

Synopsis
Synopsis
0
KDMF
0
VMF
0
Canada

0
Australia

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA
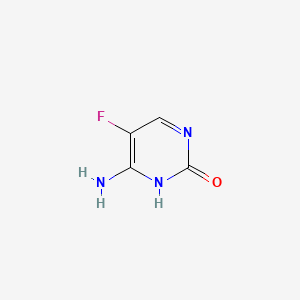

1. 5-fluorocytosine
2. Alcobon
3. Ancobon
4. Ancotil
1. 5-fluorocytosine
2. 2022-85-7
3. Ancobon
4. Ancotil
5. Fluorocytosine
6. 5-fluorocystosine
7. 5-fc
8. Flucytosin
9. Fluocytosine
10. 5-fluorocytosin
11. Fluorcytosine
12. Alcobon
13. 4-amino-5-fluoropyrimidin-2(1h)-one
14. Ro 2-9915
15. Flucytosinum
16. Cytosine, 5-fluoro-
17. 5-fluoro Cytosine
18. 4-amino-5-fluoro-2(1h)-pyrimidinone
19. 4-amino-5-fluoropyrimidin-2-ol
20. 2(1h)-pyrimidinone, 4-amino-5-fluoro-
21. 117969-88-7
22. 5-flurocytosine
23. 5-flucytosine
24. 2-hydroxy-4-amino-5-fluoropyrimidine
25. Ancobon (tn)
26. Ro 29915 E/265601
27. Nsc 103805
28. 6-amino-5-fluoro-1h-pyrimidin-2-one
29. Flucytosine (ancobon)
30. 4-amino-5-fluoro-2-hydroxypyrimidine
31. 130256-61-0
32. 2-pyrimidinol, 4-amino-5-fluoro- (9ci)
33. Ro-2-9915
34. Chebi:5100
35. Mfcd00006035
36. Ro-29915
37. Nsc-103805
38. Mls000069463
39. Flucitosina
40. 4-amino-2-hydroxy-5-fluoropyrimidine
41. 5-fluoro-4-imino-1,2,3,4-tetrahydropyrimidin-2-one
42. Nsc103805
43. Flucytosine [usan)
44. D83282dt06
45. 4-amino-5-fluoro-1h-pyrimidin-2-one
46. Ncgc00016599-01
47. Flucitosina [dcit]
48. Smr000059047
49. Cas-2022-85-7
50. 4-amino-5-fluoro-2(1h)-pyrimidine
51. Dsstox_cid_3059
52. Flucytosine [usan]
53. Dsstox_rid_76856
54. Dsstox_gsid_23059
55. Flucytosinum [inn-latin]
56. Flucytosone
57. Ancotyl
58. Hsdb 3082
59. 6-amino-2-oxo-5-fluoropyrimidine
60. Einecs 217-968-7
61. Flucytosina
62. Flourocytosine
63. Ancoban
64. 2(1h)-pyrimidinone, 4-amino-5-fluoro-)
65. 5-fluorocytocine
66. Unii-d83282dt06
67. Flucytosine,(s)
68. 5-fluoro-cytosine
69. 5 Fluoro Cytosine
70. 4-amino-5-fluoro-2-hyroxypyrimidine
71. 6-amino-5-fluoropyrimidin-2(1h)-one
72. Flucytosine (5-fc)
73. Flucytosine [usan:usp:inn:ban:jan]
74. Cpd000059047
75. St028644
76. 6-amino-5-fluoro-3-hydropyrimidin-2-one
77. Opera_id_178
78. 5-fluorocytosine Form I
79. Flucytosine [mi]
80. Prestwick0_000934
81. Prestwick1_000934
82. Prestwick2_000934
83. Prestwick3_000934
84. Flucytosine [inn]
85. Flucytosine [jan]
86. 5-fluorocytosine Form Ii
87. F0321
88. Flucytosine [hsdb]
89. Ec 217-968-7
90. Wln: T6mvnj Dz Ef
91. Chembl1463
92. Flucytosine [mart.]
93. Schembl24063
94. Bspbio_000868
95. Flucytosine [usp-rs]
96. Flucytosine [who-dd]
97. Flucytosine [who-ip]
98. Mls000759519
99. Mls001076503
100. Mls001424013
101. Flucytosine, 5-fluorocytosine
102. Spbio_003037
103. Bpbio1_000956
104. Flucytosine (jp17/usp/inn)
105. Dtxsid3023059
106. Schembl14696800
107. 4-amino-5-fluoro-2-pyrimidinol
108. Ala-ala-phep-nitroanilide
109. Flucytosine [orange Book]
110. Flucytosine For System Suitability
111. 2-pyrimidinol,4-amino-5-fluoro-
112. 4-amino-5-fluoro-pyrimidin-2-ol
113. Flucytosine [ep Monograph]
114. Flucytosine [usp Impurity]
115. Hms1570l10
116. Hms2051m04
117. Hms2093j05
118. Hms2097l10
119. Hms2233i14
120. Hms3373l05
121. Hms3393m04
122. Hms3655g17
123. Hms3714l10
124. Pharmakon1600-01505429
125. Zinc896546
126. 5-fluorocytosine [who-ip]
127. Flucytosine [usp Monograph]
128. 5-fluorocytosine, Nucleoside Analog
129. Bcp02877
130. Flucytosine 2.0 Mg/ml In Methanol
131. Flucytosinum [who-ip Latin]
132. Hy-b0139
133. Tox21_110515
134. Bdbm50248003
135. Mfcd00179326
136. Mfcd03547958
137. Nsc759130
138. S1666
139. Stk292386
140. Akos004912683
141. Akos005063821
142. Akos015896898
143. Akos030241326
144. Tox21_110515_1
145. Bcp9000692
146. Ccg-100837
147. Ccg-213434
148. Cs-1935
149. Db01099
150. Gs-5578
151. Ks-1060
152. Nc00087
153. Nsc-759130
154. Sb57097
155. Cytosine, 5-fluoro- (6ci,7ci,8ci)
156. Ncgc00016599-02
157. Ncgc00016599-04
158. Ncgc00016599-05
159. Ac-11748
160. Bp-30254
161. Emtricitabine Impurity E [who-ip]
162. Nci60_000093
163. Sy004438
164. Bcp0726000281
165. Db-005380
166. Ab00513969
167. Am20090149
168. Ft-0601273
169. Ft-0695664
170. Sw197278-3
171. 6-amino-5-fluoro-1,2-dihydropyrimidin-2-one
172. 22p857
173. D00323
174. F-3010
175. H10295
176. Ab00444223-16
177. Ab00513969-02
178. Ab00513969_03
179. Ab00513969_04
180. Flucytosine 100 Microg/ml In Acetonitrile:water
181. 5-fluorocytosine, Vetec(tm) Reagent Grade, 99%
182. A814346
183. Q238490
184. Sr-01000721885
185. Sr-01000721885-5
186. Brd-k82143716-001-15-7
187. 4-amino-5-fluoropyrimidin-2(1h)-one [who-ip]
188. 5-fluorocytosine, Antibiotic For Culture Media Use Only
189. 6-amino-5-fluoro-2(1h)-pyrimidinone;5-fluorocytosine
190. Flucytosine, European Pharmacopoeia (ep) Reference Standard
191. Flucytosine, United States Pharmacopeia (usp) Reference Standard
192. Flucytosine, Pharmaceutical Secondary Standard; Certified Reference Material
193. Flucytosine For System Suitability, European Pharmacopoeia (ep) Reference Standard
