

Synopsis
Synopsis
0
VMF
0
Australia

0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
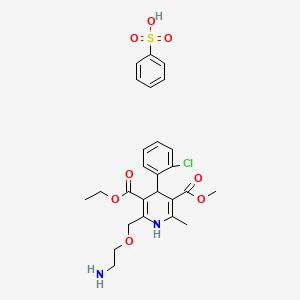

1. Amlodipine
2. Amlodipine Maleate
3. Amlodipine Maleate (1:1)
4. Amlodipine, (+-)-isomer
5. Amlodipine, (+-)-isomer, Maleate (1:1)
6. Amlodipine, (r)-isomer
7. Amlodipine, (s)-isomer, Maleate (1:1)
8. Amlodis
9. Amlor
10. Astudal
11. Istin
12. Norvasc
1. 111470-99-6
2. Amlodipine Besilate
3. Amlodipine Benzenesulfonate
4. Antacal
5. Monopina
6. Istin
7. Amlodipine (besylate)
8. Amdepin
9. Amdipin
10. Amlodin
11. Pelmec
12. 3-ethyl 5-methyl 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
13. Lodipressin
14. Amlor
15. Amlodipine (as Besilate)
16. Nsc-758922
17. (s)-amlodipine;levoamlodipine
18. Amlodipine Besylate (norvasc)
19. Chebi:2669
20. Cardiorex
21. Uk-48,340-26
22. 111470-99-6 (besylate)
23. 864v2q084h
24. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+-)-, Monobenzenesulfonate
25. Ncgc00095835-01
26. Amlogard
27. Amlopin
28. Amlosyn
29. Astudar
30. Cordarene
31. Myodura
32. Norlopin
33. Norvask
34. Tensivask
35. Amcard
36. Norvas
37. Terloc
38. Norvasc (tn)
39. Uk-48340-26
40. (s)-amlodipine Besylate;levoamlodipine Besylate
41. Dsstox_cid_23909
42. Dsstox_rid_80087
43. Dsstox_gsid_43909
44. Benzenesulfonic Acid;3-o-ethyl 5-o-methyl 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate
45. Stamlo
46. Amlodipine Besylate [usan]
47. Cas-111470-99-6
48. C20h25cln2o5.c6h6o3s
49. Ncgc00165957-04
50. Amlodipine 100 Microg/ml In Acetonitrile
51. Unii-864v2q084h
52. Amlodipine Besylate [usan:usp]
53. Uk 48340-26
54. 3-ethyl 5-methyl 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
55. Mfcd00887594
56. Norliqva
57. Spectrum_000573
58. Amlodipine Benzenesulphonate
59. Amlodipine Besylate (usp)
60. Amlodipine Besylate- Bio-x
61. Amlodipine Besilate (jp17)
62. Schembl41282
63. Kbioss_001053
64. 3-ethyl 5-methyl (+-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulfonate
65. Mls001361394
66. Spectrum1505202
67. Chembl1200402
68. Chembl3193637
69. Chembl3207778
70. Dtxsid2043909
71. Hy-b0317b
72. Kbio2_001053
73. Kbio2_003621
74. Kbio2_006189
75. Amlodipine Besilate [jan]
76. Amlodipine Besylate [hsdb]
77. Hms2094e13
78. Hms2232d22
79. Hms2235b19
80. Hms3269b19
81. Hms3372o18
82. Hms3373f15
83. Hms3413h11
84. Hms3677h11
85. Hms3884m10
86. Pharmakon1600-01505202
87. Amlodipine Besylate [vandf]
88. Amlodipine Besilate [mart.]
89. Bcp21993
90. Tox21_111525
91. Tox21_113476
92. Ac-079
93. Amlodipine Besilate [who-dd]
94. Amlodipine Besylate [usp-rs]
95. Ccg-39531
96. Nsc758922
97. S1813
98. Akos001260256
99. Akos016340493
100. Amlodipine Besylate [ema Epar]
101. Amlodipine Besylate, >=98% (hplc)
102. Bcp9000059
103. Cs-3988
104. Ks-1029
105. Nsc 758922
106. Amlodipine Benzenesulfonate [mi]
107. Amlodipine Besylate [orange Book]
108. Azor Component Amlodipine Besylate
109. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl-5-methyl Ester, Monobenzenesulfonate
110. Amlodipine Besilate [ep Monograph]
111. Amlodipine Besylate [usp Impurity]
112. Ba164162
113. Smr000875356
114. Amlodipine Besylate [usp Monograph]
115. Caduet Component Amlodipine Besylate
116. Dafiro Component Amlodipine Besilate
117. Lotrel Component Amlodipine Besylate
118. Amlessa Component Amlodipine Besylate
119. Copalia Component Amlodipine Besylate
120. Exforge Component Amlodipine Besilate
121. Exforge Component Amlodipine Besylate
122. Imprida Component Amlodipine Besilate
123. Onduarp Component Amlodipine Besylate
124. Tekamlo Component Amlodipine Besylate
125. Twynsta Component Amlodipine Besylate
126. Amlodipine Besylate Component Of Azor
127. Consensi Component Amlodipine Besylate
128. Ft-0622301
129. Sw198880-2
130. Amlodipine Besilate Component Of Dafiro
131. Amlodipine Besylate Component Of Caduet
132. Amlodipine Besylate Component Of Lotrel
133. Amturnide Component Amlodipine Besylate
134. Tribenzor Component Amlodipine Besylate
135. Vu0244594-3
136. Amlodipine Besilate Component Of Exforge
137. Amlodipine Besylate Component Of Copalia
138. Amlodipine Besylate Component Of Exforge
139. Amlodipine Besylate Component Of Onduarp
140. Amlodipine Besylate Component Of Tekamlo
