

Synopsis
Synopsis
0
VMF
0
FDF
0
Australia

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
Annual Reports
NA
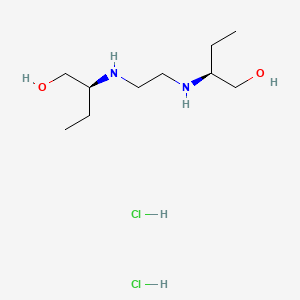

1. Dexambutol
2. Emb Fatol
3. Emb Hefa
4. Emb-fatol
5. Emb-hefa
6. Etambutol Llorente
7. Ethambutol
8. Etibi
9. Hydrochloride, Ethambutol
10. Llorente, Etambutol
11. Miambutol
12. Myambutol
1. Ethambutol Dihydrochloride
2. Ethambutol Hcl
3. 1070-11-7
4. Myambutol
5. Dadibutol
6. Dexambutol
7. Etibi
8. Ethambutol (dihydrochloride)
9. Servambutol
10. Etapiam
11. Mycobutol
12. Tambutol
13. Ebutol
14. Cl 40881
15. Tibutol
16. (2s,2's)-2,2'-(ethane-1,2-diylbis(azanediyl))bis(butan-1-ol) Dihydrochloride
17. Jng307dj5x
18. Qe4vw5fo07
19. Ethambutol Dihydrochloride, Dl-
20. (+)-2,2'-(ethylenediimino)-di-1-butanol Dihydrochloride
21. Nsc-757022
22. Cl-40881
23. 22196-75-4
24. Dsstox_cid_25345
25. Dsstox_rid_80813
26. Dsstox_gsid_45345
27. Ethambutol 2hcl
28. Emb Dihydrochloride
29. Chebi:4878
30. (2s)-2-[2-[[(1s)-1-(hydroxymethyl)propyl]amino]ethylamino]butan-1-ol Dihydrochloride
31. (2s)-2-[2-[[(2s)-1-hydroxybutan-2-yl]amino]ethylamino]butan-1-ol;dihydrochloride
32. Ccris 6606
33. Ncgc00178864-03
34. Einecs 213-970-7
35. Unii-jng307dj5x
36. Unii-qe4vw5fo07
37. (+-)-ethambutol Dihydrochloride
38. Myambutol (tn)
39. Ethambutol Hydrochloride [usan:usp:jan]
40. Ethambutoli Hydrochloridum
41. Emb
42. Ncgc00016560-01
43. Cas-1070-11-7
44. (2s)-2-[(2-{[(2s)-1-hydroxybutan-2-yl]amino}ethyl)amino]butan-1-ol Dihydrochloride
45. Schembl650049
46. Spectrum1500288
47. Chembl3140361
48. Dtxsid4045345
49. Hy-b0535a
50. (+-)-2,2'-(ethylenediimino)-di-1-butanol Dihydrochloride
51. Hms1920m18
52. Kuc109574c
53. (s-(r*,r*))-2,2'-(ethylenediimino)dibutan-1-ol Dihydrochloride
54. Ksc-27-052b
55. Tox21_110494
56. 1-butanol, 2,2'-(ethylenediimino)di-, Dihydrochloride, (+)-
57. Ccg-40232
58. Mfcd21364080
59. S4004
60. Ethambutol Hydrochloride (jp17/usp)
61. Ethambutol Hydrochloride [jan]
62. 1-butanol, (1,2-ethanediyldiimino)bis-, Dihydrochloride, (r*,r*)-(+-)-
63. Akos024258527
64. Tox21_110494_1
65. Cl40881
66. Ethambutol Dihydrochloride [mi]
67. Ethambutol Hydrochloride [usan]
68. Nsc 757022
69. 1-butanol, 2,2'-(1,2-ethanediyldiimino)bis-, Dihydrochloride, (r-(r*,r*))-
70. 1-butanol, 2,2'-(1,2-ethanediyldiimino)bis-, Dihydrochloride, (s-(r*,r*))-
71. Ethambutol Hydrochloride [mart.]
72. Ethambutol Hydrochloride [vandf]
73. Ethambutol Hydrochloride [usp-rs]
74. Ethambutol Hydrochloride [who-ip]
75. Ethambutol Dihydrochloride [who-dd]
76. Ethambutol Dihydrochloride, Antimycobacterial
77. E1011
78. Sw219109-1
79. Ethambutol Hydrochloride [ep Impurity]
80. Ethambutol Hydrochloride [orange Book]
81. D00878
82. Ethambutol Hydrochloride [ep Monograph]
83. Ethambutol Hydrochloride [usp Monograph]
84. Ethambutoli Hydrochloridum [who-ip Latin]
85. A895715
86. Q27106530
87. Z1558572530
88. 2,2'-(1,2-ethanediyldiimino)bis-1-butanol Dihydrochloride
89. 2,2'-[ethane-1,2-diyldi(imino)]dibutan-1-ol Dihydrochloride
90. (s,s)-2,2'-(1,2-ethanediyldiimino)bis-1-butanol Dihydrochloride
91. (s,s)-n,n'-bis(1-hydroxy-2-butyl)ethylenediamine Dihydrochloride
92. 1-butanol, 2,2'-(1,2-ethanediyldiimino)bis-, Dihydrochloride, (r*,r*)-
93. 1-butanol, 2,2'-(ethylenediimino)di-, Dihydrochloride, (+-)-
94. Ethambutol Hydrochloride, European Pharmacopoeia (ep) Reference Standard
95. 1-butanol, 2,2'(1,2-ethanediyldiimino)bis-, Dihydrochloride, (-(r*,r*))
96. 1-butanol, 2,2'-(1,2-ethanediyldiimino)bis-, Dihydrochloride, (2r,2'r)-rel-
97. 1-butanol, 2,2'-(1,2-ethanediyldiimino)bis-, Hydrochloride (1:2), (2s,2's)-
98. Ethambutol For System Suitability, European Pharmacopoeia (ep) Reference Standard
99. Ethambutol Hydrochloride, Pharmaceutical Secondary Standard; Certified Reference Material
100. Ethambutol Hydrochloride, United States Pharmacopeia (usp) Reference Standard
101. 29326-86-1
| Molecular Weight | 277.23 g/mol |
|---|---|
| Molecular Formula | C10H26Cl2N2O2 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 9 |
| Exact Mass | 276.1371335 g/mol |
| Monoisotopic Mass | 276.1371335 g/mol |
| Topological Polar Surface Area | 64.5 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 109 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 2 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
| 1 of 2 | |
|---|---|
| Drug Name | Ethambutol hydrochloride |
| Active Ingredient | Ethambutol hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 100mg; 400mg |
| Market Status | Prescription |
| Company | Lupin; Barr |
| 2 of 2 | |
|---|---|
| Drug Name | Ethambutol hydrochloride |
| Active Ingredient | Ethambutol hydrochloride |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 100mg; 400mg |
| Market Status | Prescription |
| Company | Lupin; Barr |
Antitubercular Agents
Drugs used in the treatment of tuberculosis. They are divided into two main classes: "first-line" agents, those with the greatest efficacy and acceptable degrees of toxicity used successfully in the great majority of cases; and "second-line" drugs used in drug-resistant cases or those in which some other patient-related condition has compromised the effectiveness of primary therapy. (See all compounds classified as Antitubercular Agents.)
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : ETHAMBUTOL HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : 400MG
Approval Date : 2001-11-26
Application Number : 76057
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : ETHAMBUTOL HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : 100MG
Approval Date : 1999-11-30
Application Number : 75095
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : ETHAMBUTOL HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : 400MG
Approval Date : 1999-11-30
Application Number : 75095
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : ETHAMBUTOL HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : 100MG
Approval Date : 2009-06-17
Application Number : 78939
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : ETHAMBUTOL HYDROCHLORIDE
Dosage Form : TABLET;ORAL
Dosage Strength : 400MG
Approval Date : 2009-06-17
Application Number : 78939
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