194. 1ld
| Molecular Weight | 129.09 g/mol |
|---|---|
| Molecular Formula | C4H4FN3O |
| XLogP3 | -0.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Exact Mass | 129.03383992 g/mol |
| Monoisotopic Mass | 129.03383992 g/mol |
| Topological Polar Surface Area | 67.5 Ų |
| Heavy Atom Count | 9 |
| Formal Charge | 0 |
| Complexity | 208 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Ancobon |
| PubMed Health | Flucytosine (By mouth) |
| Drug Classes | Antifungal |
| Drug Label | Ancobon (flucytosine), an antifungal agent, is available as 250 mg and 500 mg capsules for oral administration. Each capsule also contains corn starch, lactose and talc. Gelatin capsule shells contain parabens (butyl, methyl, propyl) and sodium propi... |
| Active Ingredient | Flucytosine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 250mg; 500mg |
| Market Status | Prescription |
| Company | Valeant |
| 2 of 4 | |
|---|---|
| Drug Name | Flucytosine |
| PubMed Health | Flucytosine (By mouth) |
| Drug Classes | Antifungal |
| Drug Label | Flucytosine Capsules, USP are an antifungal agent available as 250 mg and 500 mg capsules for oral administration. Each capsule also contains corn starch, lactose monohydrate and talc. The 250 mg capsule shell contains FD and C Yellow No.6, FD and C... |
| Active Ingredient | Flucytosine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 250mg; 500mg |
| Market Status | Prescription |
| Company | Sigmapharm Labs |
| 3 of 4 | |
|---|---|
| Drug Name | Ancobon |
| PubMed Health | Flucytosine (By mouth) |
| Drug Classes | Antifungal |
| Drug Label | Ancobon (flucytosine), an antifungal agent, is available as 250 mg and 500 mg capsules for oral administration. Each capsule also contains corn starch, lactose and talc. Gelatin capsule shells contain parabens (butyl, methyl, propyl) and sodium propi... |
| Active Ingredient | Flucytosine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 250mg; 500mg |
| Market Status | Prescription |
| Company | Valeant |
| 4 of 4 | |
|---|---|
| Drug Name | Flucytosine |
| PubMed Health | Flucytosine (By mouth) |
| Drug Classes | Antifungal |
| Drug Label | Flucytosine Capsules, USP are an antifungal agent available as 250 mg and 500 mg capsules for oral administration. Each capsule also contains corn starch, lactose monohydrate and talc. The 250 mg capsule shell contains FD and C Yellow No.6, FD and C... |
| Active Ingredient | Flucytosine |
| Dosage Form | Capsule |
| Route | Oral |
| Strength | 250mg; 500mg |
| Market Status | Prescription |
| Company | Sigmapharm Labs |
Antifungal Agents; Antimetabolites
National Library of Medicine's Medical Subject Headings. Citalopram. Online file (MeSH, 2018). Available from, as of February 2, 2018: https://meshb.nlm.nih.gov/search
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Flucytosine is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of February 2, 2018: https://clinicaltrials.gov/
Flucytosine Capsules are indicated only in the treatment of serious infections caused by susceptible strains of Candida and/or Cryptococcus. Candida: Septicemia, endocarditis and urinary system infections have been effectively treated with flucytosine. Limited trials in pulmonary infections justify the use of flucytosine. Cryptococcus: Meningitis and pulmonary infections have been treated effectively. Studies in septicemias and urinary tract infections are limited, but good responses have been reported. /Included in US product label/
NIH; DailyMed. Current Medication Information for Flucytosine Capsule (Updated: January 8, 2018). Available from, as of February 8, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=58189ec7-118b-458a-8e4f-7867a97cfcbd
Flucytosine Capsules should be used in combination with amphotericin B for the treatment of systemic candidiasis and cryptococcosis because of the emergence of resistance to Flucytosine Capsules. /Included in US product label/
NIH; DailyMed. Current Medication Information for Flucytosine Capsule (Updated: January 8, 2018). Available from, as of February 8, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=58189ec7-118b-458a-8e4f-7867a97cfcbd
For more Therapeutic Uses (Complete) data for Flucytosine (10 total), please visit the HSDB record page.
/BOXED WARNING/ Use with extreme caution in patients with impaired renal function. Close monitoring of hematologic, renal and hepatic status of all patients is essential. These instructions should be thoroughly reviewed before administration of Flucytosine Capsules, USP.
NIH; DailyMed. Current Medication Information for Flucytosine Capsule (Updated: January 8, 2018). Available from, as of February 8, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=58189ec7-118b-458a-8e4f-7867a97cfcbd
