
Synopsis
Synopsis
0
CEP/COS
0
EU WC
0
KDMF
0
NDC API
0
Europe

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
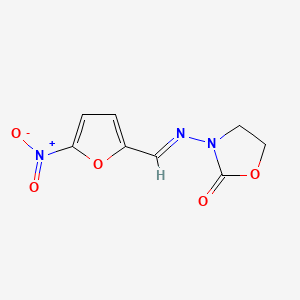

1. Furazol
2. Furazolidine
3. Furoxone
4. Nifurazolidone
1. Furoxone
2. Nitrofurazolidone
3. 67-45-8
4. Furazolidine
5. Nitrofuroxon
6. Furazolidon
7. Nifurazolidone
8. Diafuron
9. Furaxone
10. Furazol
11. Furazon
12. Furidon
13. Nitrofurazolidonum
14. Furozolidine
15. Coryzium
16. Furoxane
17. Nifulidone
18. Roptazol
19. Tikofuran
20. Topazone
21. Trichofuron
22. Tricofuron
23. Tricoron
24. Trifurox
25. Furall
26. Furaxon
27. Furoxal
28. Furoxon
29. Giardil
30. Giarlam
31. Medaron
32. Neftin
33. Nicolen
34. Optazol
35. Ortazol
36. Puradin
37. Corizium
38. Enterotoxon
39. Sclaventerol
40. Viofuragyn
41. Bifuron
42. Furovag
43. Nifuran
44. Furox
45. Furoxone Swine Mix
46. Usaf Ea-1
47. 3-(5'-nitrofurfuralamino)-2-oxazolidone
48. N-(5-nitro-2-furfurylidene)-3-amino-2-oxazolidone
49. Furazolidona
50. Furazolidonum
51. 3-[(5-nitrofurfurylidene)amino]-2-oxazolidinone
52. 2-oxazolidinone, 3-[[(5-nitro-2-furanyl)methylene]amino]-
53. 5-nitro-n-(2-oxo-3-oxazolidinyl)-2-furanmethanimine
54. N-(5-nitro-2-furfurylidene)-3-aminooxazolidine-2-one
55. 3-((5-nitrofurfurylidene)amino)-2-oxazolidinone
56. 3-((5-nitrofurylidene)amino)-2-oxazolidone
57. 3-((5-nitrofurfurylidene)amino)-2-oxazolidone
58. 3-[(e)-(5-nitrofuran-2-yl)methylideneamino]-1,3-oxazolidin-2-one
59. 3-[(5-nitrofurylidene)amino]-2-oxazolidone
60. 3-(((5-nitro-2-furanyl)methylene)amino)-2-oxazolidinone
61. 3-[(5-nitrofurfurylidene)amino]-2-oxazolidone
62. Nf 180 Custom Mix Ten
63. 3-[[(5-nitro-2-furanyl)methylene]amino]-2-oxazolidinone
64. Nsc-6469
65. 2-furanmethanimine, 5-nitro-n-(2-oxo-3-oxazolidinyl)-
66. Mls000069727
67. 3-{[(5-nitro-2-furanyl)methylene]amino}-2-oxazolidinone
68. 2-oxazolidinone, 3-(((5-nitro-2-furanyl)methylene)amino)-
69. 3-{[(5-nitrofuran-2-yl)methylidene]amino}-1,3-oxazolidin-2-one
70. Nsc6469
71. 2-oxazolidinone, 3-((5-nitrofurfurylidine)amino)-
72. Nf 180
73. Ncgc00095304-02
74. Smr000058228
75. Dsstox_cid_21997
76. Dsstox_rid_79899
77. Dsstox_gsid_41997
78. 2-oxazolidinone, 3-((5-nitrofurfurylidene)amino)-
79. Fiurox Aerosol Powder
80. 3-(((5-nitrofuran-2-yl)methylene)amino)oxazolidin-2-one
81. Furoxone (tn)
82. 3-((5-nitrofurfurylidine)amino)-2-oxazolidinone
83. 3-[(5-nitrofurfurylidine)amino]-2-oxazolidinone
84. 5j9cpu3re0
85. Sr-05000001632
86. 2-oxazolidinone, 3-[(5-nitrofurfurylidene)amino]-
87. 2-oxazolidinone, 3-[(5-nitrofurfurylidine)amino]-
88. Furazolidone (usp/inn)
89. Chebi:5195
90. Cas-67-45-8
91. Fzl
92. Mfcd00010550
93. Component Of Topazone
94. Component Of Tricofuron
95. Opera_id_256
96. Prestwick2_000808
97. Prestwick3_000808
98. Spectrum5_000742
99. Furazolidone [mi]
100. Furazolidone [inn]
101. F-8900
102. Furazolidone [hsdb]
103. Furazolidone [iarc]
104. Chembl1103
105. Schembl64996
106. Bspbio_000675
107. Bspbio_002052
108. Furazolidone [mart.]
109. Mls001304180
110. Bidd:gt0182
111. Furazolidone [usp-rs]
112. Furazolidone [who-dd]
113. Spectrum1500309
114. Bpbio1_000743
115. Hms500e07
116. 3-[(e)-(5-nitro-2-furyl)methyleneamino]oxazolidin-2-one
117. 3-[[(5-nitro-2-furanyl)methylene]-amino]-2-oxazolidinone
118. Furazolidone [green Book]
119. 3-{[(5-nitro-2-furyl)methylene]amino}-1,3-oxazolidin-2-one
120. Furazolidone [orange Book]
121. Hms1920o22
122. Hms2091h03
123. Pharmakon1600-01500309
124. Furazolidone [usp Impurity]
125. Hy-b1336
126. Zinc7997571
127. Furazolidone [usp Monograph]
128. Tox21_111502
129. 3-{[(e)-(5-nitrofuran-2-yl)methylidene]amino}-1,3-oxazolidin-2-one
130. Ccg-40233
131. Nsc757038
132. S4528
133. Stk388219
134. Akos001678302
135. Furazolidone, Vetec(tm) Reagent Grade