141. Amlodipine Besylate Component Of Twynsta
142. D00615
143. Dafiro-hct Component Amlodipine Besilate
144. F17340
145. Amlodipine Besylate [ema Epar Veterinary]
146. Amlodipine Besylate Component Of Amturnide
147. Amlodipine Besylate Component Of Consensi
148. Amlodipine Besylate Component Of Tribenzor
149. Exforge Hct Component Amlodipine Besylate
150. Exforge-hct Component Amlodipine Besilate
151. Imprida-hct Component Amlodipine Besilate
152. 470a996
153. A802368
154. Amlodipine Besilate Component Of Dafiro-hct
155. Amlodipine Besilate Component Of Exforge-hct
156. Amlodipine Besilate Component Of Imprida-hct
157. Amlodipine Besylate Component Of Exforge Hct
158. Q-101934
159. Q-200627
160. Sr-05000001461-2
161. Q27105760
162. F6170-0024
163. Amlodipine Besylate, European Pharmacopoeia (ep) Reference Standard
164. Amlodipine Besylate, United States Pharmacopeia (usp) Reference Standard
165. Amlodipine Besylate, Pharmaceutical Secondary Standard; Certified Reference Material
166. Amlodipine For Peak Identification, European Pharmacopoeia (ep) Reference Standard
167. (+/-)-3-ethyl 5-methyl 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate Monobenzenesulfonate
168. 2-(2-aminoethoxy)methyl-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylic Acid 3-ethyl 5-methyl Ester Benzenesulfona
169. 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylic Acid O3-ethyl Ester O5-methyl Ester; Benzenesulfonic Acid
170. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbo Xylic Acid 3-ethyl 5-methyl Ester Benzenesulfonate
171. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylic Acid 3-ethyl 5-methyl Ester Benzenesulfonate
172. 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-3-ethoxycarbonyl-5-methoxycarbonyl-6-methyl-1,4-dihydropyridine Benzenesulfonate
173. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+/-)-, Monobenzenesulfonate
174. 3,5-pyridinedicarboxylic Acid, 2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, (+/-)-, Monobenzenesulphonate
175. 3,5-pyridinedicarboxylic Acid, 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-, 3-ethyl 5-methyl Ester, Benzenesulfonate (1:1)
176. 3-ethyl 5-methyl (+/-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulfonate
177. 3-ethyl 5-methyl (+/-)-2-((2-aminoethoxy)methyl)-4-(o-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, Monobenzenesulphonate
178. 3-ethyl 5-methyl 2-(2-aminoethoxy)methyl-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate Benzenesulfonate
179. 3-ethyl 5-methyl 2-(2-aminoethoxymethyl)-4-(2-chlorophenyl)-1,4-dihydro-6-methylpyridine-3,5-dicarboxylate Monobenzenesulphonate
180. 3-ethyl5-methyl2-((2-aminoethoxy)methyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylatebenzenesulfonate
181. Benzenesulfonic Acid; O3-ethyl O5-methyl 2-(2-azanylethoxymethyl)-4-(2-chlorophenyl)-6-methyl-1,4-dihydropyridine-3,5-dicarboxylate
1. Prestalia
| Molecular Weight | 567.1 g/mol |
|---|---|
| Molecular Formula | C26H31ClN2O8S |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 11 |
| Exact Mass | 566.1489648 g/mol |
| Monoisotopic Mass | 566.1489648 g/mol |
| Topological Polar Surface Area | 163 Ų |
| Heavy Atom Count | 38 |
| Formal Charge | 0 |
| Complexity | 830 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 4 | |
|---|---|
| Drug Name | Amlodipine besylate |
| PubMed Health | Amlodipine (By mouth) |
| Drug Classes | Antianginal, Antihypertensive, Cardiovascular Agent |
| Drug Label | Amlodipine besylate is the besylate salt of amlodipine, a long-acting calcium channel blocker. Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbox... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ranbaxy; Amneal Pharms Ny; Upsher Smith; Secan Pharms; Matrix Labs; Epic Pharma; Macleods Pharms; Vivimed Labs; Teva; Apotex; Accord Hlthcare; Vintage; Alkem; Sun Pharm Inds; Aurobindo Pharma; Torrent Pharms; Zydus Pharms Usa; Lupi |
| 2 of 4 | |
|---|---|
| Drug Name | Norvasc |
| Drug Label | NORVASC is the besylate salt of amlodipine, a long-acting calcium channel blocker.Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobe... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Pfizer |
| 3 of 4 | |