Flucytosine capsules must be given with extreme caution to patients with bone marrow depression. Patients may be more prone to depression of bone marrow function if they: 1) have a hematologic disease, 2) are being treated with radiation or drugs which depress bone marrow, or 3) have a history of treatment with such drugs or radiation. Bone marrow toxicity can be irreversible and may lead to death in immunosuppressed patients. Frequent monitoring of hepatic function and of the hematopoietic system is indicated during therapy.
NIH; DailyMed. Current Medication Information for Flucytosine Capsule (Updated: January 8, 2018). Available from, as of February 8, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=58189ec7-118b-458a-8e4f-7867a97cfcbd
In addition to antiproliferative effects on the GI lining, adverse GI effects reported with flucytosine, which are sometimes severe, include anorexia, abdominal bloating, abdominal pain, diarrhea, dry mouth, duodenal ulcer, GI hemorrhage, nausea, vomiting, and ulcerative colitis.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 558
There are no adequate or controlled studies to date using flucytosine in pregnant women, and the drug should be used during pregnancy only when the potential benefits justify the possible risks to the fetus.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
For more Drug Warnings (Complete) data for Flucytosine (17 total), please visit the HSDB record page.
For the treatment (in combination with amphotericin B) of serious infections caused by susceptible strains of Candida (septicemia, endocarditis and urinary system infections) and/or Cryptococcus (meningitis and pulmonary infections).
FDA Label
Flucytosine is an antimetabolite that acts as an antifungal agent with in vitro and in vivo activity against Candida and Cryptococcus. Flucytosine enters the fungal cell via cytosine permease; thus, flucytosine is metabolized to 5-fluorouracil within fungal organisms. The 5-fluorouracil is extensively incorporated into fungal RNA and inhibits synthesis of both DNA and RNA. The result is unbalanced growth and death of the fungal organism. Antifungal synergism between Ancobon and polyene antibiotics, particularly amphotericin B, has been reported.
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
Antimetabolites
Drugs that are chemically similar to naturally occurring metabolites, but differ enough to interfere with normal metabolic pathways. (From AMA Drug Evaluations Annual, 1994, p2033) (See all compounds classified as Antimetabolites.)
D - Dermatologicals
D01 - Antifungals for dermatological use
D01A - Antifungals for topical use
D01AE - Other antifungals for topical use
D01AE21 - Flucytosine
J - Antiinfectives for systemic use
J02 - Antimycotics for systemic use
J02A - Antimycotics for systemic use
J02AX - Other antimycotics for systemic use
J02AX01 - Flucytosine
Absorption
Rapidly and virtually completely absorbed following oral administration. Bioavailability 78% to 89%.
Route of Elimination
Flucytosine is excreted via the kidneys by means of glomerular filtration without significant tubular reabsorption. A small portion of the dose is excreted in the feces.
Flucytosine is rapidly and well absorbed from the GI tract, with plasma levels peaking in 1-2 hr in animals that have received the drug for several days. The drug is widely distributed in the body, with a volume of distribution approximating the total body water. Flucytosine is minimally bound to plasma proteins. There is excellent penetration into body fluids such as the CSF, synovial fluids, and aqueous humor.
Kahn, C.M (ed.).; The Merck Veterinary Manual 10th Edition. Merck & Co. Whitehouse Station NJ. 2010, p. 2310
Flucytosine is rapidly and almost completed absorbed from the GI tract. Bioavailability is 78-89% following oral administration. Food decreases the rate, but not the extent, of absorption.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
In a limited number of neonates receiving oral flucytosine in a dosage of 25, 50, or 100 mg/kg daily for the treatment of systemic candidiasis, median peak serum concentrations after 5 days of treatment were 19.6, 27.7, and 83.9 ug/mL, respectively, and the mean time to peak concentrations was 2.5 hours. There was considerable interindividual variation in serum concentrations, which did not correlate with gestational age, and some neonates had serum flucytosine concentrations greater than 100 ug/mL.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
In patients with normal renal function, peak serum flucytosine concentrations of 30-40 mcg/mL are reached within 2 hours following a single 2-g oral dose. In other studies in patients with normal renal function receiving a 6-week regimen of oral flucytosine (150 mg/kg daily given in divided doses every 6 hours) and concomitant IV amphotericin B, mean serum concentrations of flucytosine 1-2 hours after a dose were approximately 70-80 ug/mL.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