136. Tox21_111502_1
137. Ccg-266758
138. Cs-4897
139. Db00614
140. Nsc-757038
141. Furazolidone 100 Microg/ml In Methanol
142. Idi1_000085
143. Wln: T5oj Bnw E1un- At5nvotj
144. Ncgc00095304-01
145. Ncgc00095304-03
146. Ncgc00095304-04
147. Ncgc00095304-05
148. Ncgc00095304-06
149. Ncgc00188974-01
150. Ac-11695
151. Ac-32486
152. Sbi-0051388.p003
153. Ab00443658
154. A16011
155. C07999
156. D00830
157. Ab00052000_03
158. Furazolidone, Vetranal(tm), Analytical Standard
159. A835757
160. Furazolidone, Antibiotic For Culture Media Use Only
161. Sr-05000001632-1
162. Sr-05000001632-3
163. Brd-k10647544-001-02-0
164. Brd-k11756522-001-03-2
165. Brd-k11756522-001-04-0
166. 3-[(e)-(5-nitro-2-furanyl)methylideneamino]-2-oxazolidinone
167. 3-[[(5-nitro-2-furanyl)methylene]amino]-2-oxazolidinone, 9ci
168. 3-([(5-nitro-2-furyl)methylidene]amino)-1,3-oxazolidin-2-one #
169. 3-{[(1e)-(5-nitro-2-furyl)methylene]amino}-1,3-oxazolidin-2-one
170. Furazolidone, United States Pharmacopeia (usp) Reference Standard
| Molecular Weight | 225.16 g/mol |
|---|---|
| Molecular Formula | C8H7N3O5 |
| XLogP3 | -0.1 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Exact Mass | 225.03857033 g/mol |
| Monoisotopic Mass | 225.03857033 g/mol |
| Topological Polar Surface Area | 101 Ų |
| Heavy Atom Count | 16 |
| Formal Charge | 0 |
| Complexity | 326 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 1 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Furazolidone is used in the treatment of cholera when anti-infective therapy is indicated as an adjunct to fluid and electrolyte replacement.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 861
Furazolidone is used for the specific and symptomatic treatment of diarrhea and enteritis caused by susceptible bacteria or protozoa.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 861
Furazolidone ... often is prescribed /to treat giardiasis in/ children because the drug is available in a pleasant liquid formulation. ... Furazolidone is the only drug currently approved by the US FDA for treatment of giardiasis.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 1098
After use of furazolidone as a drug, acute nausea, emesis, occasional diarrhea, abdominal pain and intestinal bleeding were observed; hepatic damage, as evidenced by biochemical tests, and peripheral neuropathy were also seen.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V31 146 (1983)
Nausea and vomiting are the most common side effects of oral furazolidone therapy; abdominal pain and diarrhea occasionally occur. These effects can be minimized or eliminated by reducing dosage or discontinuing the drug.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 861
Hypersensitivity reactions to oral furazolidone have occurred in a small number of patients and generally subside with discontinuance of the drug. Hypersensitivity reactions include a fall in blood pressure, angioedema, fever, arthralgia, urticaria, and a vesicular or morbilliform rash. Erythema multiforme, pulmonary infiltration, and pulmonary eosinophilia also have been reported and may be due to hypersensitivity.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 861
Headache and malaise occur occasionally with oral furazolidone therapy and can be minimized or eliminated by reducing dosage or discontinuing the drug. Following oral furazolidone administration, hypoglycemia, agranulocytosis, and, in one patient, partial deafness and dizziness have also been reported. Rarely, some patients receiving oral furazolidone experience a disulfiram-like reaction to alcohol. Polyneuritis and hemolytic anemia (in patients with glucose-6-phosphate dehydrogenase deficiency and in neonates) also have been reported rarely.