Brand Name : MYAMBUTOL
Dosage Form : TABLET;ORAL
Dosage Strength : 100MG
Approval Date : 1982-01-01
Application Number : 16320
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AB

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Brand Name : MYAMBUTOL
Dosage Form : TABLET;ORAL
Dosage Strength : 200MG
Approval Date : 1982-01-01
Application Number : 16320
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Brand Name : MYAMBUTOL
Dosage Form : TABLET;ORAL
Dosage Strength : 400MG
Approval Date : 1982-01-01
Application Number : 16320
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AB

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Brand Name : MYAMBUTOL
Dosage Form : TABLET;ORAL
Dosage Strength : 500MG
Approval Date : 1982-01-01
Application Number : 16320
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Rifampicin, Isoniazid, Pyrazinamide, Ethambutol hydrochloride
Brand Name : Rimstar
Dosage Form : Tablet, film-coated
Dosage Strength :
Packaging : Blisterpakning 60item
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Norway
Rifampicin, Isoniazid, Pyrazinamide, Ethambutol hydrochloride
Brand Name : Rimstar
Dosage Form : Tablet, film-coated
Dosage Strength :
Packaging : Blisterpakning 60item
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Norway

Regulatory Info :
Registration Country : Sweden
Brand Name : Myambutol
Dosage Form : DRAGERAD TABLETT
Dosage Strength : 400 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Sweden
Brand Name : Myambutol
Dosage Form : DRAGERAD TABLETT
Dosage Strength : 400 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Sweden
Brand Name : Myambutol
Dosage Form : DRAGERAD TABLETT
Dosage Strength : 400 MG
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 3 companies offering Ethambutol Hydrochloride
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