For more Absorption, Distribution and Excretion (Complete) data for Flucytosine (8 total), please visit the HSDB record page.
Flucytosine is deaminated, possibly by gut bacteria or by the fungal targets, to 5-fluorouracil, the active metabolite.
Flucytosine may be fungistatic or fungicidal in action depending on the concentration of the drug. Two possible mechanisms of action have been identified for flucytosine. Flucytosine appears to enter fungal cells via the action of fungal-specific cytosine permease. Inside the cell, flucytosine is converted into fluorouracil (5-FU) by cytosine deaminase and then after several intermediate steps is converted into 5-fluorouridine triphosphate (FUTP). FUTP is incorporated into fungal RNA and interferes with protein synthesis. Flucytosine also appears to be converted to 5-fluorodeoxyuridine monophosphate, which noncompetitively inhibits thymidylate synthetase and interferes with DNA synthesis. Flucytosine does not appear to have antineoplastic activity.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
The aim of this study is to investigate whether fluorouracil (5-FU) could be responsible for bone-marrow depression occurring in fluorocytosine (5-FC) treated patients. Six 5-FC treated patients were included in this pilot study. Toxicity was monitored by means of thrombocyte and leucocyte counts. 5-FC and 5-FU serum levels were measured using a high-performance liquid chromatography (HPLC) assay that allows simultaneous determination of both compounds. The amounts of 5-FU in the 34 available serum samples remained below the limit of quantitation (< 0.05 mg/L), whereas 5-FC levels could be detected in all samples. Instead, low levels of the 5-FU catabolite alpha-fluoro-beta-alanine (FBAL) were detected in several of the investigated serum samples. In case of three patients thrombocyte counts remained within the normal range during 5-FC treatment, whereas one patient developed thrombocytopenia (50 x 10(9) thrombocytes/L) during therapy. Furthermore, one patient developed leucocytopenia (2.6 x 10(9) leucocytes/L) during 5-FC therapy, whereas the remaining five patients were suffering from leucocytosis prior to 5-FC therapy. In conclusion, we found nondetectable 5-FU serum concentrations (< 0.05 mg/L) in ICU patients treated with intravenous 5-FC, making it unlikely that 5-FC-associated toxicity results from 5-FU exposure in patients receiving intravenous 5-FC therapy. These findings may be explained by the fact that our patients received 5-FC intravenously instead of orally, therefore not allowing active conversion of 5-FC to 5-FU by the human intestinal microflora. /5-Fluorouracil/
PMID:11903511 Vermes A et al; Fundam Clin Pharmacol 16 (1): 39-47 (2002)
A gas chromatographic-mass spectrometric method for detecting 5-fluorouracil (5-FU) in serum at concentrations as low as 10 ng/mL was used to determine to what extent 5-FU was present in the serum of patients taking oral 5-fluorocytosine (5-FC). Preliminary studies in two patients and two healthy volunteers given an initial 2-g oral dose of 5-FC demonstrated sustained serum 5-FU levels (>100 ng/mL) during the 5 hr after ingestion of drug. Pharmaceutical preparations of 5-FC used in these studies were shown to be insignificantly contaminated with 5-FU (<0.03%), suggesting in vivo conversion of 5-FC to 5-FU had occurred. Serum samples from seven patients with cryptococcal meningitis treated with amphotericin B and 5-FC were examined for 5-FU. Five of these patients had experienced hematological or other toxicity attributed to 5-FC at some time during the course of therapy. Of 41 serum samples, 20 were observed to have 5-FU levels greater than 1,000 ng/mL in the range observed with cancer chemotherapeutic doses of 5-FU known to be associated with hematological toxicity. It is concluded that conversion of 5-FC to 5-FU occurs in humans and furthermore that 5-FU may account for some of the toxicity observed with 5-FC. /5-Fluorouracil/
PMID:742878 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC352577 Diasio RB et al; Antimicrob Agents Chemother 14 (6): 903-8 (1978)