McEvoy, G.K. (ed.). American Hospital Formulary Service- Drug Information 2002. Bethesda, MD: American Society of Health-System Pharmacists, Inc. 2002 (Plus Supplements)., p. 861
For more Drug Warnings (Complete) data for FURAZOLIDONE (6 total), please visit the HSDB record page.
For the specific and symptomatic treatment of bacterial or protozoal diarrhea and enteritis caused by susceptible organisms.
Furoxone has a broad antibacterial spectrum covering the majority of gastrointestinal tract pathogens including E. coli, staphylococci, Salmonella, Shigella, Proteus, Aerobacter aerogenes, Vibrio cholerae and Giardia lamblia. Its bactericidal activity is based upon its interference with DNA replication and protein production; this antimicrobial action minimizes the development of resistant organisms.
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
Antitrichomonal Agents
Agents used to treat trichomonas infections. (See all compounds classified as Antitrichomonal Agents.)
Monoamine Oxidase Inhibitors
A chemically heterogeneous group of drugs that have in common the ability to block oxidative deamination of naturally occurring monoamines. (From Gilman, et al., Goodman and Gilman's The Pharmacological Basis of Therapeutics, 8th ed, p414) (See all compounds classified as Monoamine Oxidase Inhibitors.)
Anti-Infective Agents, Urinary
Substances capable of killing agents causing urinary tract infections or of preventing them from spreading. (See all compounds classified as Anti-Infective Agents, Urinary.)
G - Genito urinary system and sex hormones
G01 - Gynecological antiinfectives and antiseptics
G01A - Antiinfectives and antiseptics, excl. combinations with corticosteroids
G01AX - Other antiinfectives and antiseptics
G01AX06 - Furazolidone
Absorption
Radiolabeled drug studies indicate that furazolidone is well absorbed following oral administration
After a single oral dose of 100 mg/kg body weight to rats, only 3% of the dose was recovered in the feces as unmetabolized compound.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V31 145 (1983)
Furazolidone is rapidly and extensively metabolized; the primary metabolic pathway identified begins with nitro-reduction to the aminofuran derivative. Two major metabolites are produced: 3-amino-2-oxazolidone (AOZ) or beta-hydroxyethylhydrazine (HEH). AOZ is responsible for monoamine oxidase inhibition. Detoxification and elimination of the drug is done primarily by conjugation with glutathione.
In vitro metabolism of furazolidone by milk xanthine oxidase and rat liver homogenate yielded approximately equal amounts (30%) of 2,3-dihydro-3-cyano-methyl-2-hydroxyl-5-nitro-1a,2-di(2-oxo-oxazolidine-3-yl)imi nomethylfuro(2,3b)furan, and 3-(4-cyano-2-oxobutyl-lideneamino)-2-oxazolidone. The latter was also isolated from the urine of rabbits given on oral dose of furazolidone.
IARC. Monographs on the Evaluation of the Carcinogenic Risk of Chemicals to Humans. Geneva: World Health Organization, International Agency for Research on Cancer, 1972-PRESENT. (Multivolume work). Available at: https://monographs.iarc.fr/ENG/Classification/index.php, p. V31 146 (1983)
Furazolidone is an antimicrobial compound used in human and veterinary medicine. The aim of this investigation was to determine its genotoxic capacity in vitro and in vivo. We used the human lymphocyte culture system to detect the effect of 2.0, 4.0, 6.0, 8.0, or 10.0 ug/ml, and the mouse bone marrow assay to determine the effect of 8.6, 30.0, or 75.0 mg/kg furazolidone. In both systems we determined the frequency of sister-chromatid exchanges (SCE), the cell proliferation kinetics (CPK), and the mitotic index (MI). The in vitro results showed a significant SCE increase starting from the second dose tested and a CPK and MI decrease starting from the third dose. The in vivo results showed a SCE increase with the two high doses tested, but no significant modification was found in the CPK and MI with the three doses tested in the experiment.
