
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Europe

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
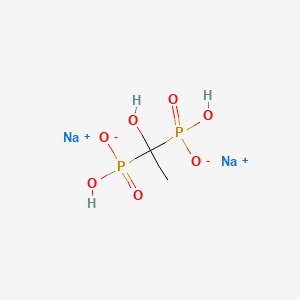

1. (1-hydroxyethylene)diphosphonic Acid
2. (1-hydroxyethylene)diphosphonic Acid, Tetrapotassium Salt
3. 1 Hydroxyethane 1,1 Diphosphonate
4. 1 Hydroxyethylidene 1,1 Bisphosphonate
5. 1,1 Hydroxyethylenediphosphonate
6. 1,1-hydroxyethylenediphosphonate
7. 1-hydroxyethane-1,1-diphosphonate
8. 1-hydroxyethylene Diphosphonate, Disodium
9. 1-hydroxyethylidene-1,1-bisphosphonate
10. Dicalcium Ehdp
11. Dicalcium Etidronate
12. Didronel
13. Diphosphonate, Disodium 1-hydroxyethylene
14. Diphosphonic Acid, Hydroxyethylidene
15. Disodium 1 Hydroxyethylene Diphosphonate
16. Disodium 1-hydroxyethylene Diphosphonate
17. Disodium Etidronate
18. Ehdp
19. Ehdp, Dicalcium
20. Ethanehydroxydiphosphonate
21. Ethanehydroxyphosphate
22. Etidronate
23. Etidronate, Dicalcium
24. Etidronate, Disodium
25. Etidronate, Sodium
26. Etidronate, Tetrapotassium Salt
27. Etidronic Acid
28. Hedp
29. Hedspa
30. Hydroxyethanediphosphonate
31. Hydroxyethylidene Diphosphonic Acid
32. Phosphonic Acid, (1-hydroxyethylidene)bis-, Disodium Salt
33. Salt Etidronate, Tetrapotassium
34. Sodium Etidronate
35. Tetrapotassium Salt Etidronate
36. Xidifon
37. Xidiphon
38. Xydiphone
1. 7414-83-7
2. Didronel
3. Disodium Etidronate
4. Sodium Etidronate
5. Sodium Ethidronate
6. Sodium Ethydronate
7. Disodium Ethydronate
8. Turpinal 2nz
9. Disodium Ethanol-1,1-diphosphonate
10. Etidronic Acid Disodium Salt
11. Etidronatedisodium
12. Disodium (1-hydroxyethylidene)diphosphonate
13. Phosphonic Acid, (1-hydroxyethylidene)bis-, Disodium Salt
14. Disodium 1-hydroxyethylidene Phosphonate
15. Chebi:4906
16. Hedpa;hedp
17. Disodium Dihydrogen (1-hydroxyethylidene)diphosphonate
18. Disodium Ethane-1-hydroxy-1,1-diphosphonate
19. (1-hydroxyethylidene)diphosphonic Acid, Disodium Salt
20. Etidronic Acid, Disodium Salt
21. 1-hydroxyethylidene-1,1-diphosphonic Acid Disodium Salt
22. (1-hydroxyethane-1,1-diyl)diphosphonic Acid Disodium Salt
23. M16pxg993g
24. Didronel R
25. 1-hydroxyethane-1,1-diphosphonic Acid Disodium Salt
26. Nsc-759157
27. Cas-7414-83-7
28. Dsstox_cid_9671
29. Disodium;hydroxy-[1-hydroxy-1-[hydroxy(oxido)phosphoryl]ethyl]phosphinate
30. Dsstox_rid_78804
31. Dsstox_gsid_29671
32. Disodium (1-hydroxyethane-1,1-diyl)bis[hydrogen (phosphonate)]
33. Etidronsaeure Dinatriumsalz
34. Etidronate Disodium Hydrate
35. Etidronic Acid Disodium
36. Ncgc00159352-02
37. 29329-71-3
38. Einecs 231-025-7
39. Sm-5600
40. Unii-m16pxg993g
41. Didronel Iv
42. Didronel Pmo
43. Phosphonic Acid, (1-hydroxyethylidene)bis-, Sodium Salt
44. Didronel (tn)
45. Ethane-1-hydroxy-1,1-diphosphonate, Disodium Salt
46. Etidronate Disodium [usan:usp:jan]
47. Mfcd00152567
48. Prestwick_1061
49. Disodium Dihydrogen (1-hydroxyethylidene)bisphosphonate
50. Ethane-1-hydroxy-1,1-diphosphonic Acid, Disodium Salt
51. Phosphonic Acid, (1-hydroxyethylidene)di-, Disodium Salt
52. Disodium Hydroxy-[1-hydroxy-1-[hydroxy(oxido)phosphoryl]ethyl]phosphinate
53. Chembl1201042
54. Dtxsid1029671
55. Etidronate Disodium [jan]
56. Etidronate Disodium (jp17/usp)
57. Disodium Etidronate [inci]
58. Etidronate Disodium [usan]
59. Hms1570n07
60. Hms2093p03
61. Hms2097n07