Metabolism of 5-fluorocytosine-6-14C (5-FC) was studied in mice, rats, rabbits and dogs after oral and subcutaneous, single and repeated administration. In the urines of all species, intact 5-FC accounted for more than 90% of the total radioactivity at any time of the various treatment schedules. The average proportion of the urinary metabolites was around 5% in dogs, 3% in rabbits, 2.5% in rats, and 2% in mice of the total radioactivity. At repeated dosage, there was an increase of metabolites in mice but a decrease in rats treated subcutaneously. Neither increase nor decrease was observed in rabbits (treated orally) and dogs. Two metabolites were identified, alpha-fluoro-beta-ureido-propionic acid (FUPA) and alpha-fluoro-beta-alanine, the latter occurring mainly after oral treatment. These compounds represent probably that part of 5-FC which was deaminated to 5-fluorouracil (5-FU) or directly to 5-fluorodihydrouracil. FUPA was the only metabolite found in the urines collected from 4 out of 5 human volunteers during the first 12 h after single oral administration of 3.5 g of the radiolabelled drug. Its maximum proportion was 1.1% of the total radioactivity. No metabolites were detected in the urine neither of the 5th volunteer nor in those of 3 mycosis patients who were given the radioactive dose after they had received regular chemotherapy with unlabelled 5-FC (150 mg/kg/day) for at least 2 weeks. The sensitivity threshold of the method was 0.1-0.4% of the total radioactivity. One of the patients had developed thrombocytopenia which was probably due to 5-FC chemotherapy. The symptoms of 5-FC intolerance were in most of the examined species similar to those observed with 5-FU [9]. However, no quantitative correlation between proportion of metabolites and 5-FC toxicity is apparent except that man is the species in which both metabolism and toxicity are the lowest. It has not been proved yet that 5-FC intolerance occurring in a small percentage of patients receiving 5-FC chemotherapy (mainly leukopenia, thrombocytopenia) results in fact from conversion to 5-FU.
PMID:773604 Polak A et al; Chemotherapy 22 (3-4): 137-53 (1976)
For more Metabolism/Metabolites (Complete) data for Flucytosine (6 total), please visit the HSDB record page.
2.4 to 4.8 hours.
In a limited number of infants, the median half-life of flucytosine was 7.4 hours.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 560
The half-life of flucytosine is prolonged in patients with renal insufficiency; the average half-life in nephrectomized or anuric patients was 85 hours (range: 29.9 to 250 hours). A linear correlation was found between the elimination rate constant of flucytosine and creatinine clearance.
NIH; DailyMed. Current Medication Information for Flucytosine Capsule (Updated: January 8, 2018). Available from, as of February 8, 2018: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=58189ec7-118b-458a-8e4f-7867a97cfcbd
The elimination half-life of flucytosine has been variously reported to be 2.4-6 hours in patients with normal renal function, 6-14 hours in patients with creatinine clearances of 40 mL/minute, 12-15 hours in patients with creatinine clearances of 20 mL/minute, 21-27 hours in patients with creatinine clearances of 10 mL/minute, and 30-250 hours in patients with creatinine clearances less than 10 mL/minute. Half-lives up to 1160 hours have been reported in a few patients with creatinine clearances less than 2 mL/minute. Some clinicians have suggested that the half-life of flucytosine in hours is approximately 5 or 6 times the serum creatinine concentration in mg/dL.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 560
Although the exact mode of action is unknown, it has been proposed that flucytosine acts directly on fungal organisms by competitive inhibition of purine and pyrimidine uptake and indirectly by intracellular metabolism to 5-fluorouracil. Flucytosine enters the fungal cell via cytosine permease; thus, flucytosine is metabolized to 5-fluorouracil within fungal organisms. The 5-fluorouracil is extensively incorporated into fungal RNA and inhibits synthesis of both DNA and RNA. The result is unbalanced growth and death of the fungal organism. It also appears to be an inhibitor of fungal thymidylate synthase.
Flucytosine may be fungistatic or fungicidal in action depending on the concentration of the drug. Two possible mechanisms of action have been identified for flucytosine. Flucytosine appears to enter fungal cells via the action of fungal-specific cytosine permease. Inside the cell, flucytosine is converted into fluorouracil (5-FU) by cytosine deaminase and then after several intermediate steps is converted into 5-fluorouridine triphosphate (FUTP). FUTP is incorporated into fungal RNA and interferes with protein synthesis. Flucytosine also appears to be converted to 5-fluorodeoxyuridine monophosphate, which noncompetitively inhibits thymidylate synthetase and interferes with DNA synthesis. Flucytosine does not appear to have antineoplastic activity.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017, p. 559
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2014-10-28
Pay. Date : 2014-06-15
DMF Number : 28247
Submission : 2014-06-16
Status : Active
Type : II


