analysis) is a formal document that attests to Emamectin Benzoate's compliance with Emamectin Benzoate specifications and serves as a tool for batch-level quality control.
Emamectin Benzoate CoA mostly includes findings from lab analyses of a specific batch. For each Emamectin Benzoate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Emamectin Benzoate may be tested according to a variety of international standards, such as European Pharmacopoeia (Emamectin Benzoate EP), Emamectin Benzoate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Emamectin Benzoate USP).