62. Etidronate Disodium [vandf]
63. Bcp22743
64. Etidronate Disodium [mart.]
65. Tox21_111596
66. Bdbm50247999
67. Etidronate Disodium [usp-rs]
68. Etidronate Disodium [who-dd]
69. Phosphonic Acid, P,p'-(1-hydroxyethylidene)bis-, Sodium Salt (1:2)
70. Akos015961949
71. Tox21_111596_1
72. Ac-2078
73. Ccg-213569
74. Ks-1297
75. Nsc 759157
76. Etidronate Disodium [orange Book]
77. Ncgc00017072-01
78. Ncgc00179380-04
79. Be164429
80. Etidronate Disodium [ep Monograph]
81. Etidronic Acid Disodium Salt [mi]
82. Etidronate Disodium [usp Monograph]
83. Disodium 1-hydroxyethane-1,1-diphosphonate
84. D4159
85. Ft-0630369
86. D00314
87. H11387
88. Etidronate Disodium;hedpa Disodium;hedp Disodium
89. A838033
90. Etidronate Disodium Hydrate, >=97% (nmr), Solid
91. W-104431
92. 1-hydroxyethane 1,1-diphosphonic Acid Disodium Salt
93. Disodium Etidronate;sm-5600; Sm-5600; Sm-5600
94. Q27106537
95. Sodium 1-hydroxyethane-1,1-diylbis(hydrogenphosphonate)
96. Disodium [1-[oxidanidyl(oxidanyl)phosphoryl]-1-oxidanyl-ethyl]-oxidanyl-phosphinate
| Molecular Weight | 249.99 g/mol |
|---|---|
| Molecular Formula | C2H6Na2O7P2 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 2 |
| Exact Mass | 249.93841509 g/mol |
| Monoisotopic Mass | 249.93841509 g/mol |
| Topological Polar Surface Area | 141 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 206 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
| 1 of 4 | |
|---|---|
| Drug Name | Didronel |
| Active Ingredient | Etidronate disodium |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 400mg |
| Market Status | Prescription |
| Company | Procter And Gamble |
| 2 of 4 | |
|---|---|
| Drug Name | Etidronate disodium |
| PubMed Health | Etidronate (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Etidronate disodium tablets, USP contain either 200 mg or 400 mg of etidronate disodium, the disodium salt of (1-hydroxyethylidene) diphosphonic acid, for oral administration. This compound, also known as EHDP, regulates bone metabolism. Etidronate d... |
| Active Ingredient | Etidronate disodium |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 400mg |
| Market Status | Prescription |
| Company | Mylan |
| 3 of 4 | |
|---|---|
| Drug Name | Didronel |
| Active Ingredient | Etidronate disodium |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 400mg |
| Market Status | Prescription |
| Company | Procter And Gamble |
| 4 of 4 | |
|---|---|
| Drug Name | Etidronate disodium |
| PubMed Health | Etidronate (By mouth) |
| Drug Classes | Calcium Regulator |
| Drug Label | Etidronate disodium tablets, USP contain either 200 mg or 400 mg of etidronate disodium, the disodium salt of (1-hydroxyethylidene) diphosphonic acid, for oral administration. This compound, also known as EHDP, regulates bone metabolism. Etidronate d... |
| Active Ingredient | Etidronate disodium |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg; 400mg |
| Market Status | Prescription |
| Company | Mylan |
Bone Density Conservation Agents
Agents that inhibit BONE RESORPTION and/or favor BONE MINERALIZATION and BONE REGENERATION. They are used to heal BONE FRACTURES and to treat METABOLIC BONE DISEASES such as OSTEOPOROSIS. (See all compounds classified as Bone Density Conservation Agents.)