GDUFA
DMF Review : Reviewed
Rev. Date : 2015-06-05
Pay. Date : 2015-02-04
DMF Number : 25094
Submission : 2011-07-14
Status : Active
Type : II

Certificate Number : R1-CEP 2016-161 - Rev 00
Issue Date : 2023-07-03
Type : Chemical
Substance Number : 766
Status : Valid

NDC Package Code : 59348-0016
Start Marketing Date : 1982-01-01
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2018-09-13
Pay. Date : 2018-07-20
DMF Number : 32090
Submission : 2017-11-10
Status : Active
Type : II
Date of Issue : 2022-04-08
Valid Till : 2024-01-11
Written Confirmation Number : WC-0290A3
Address of the Firm :


GDUFA
DMF Review : Reviewed
Rev. Date : 2022-08-24
Pay. Date : 2022-07-13
DMF Number : 31959
Submission : 2017-08-30
Status : Active
Type : II

Date of Issue : 2022-11-18
Valid Till : 2025-09-30
Written Confirmation Number : WC-0455
Address of the Firm :


GDUFA
DMF Review : Reviewed
Rev. Date : 2023-01-05
Pay. Date : 2022-11-09
DMF Number : 37631
Submission : 2022-11-30
Status : Active
Type : II

Date of Issue : 2023-07-03
Valid Till : 2025-07-02
Written Confirmation Number : WC-0213
Address of the Firm :


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 23503
Submission : 2010-01-22
Status : Active
Type : II

Certificate Number : R1-CEP 2016-019 - Rev 00
Issue Date : 2023-07-05
Type : Chemical
Substance Number : 766
Status : Valid

NDC Package Code : 66639-008
Start Marketing Date : 2009-06-30
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9259
Submission : 1991-07-25
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 15476
Submission : 2001-06-07
Status : Inactive
Type : II


Certificate Number : R1-CEP 2009-091 - Rev 01
Issue Date : 2022-01-11
Type : Chemical
Substance Number : 766
Status : Valid

Registration Number : 222MF10042
Registrant's Address : Wurmisweg 576, CH-4303, Kaiseraugst, Switzerland
Initial Date of Registration : 2010-02-04
Latest Date of Registration : --

USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others

Certificate Number : CEP 2020-423 - Rev 01
Issue Date : 2024-07-25
Type : Chemical
Substance Number : 766
Status : Valid

