
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
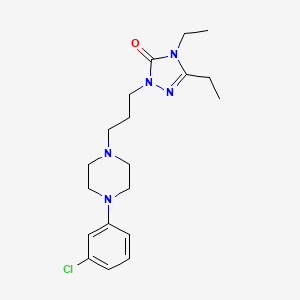

1. (1,3-(4-m-chlorophenyl-1-piperazinyl)propyl)-3,4-diethyl-delta(2)-1,2,4-triazolin-5-one
2. Etoperidone Monohydrochloride
1. 52942-31-1
2. Etoperidone [inn]
3. Kai6mvo39z
4. 2-[3-[4-(3-chlorophenyl)piperazin-1-yl]propyl]-4,5-diethyl-1,2,4-triazol-3-one
5. Etoperidona
6. Etoperidonum
7. Etoperidona [spanish]
8. Unii-kai6mvo39z
9. Etoperidonum [inn-latin]
10. Etoperidona [inn-spanish]
11. Etoperidone [mi]
12. Schembl49314
13. Etoperidone [who-dd]
14. Chembl1743259
15. Dtxsid0023034
16. Bdbm82438
17. Chebi:135589
18. Zinc3830815
19. Nsc_40589
20. Pdsp1_000523
21. Pdsp2_000521
22. Db09194
23. Cas_52942-31-1
24. L001188
25. Q5404839
26. 1,2,4-triazol-3-one, 2-(3-(4-(3-chlorophenyl)-1-piperazinyl)propyl)-4,5-diethyl-2,4-dihydro-
27. 1-(3-(4-(m-chlorophenyl)-1-piperazinyl)propyl)-3,4-diethyl-d2-1,2,4-triazolin-5-one
28. 2-{3-[4-(3-chlorophenyl)piperazin-1-yl]propyl}-4,5-diethyl-2,4-dihydro-3h-1,2,4-triazol-3-one
| Molecular Weight | 377.9 g/mol |
|---|---|
| Molecular Formula | C19H28ClN5O |
| XLogP3 | 3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 7 |
| Exact Mass | 377.1982382 g/mol |
| Monoisotopic Mass | 377.1982382 g/mol |
| Topological Polar Surface Area | 42.4 Ų |
| Heavy Atom Count | 26 |
| Formal Charge | 0 |
| Complexity | 506 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Etoperidone has been studied for the treatment of depression, tremors in Parkinson, extrapyramidal symptoms and male impotence. It is not certain if it was ever approved and marketed but its current status is withdrawn.
Etoperidone has a biphasic effect on the central transmission of serotonin. It presents the capacity to inhibit serotonin receptor but also to inhibit the reuptake of serotonin, norepinephrine and dopamine. As part of its actions, etoperidone also inhibits the -adrenergic receptors which directly corresponds to the sedative and cardiovascular effects. The presence of both effects caused that the effective dose of etoperidone was poorly tolerated thus, efforts have been made to separate the serotonergic and adrenergic functions in order to generate etoperidone-derivatives like nefazodone.
N - Nervous system
N06 - Psychoanaleptics
N06A - Antidepressants
N06AB - Selective serotonin reuptake inhibitors
N06AB09 - Etoperidone
Absorption
The absorption and bioavailability is highly variable between individuals and can be as low as 12%. The lower bioavailability is explained due to its high metabolism. The mean time to peak plasma concentration is ranged from 1.4-4.8 hours.
Route of Elimination
The elimination of an oral dose of etoperidone presents a division of 78.8% found in urine and 9.6% found in faeces. On the elimination route, less than 0.01% of the etoperidone dose is represented by the unchanged drug while the rest is formed by 21 different metabolites.
Volume of Distribution
The high protein binding presented in etoperidone modulates its volume of distribution to a range of 0.23 to 0.69 L/kg.
Clearance
The apparent clearance of etoperidone was 1.01 ml/min.
Etoperidone is highly metabolized and it forms 21 different metabolites that can be found in plasma, urine and faeces. The metabolism of etoperidone is thought to be related to 5 different reaction pathways that are alkyl oxidation, piperazinyl oxidation, N-dealkylation, phenyl hydroxylation and conjugation.
Etoperidone has known human metabolites that include 1-(3-Chlorophenyl)piperazine, 2-[3-[4-(3-Chlorophenyl)piperazin-1-yl]propyl]-4-ethyl-5-(1-hydroxyethyl)-1,2,4-triazol-3-one, 2-[3-[4-(3-chloro-4-hydroxyphenyl)piperazin-1-yl]propyl]-4,5-diethyl-1,2,4-triazol-3-one, and 4,5-diethyl-2-propyl-1,2,4-triazol-3-one.
S73 | METXBIODB | Metabolite Reaction Database from BioTransformer | DOI:10.5281/zenodo.4056560
After oral administration of etoperidone the terminal half-life was 21.7 hours.
The activity of etoperidone is made mainly by its major metabolite 1-(3'-chlorophenyl)piperazine (mCPP). mCPP binds with different affinity to most of the serotonergic receptors and adrenergic receptors. This metabolite is an agonist of 5-HT2c and an antagonist of 5-HT2a. Part of etoperidone structure contibutes to the activity in the -adrenergic receptors.
ABOUT THIS PAGE
46
PharmaCompass offers a list of Etoperidone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Etoperidone manufacturer or Etoperidone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Etoperidone manufacturer or Etoperidone supplier.
PharmaCompass also assists you with knowing the Etoperidone API Price utilized in the formulation of products. Etoperidone API Price is not always fixed or binding as the Etoperidone Price is obtained through a variety of data sources. The Etoperidone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Etoperidone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Etoperidone, including repackagers and relabelers. The FDA regulates Etoperidone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Etoperidone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Etoperidone supplier is an individual or a company that provides Etoperidone active pharmaceutical ingredient (API) or Etoperidone finished formulations upon request. The Etoperidone suppliers may include Etoperidone API manufacturers, exporters, distributors and traders.
Etoperidone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Etoperidone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Etoperidone GMP manufacturer or Etoperidone GMP API supplier for your needs.
A Etoperidone CoA (Certificate of Analysis) is a formal document that attests to Etoperidone's compliance with Etoperidone specifications and serves as a tool for batch-level quality control.
Etoperidone CoA mostly includes findings from lab analyses of a specific batch. For each Etoperidone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Etoperidone may be tested according to a variety of international standards, such as European Pharmacopoeia (Etoperidone EP), Etoperidone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Etoperidone USP).