Related Excipient Companies

Excipients by Applications
Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
18
PharmaCompass offers a list of Etidronate Disodium API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Etidronate Disodium manufacturer or Etidronate Disodium supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Etidronate Disodium manufacturer or Etidronate Disodium supplier.
PharmaCompass also assists you with knowing the Etidronate Disodium API Price utilized in the formulation of products. Etidronate Disodium API Price is not always fixed or binding as the Etidronate Disodium Price is obtained through a variety of data sources. The Etidronate Disodium Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Etidronate Disodium manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Etidronate Disodium, including repackagers and relabelers. The FDA regulates Etidronate Disodium manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Etidronate Disodium API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Etidronate Disodium manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Etidronate Disodium supplier is an individual or a company that provides Etidronate Disodium active pharmaceutical ingredient (API) or Etidronate Disodium finished formulations upon request. The Etidronate Disodium suppliers may include Etidronate Disodium API manufacturers, exporters, distributors and traders.
click here to find a list of Etidronate Disodium suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Etidronate Disodium DMF (Drug Master File) is a document detailing the whole manufacturing process of Etidronate Disodium active pharmaceutical ingredient (API) in detail. Different forms of Etidronate Disodium DMFs exist exist since differing nations have different regulations, such as Etidronate Disodium USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Etidronate Disodium DMF submitted to regulatory agencies in the US is known as a USDMF. Etidronate Disodium USDMF includes data on Etidronate Disodium's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Etidronate Disodium USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Etidronate Disodium suppliers with USDMF on PharmaCompass.
A Etidronate Disodium CEP of the European Pharmacopoeia monograph is often referred to as a Etidronate Disodium Certificate of Suitability (COS). The purpose of a Etidronate Disodium CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Etidronate Disodium EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Etidronate Disodium to their clients by showing that a Etidronate Disodium CEP has been issued for it. The manufacturer submits a Etidronate Disodium CEP (COS) as part of the market authorization procedure, and it takes on the role of a Etidronate Disodium CEP holder for the record. Additionally, the data presented in the Etidronate Disodium CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Etidronate Disodium DMF.
A Etidronate Disodium CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Etidronate Disodium CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Etidronate Disodium suppliers with CEP (COS) on PharmaCompass.
Etidronate Disodium Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Etidronate Disodium GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Etidronate Disodium GMP manufacturer or Etidronate Disodium GMP API supplier for your needs.
A Etidronate Disodium CoA (Certificate of Analysis) is a formal document that attests to Etidronate Disodium's compliance with Etidronate Disodium specifications and serves as a tool for batch-level quality control.
Etidronate Disodium CoA mostly includes findings from lab analyses of a specific batch. For each Etidronate Disodium CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Etidronate Disodium may be tested according to a variety of international standards, such as European Pharmacopoeia (Etidronate Disodium EP), Etidronate Disodium JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Etidronate Disodium USP).