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Details:
USFDA approved generic version of Flucytosine Capsule (thymidylate synthetase inhibitor) indicated for treating serious infections caused by susceptible strains of Candida and/or Cryptococcus (meningitis and pulmonary infections)
Lead Product(s): Fluocytosine
Therapeutic Area: Infections and Infectious Diseases Brand Name: Ancobon-Generic
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable April 20, 2020
Lead Product(s) : Fluocytosine
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Strides receives USFDA Approval for Flucytosine Capsules
Details : USFDA approved generic version of Flucytosine Capsule (thymidylate synthetase inhibitor) indicated for treating serious infections caused by susceptible strains of Candida and/or Cryptococcus (meningitis and pulmonary infections)
Brand Name : Ancobon-Generic
Molecule Type : Small molecule
Upfront Cash : Not Applicable
April 20, 2020
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The proceeds will advance the development of DB107 which consists of two components: DB107–RRV (vocimagene amiretrorepvec) as a prodrug activator and DB107–FC (extended-release 5–fluorocytosine [5–FC]) as an oral prodrug, for high–grade glioma including glioblastoma.
Lead Product(s): Vocimagene Amiretrorepvec,Fluocytosine
Therapeutic Area: Oncology Brand Name: DB107-RRV
Study Phase: Phase IIProduct Type: Cell and Gene therapy
Sponsor: California Institute for Regenerative Medicine
Deal Size: $11.8 million Upfront Cash: Undisclosed
Deal Type: Funding April 30, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Vocimagene Amiretrorepvec,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase II
Partner/Sponsor/Collaborator : California Institute for Regenerative Medicine
Deal Size : $11.8 million
Deal Type : Funding
Details : The proceeds will advance the development of DB107 which consists of two components: DB107–RRV (vocimagene amiretrorepvec) as a prodrug activator and DB107–FC (extended-release 5–fluorocytosine [5–FC]) as an oral prodrug, for high–grade glioma ...
Brand Name : DB107-RRV
Molecule Type : Cell and Gene therapy
Upfront Cash : Undisclosed
April 30, 2024

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
DB107 (vocimagene amiretrorepvec) is a two-part cancer-selective immunotherapy comprising an investigational gene therapy treatment and an investigational small molecule, which is investigated for the treatment of newly diagnosed high grade glioma.
Lead Product(s): Vocimagene Amiretrorepvec,Fluocytosine
Therapeutic Area: Oncology Brand Name: DB107
Study Phase: Phase II/ Phase IIIProduct Type: Cell and Gene therapy
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable July 25, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Vocimagene Amiretrorepvec,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase II/ Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : DB107 (vocimagene amiretrorepvec) is a two-part cancer-selective immunotherapy comprising an investigational gene therapy treatment and an investigational small molecule, which is investigated for the treatment of newly diagnosed high grade glioma.
Brand Name : DB107
Molecule Type : Cell and Gene therapy
Upfront Cash : Not Applicable
July 25, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
TG6002 is novel oncolytic virus that has been engineered to combine multiple mechanisms of action. It has been designed to,selectively replicate within cancer cells. This is due to deletion of viral genes encoding TK and RR.
Lead Product(s): TG6002,Fluocytosine
Therapeutic Area: Oncology Brand Name: TG6002
Study Phase: Phase I/ Phase IIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : TG6002,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Positive Readouts for Transgene’s Clinical Stage Candidates Generated by its Two Innovative Plat...
Details : TG6002 is novel oncolytic virus that has been engineered to combine multiple mechanisms of action. It has been designed to,selectively replicate within cancer cells. This is due to deletion of viral genes encoding TK and RR.
Brand Name : TG6002
Molecule Type : Small molecule
Upfront Cash : Not Applicable
September 07, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
These data provide a clinical proof of concept for Transgene’s double deleted VVcopTK-RR- patented virus backbone: after IV administration, TG6002 reached the tumor, multiplied within tumor cells, and induced the local expression of its payload (the FCU1 gene).
Lead Product(s): TG6002,Fluocytosine
Therapeutic Area: Oncology Brand Name: TG6002
Study Phase: Phase I/ Phase IIProduct Type: Cell and Gene therapy
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable April 09, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : TG6002,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : These data provide a clinical proof of concept for Transgene’s double deleted VVcopTK-RR- patented virus backbone: after IV administration, TG6002 reached the tumor, multiplied within tumor cells, and induced the local expression of its payload (the FC...
Brand Name : TG6002
Molecule Type : Cell and Gene therapy
Upfront Cash : Not Applicable
April 09, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The trial aims at confirming the good tolerability of ascending doses of intravenous TG6002 and at providing the first translational data from this novel route of administration.
Lead Product(s): TG6002,Fluocytosine
Therapeutic Area: Oncology Brand Name: TG6002
Study Phase: Phase I/ Phase IIProduct Type: Large molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 08, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : TG6002,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Positive Data From a Phase 1 Trial With Transgene’s Oncolytic Virus TG6002
Details : The trial aims at confirming the good tolerability of ascending doses of intravenous TG6002 and at providing the first translational data from this novel route of administration.
Brand Name : TG6002
Molecule Type : Large molecule
Upfront Cash : Not Applicable
September 08, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Transgene has been a shareholder in Tasly BioPharmaceuticals when it received shares of Tasly Biopharmaceuticals in exchange for the sale of the Greater China rights of T101 and T601, two products respectively incorporating Transgene’s TG6002 and TG1050 patented technologies.
Lead Product(s): TG6002,Fluocytosine
Therapeutic Area: Oncology Brand Name: TG6002
Study Phase: Phase I/ Phase IIProduct Type: Large molecule
Sponsor: Tasly BioPharmaceuticals
Deal Size: $22.2 million Upfront Cash: Undisclosed
Deal Type: Financing August 04, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : TG6002,Fluocytosine
Therapeutic Area : Oncology
Highest Development Status : Phase I/ Phase II
Partner/Sponsor/Collaborator : Tasly BioPharmaceuticals
Deal Size : $22.2 million
Deal Type : Financing
Details : Transgene has been a shareholder in Tasly BioPharmaceuticals when it received shares of Tasly Biopharmaceuticals in exchange for the sale of the Greater China rights of T101 and T601, two products respectively incorporating Transgene’s TG6002 and TG105...
Brand Name : TG6002
Molecule Type : Large molecule
Upfront Cash : Undisclosed
August 04, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Flucytosine capsules are generic version of Bausch Health's Ancobon capsules. The product will be launched in June 2020.
Lead Product(s): Fluocytosine
Therapeutic Area: Infections and Infectious Diseases Brand Name: Undisclosed
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable May 04, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Fluocytosine
Therapeutic Area : Infections and Infectious Diseases
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Aurobindo Pharma Gets USFDA Nod For Flucytosine Capsules
Details : Flucytosine capsules are generic version of Bausch Health's Ancobon capsules. The product will be launched in June 2020.
Brand Name : Undisclosed
Molecule Type : Small molecule
Upfront Cash : Not Applicable
May 04, 2020