|---|---|
| Drug Name | Amlodipine besylate |
| PubMed Health | Amlodipine (By mouth) |
| Drug Classes | Antianginal, Antihypertensive, Cardiovascular Agent |
| Drug Label | Amlodipine besylate is the besylate salt of amlodipine, a long-acting calcium channel blocker. Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarbox... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Watson Labs; Wockhardt; Ranbaxy; Amneal Pharms Ny; Upsher Smith; Secan Pharms; Matrix Labs; Epic Pharma; Macleods Pharms; Vivimed Labs; Teva; Apotex; Accord Hlthcare; Vintage; Alkem; Sun Pharm Inds; Aurobindo Pharma; Torrent Pharms; Zydus Pharms Usa; Lupi |
| 4 of 4 | |
|---|---|
| Drug Name | Norvasc |
| Drug Label | NORVASC is the besylate salt of amlodipine, a long-acting calcium channel blocker.Amlodipine besylate is chemically described as 3-Ethyl-5-methyl ()-2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-1,4-dihydro-6-methyl-3,5-pyridinedicarboxylate, monobe... |
| Active Ingredient | Amlodipine besylate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | eq 5mg base; eq 2.5mg base; eq 10mg base |
| Market Status | Prescription |
| Company | Pfizer |
Treatment of systemic arterial hypertension in cats.
Antihypertensive Agents
Drugs used in the treatment of acute or chronic vascular HYPERTENSION regardless of pharmacological mechanism. Among the antihypertensive agents are DIURETICS; (especially DIURETICS, THIAZIDE); ADRENERGIC BETA-ANTAGONISTS; ADRENERGIC ALPHA-ANTAGONISTS; ANGIOTENSIN-CONVERTING ENZYME INHIBITORS; CALCIUM CHANNEL BLOCKERS; GANGLIONIC BLOCKERS; and VASODILATOR AGENTS. (See all compounds classified as Antihypertensive Agents.)
Calcium Channel Blockers
A class of drugs that act by selective inhibition of calcium influx through cellular membranes. (See all compounds classified as Calcium Channel Blockers.)
Vasodilator Agents
Drugs used to cause dilation of the blood vessels. (See all compounds classified as Vasodilator Agents.)
QC08CA01
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
GDUFA
DMF Review : Complete
Rev. Date : 2013-05-21
Pay. Date : 2013-05-10
DMF Number : 16473
Submission : 2003-03-19
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 17070
Submission : 2003-12-29
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2013-06-19
Pay. Date : 2013-06-07
DMF Number : 16911
Submission : 2003-09-13
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2017-03-17
Pay. Date : 2017-03-06
DMF Number : 17032
Submission : 2003-12-17
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15127
Submission : 2000-11-02
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 17584
Submission : 2004-08-02
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 16478
Submission : 2003-03-18
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 16887
Submission : 2003-10-02
Status : Inactive
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2012-11-30
Pay. Date : 2012-11-13
DMF Number : 15777
Submission : 2001-12-21
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-09-14
Pay. Date : 2013-09-06
DMF Number : 16509
Submission : 2003-04-01
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Registration Number : 219MF10352
Registrant's Address : 8-2-337, Road No. 3, Banjara Hills, Hyderabad 500 034, TELANGANA, INDIA
Initial Date of Registration : 2007-12-10
Latest Date of Registration : 2009-10-13
Registration Number : 304MF10142
Registrant's Address : Cesar Martinell i Brunet 12A, Poligono Rubi Sur, Rubi (Barcelona), Spain
Initial Date of Registration : 2022-10-19
Latest Date of Registration : 2022-10-19
Registration Number : 219MF10350
Registrant's Address : Cesar Martinell i Brunet 12A, Poligono Rubi Sur, Rubi (Barcelona), Spain
Initial Date of Registration : 2007-12-06
Latest Date of Registration : 2012-11-12
Registration Number : 218MF10894
Registrant's Address : Cesar Martinell i Brunet 12A, Poligono Rubi Sur, Rubi (Barcelona), Spain
Initial Date of Registration : 2006-11-10
Latest Date of Registration : 2006-11-10
Registration Number : 229MF10060
Registrant's Address : 2-36-1 Kanda Jinbocho, Chiyoda-ku, Tokyo
Initial Date of Registration : 2017-03-03
Latest Date of Registration : 2020-03-16

Registration Number : 301MF10107
Registrant's Address : No. 1558, Longjiang Bei Road, Chunjiang Town, New-North District, Changzhou City, Jia...
Initial Date of Registration : 2019-12-10
Latest Date of Registration : 2021-06-18

Registration Number : 221MF10131
Registrant's Address : Cipla House, Peninsula Business Park, Ganpatrao Kadam Marg, Lower Parel, Mumbai - 400...
Initial Date of Registration : 2009-06-30
Latest Date of Registration : 2021-05-20

Registration Number : 302MF10027
Registrant's Address : GV Chambers, 7-2-C8 & C8/2, IDA Sanath nagar, HYDERABAD-500 018, Telangana state, IND...
Initial Date of Registration : 2020-02-06
Latest Date of Registration : 2020-02-06

Registration Number : 222MF10151
Registrant's Address : Verovskova ulica 57, 1000 Ljubljana, Slovenia
Initial Date of Registration : 2010-05-17
Latest Date of Registration : 2022-01-20

Registration Number : 306MF10008
Registrant's Address : Plot No. 10, Road No. 8, Film Nagar, Jubilee Hills, Hyderabad-500 096, Telangana, Ind...
Initial Date of Registration : 2024-01-17
Latest Date of Registration : 2024-01-17

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Norliqva (Amlodipine) oral solution, 1 mg/mL, first and only FDA-approved oral liquid solution of the besylate salt of amlodipine, a long-acting calcium channel blocker, for treatment of hypertension in patients 6 years of age and older.
Lead Product(s): Amlodipine Besylate
Therapeutic Area: Cardiology/Vascular Diseases Brand Name: Norliqva
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 01, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Amlodipine Besylate
Therapeutic Area : Cardiology/Vascular Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
CMP Pharma, Inc Announces that Norliqva®, the First and Only FDA-approved Liquid Solution of Amlo...
Details : Norliqva (Amlodipine) oral solution, 1 mg/mL, first and only FDA-approved oral liquid solution of the besylate salt of amlodipine, a long-acting calcium channel blocker, for treatment of hypertension in patients 6 years of age and older.
Brand Name : Norliqva
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 01, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The objective is the contract development of a Single Pill with a combination of three leading active ingredients, including candesartan cilexetil, amlodipine besylate and atorvastatin calcium trihydrate for the treatment of high blood pressure (hypertension).
Lead Product(s): Candesartan Cilexetil,Amlodipine Besylate,Atorvastatin
Therapeutic Area: Cardiology/Vascular Diseases Brand Name: Undisclosed
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Midas Pharma
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Partnership December 14, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Candesartan Cilexetil,Amlodipine Besylate,Atorvastatin
Therapeutic Area : Cardiology/Vascular Diseases
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Midas Pharma
Deal Size : Undisclosed
Deal Type : Partnership
Details : The objective is the contract development of a Single Pill with a combination of three leading active ingredients, including candesartan cilexetil, amlodipine besylate and atorvastatin calcium trihydrate for the treatment of high blood pressure (hyperten...
Brand Name : Undisclosed
Molecule Type : Small molecule
Upfront Cash : Undisclosed
December 14, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Burke will market the fixed-dose combination drug, Consensi to lower blood pressure, and celecoxib for the management of the signs and symptoms of osteoarthritis.
Lead Product(s): Amlodipine Besylate,Celecoxib
Therapeutic Area: Neurology Brand Name: Undisclosed
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Coeptis Pharmaceuticals
Deal Size: Undisclosed Upfront Cash: Undisclosed
Deal Type: Agreement March 11, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Amlodipine Besylate,Celecoxib
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Coeptis Pharmaceuticals
Deal Size : Undisclosed
Deal Type : Agreement
Burke Therapeutics, LLC Signs Exclusive US Marketing and Supply Agreement
Details : Burke will market the fixed-dose combination drug, Consensi to lower blood pressure, and celecoxib for the management of the signs and symptoms of osteoarthritis.
Brand Name : Undisclosed
Molecule Type : Small molecule
Upfront Cash : Undisclosed
March 11, 2020

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

CAS Number : 111470-99-6
Quantity Per Vial : 350
Sale Unit : mg
Price : $245.00
Details : Material Origin- Chemical Synthesis; USMCA- N...
Monograph :
Storage :
Code/Batch No : Catalog #1029501 / R09060

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