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]RLD : No
TE Code :
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 250MG
Approval Date : 2018-06-18
Application Number : 207536
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
RLD : No
TE Code :
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 500MG
Approval Date : 2018-06-18
Application Number : 207536
RX/OTC/DISCN : DISCN
RLD : No
TE Code :
RLD : No
TE Code : AB
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 250MG
Approval Date : 2020-04-17
Application Number : 212632
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
RLD : No
TE Code : AB
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 500MG
Approval Date : 2020-04-17
Application Number : 212632
RX/OTC/DISCN : RX
RLD : No
TE Code : AB
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 250MG
Approval Date : 2020-05-01
Application Number : 213665
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 500MG
Approval Date : 2020-05-01
Application Number : 213665
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code : AB
Brand Name : ANCOBON
Dosage Form : CAPSULE;ORAL
Dosage Strength : 250MG
Approval Date : 1982-01-01
Application Number : 17001
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : Yes
TE Code : AB
Brand Name : ANCOBON
Dosage Form : CAPSULE;ORAL
Dosage Strength : 500MG
Approval Date : 1982-01-01
Application Number : 17001
RX/OTC/DISCN : RX
RLD : Yes
TE Code : AB

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code :
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 500MG
Approval Date : 2017-10-17
Application Number : 206550
RX/OTC/DISCN : DISCN
RLD : No
TE Code :

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
RLD : No
TE Code : AB
Brand Name : FLUCYTOSINE
Dosage Form : CAPSULE;ORAL
Dosage Strength : 250MG
Approval Date : 2011-06-28
Application Number : 201566
RX/OTC/DISCN : RX
RLD : No
TE Code : AB

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info :
Registration Country : Italy
Brand Name : Ancotil
Dosage Form : Flucytosine 2.500Mg 250Ml 5 Units Parenteral Use
Dosage Strength : 5 bottles EV 2.5 g 250 ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Regulatory Info :
Registration Country : Sweden
Brand Name : Ancotil
Dosage Form : SOLUTION FOR INFUSION
Dosage Strength : 10 MG / ML
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Sweden

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results] Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Generic
Registration Country : South Africa
Brand Name : Flucytosine Mylan 500mg
Dosage Form : TAB
Dosage Strength : 500mg
Packaging : 100X1mg
Approval Date :
Application Number :
Regulatory Info : Generic
Registration Country : South Africa

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