PMID:9169063 Madrigal-Bujaidar E et al; J Toxicol Environ Health 51 (1): 89-96 (1997)
In vitro metabolism of furazolidone (N-(5-nitro-2-furfuryliden)-3-amino-2-oxazolidone) was investigated by using milk xanthine oxidase and rat liver 9000 g supernatant. A new type of reduction product was isolated as 1 of the main metabolites from the incubation mixture and it was tentatively identified as 2,3-dihydro-3-cyanomethyl-2-hydroxyl-5-nitro-1a,2-di(2-oxo-oxazolidin-3-yl)imino methyl-furo(2,3-b)furan. The formation of N-(5-amino-2-furfurylidene)-3-amino-2-oxazolidone as a minor metabolite of nitrofuran in a milk xanthine oxidase system was demonstrated. The aminofuran derivative was easily degraded by milk xanthine oxidase under aerobic, but not anaerobic, conditions. The degradation appears to be due to superoxide anion radicals, hydroxy radicals and/or singlet O2, which are produced in this enzyme system. (Furazolidone, an antibacterial nitrofuran widely used as a veterinary medicine, was mutagenic in Escherichia coli WP2 and Salmonella typhimurium TA100 and tumorigenic in rats).
PMID:6894838 Tatsumi K et al; Arch Biochem Biophys 208 (1): 167-174 (1981)
10 minutes
Furazolidone and its related free radical products are believed to bind DNA and induce cross-links. Bacterial DNA is particularly susceptible to this drug leading to high levels of mutations (transitions and transversions) in the bacterial chromosome.
We are studying the development of unilateral malformations. Several chemicals elicit right sided limb defects both in vivo and in vitro. For example, nitroheterocyclics such as furazolidone (FZ) induce asymmetric defects in rat embryos in vitro. Potential mechanisms include asymmetric drug delivery; intrinsic difference between the cells of the left and right limbs; physiological asymmetry (e.g. in tissue oxygen), secondary to another primary asymmetry (e.g. in limb vasculature). In one series of experiments, we have investigated the role of an asymmetry in drug delivery and/or tissue oxygen. 10.3 days embryonic age rat embryos were explanted and cultured for two hours to allow 'recovery'. Embryos were then exteriorised (removal from yolk sac and amnion, retaining intact circulation) and cultured in the presence of 20 uM FZ with 5% O2 for 24h. The assumption was that exteriorisation would abolish any drug delivery or tissue oxygen asymmetry, by direct embryonic exposure to the medium. As previously described, FZ induced right sided defects (limb, eye and fore-brain) in 42% of intact yolk sac embryos. In contrast, exposure of exteriorised embryos to FZ induced a 34% incidence of abnormalities which were identical, but exclusively left sided. The results do not support a simple hypothesis that asymmetric drug delivery or tissue oxygen levels are responsible for the unilateral defects. We are investigating two potential explanations: a) Exteriorisation specifically reverses limb bud susceptibility, b) A secondary asymmetry e.g. mitochondrial maturity is altered by exteriorisation which then acts to invert the limb response. In a second series of experiments, we have also investigated a possible primary asymmetry in the development of the limb vasculature, using whole mount in situ hybridisation with a probe specific for early endothelial cell precursors (flk-1). The results are being analysed using confocal microscopy to image the limb vascular architecture.
Narburgh LJ, Brown NA; Teratology 50 (5): 24A (1994)
Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
27
PharmaCompass offers a list of Furazolidone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Furazolidone manufacturer or Furazolidone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Furazolidone manufacturer or Furazolidone supplier.
PharmaCompass also assists you with knowing the Furazolidone API Price utilized in the formulation of products. Furazolidone API Price is not always fixed or binding as the Furazolidone Price is obtained through a variety of data sources. The Furazolidone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Furazolidone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Furazolidone, including repackagers and relabelers. The FDA regulates Furazolidone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Furazolidone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Furazolidone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Furazolidone supplier is an individual or a company that provides Furazolidone active pharmaceutical ingredient (API) or Furazolidone finished formulations upon request. The Furazolidone suppliers may include Furazolidone API manufacturers, exporters, distributors and traders.
click here to find a list of Furazolidone suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Furazolidone DMF (Drug Master File) is a document detailing the whole manufacturing process of Furazolidone active pharmaceutical ingredient (API) in detail. Different forms of Furazolidone DMFs exist exist since differing nations have different regulations, such as Furazolidone USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Furazolidone DMF submitted to regulatory agencies in the US is known as a USDMF. Furazolidone USDMF includes data on Furazolidone's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Furazolidone USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Furazolidone suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Furazolidone Drug Master File in Japan (Furazolidone JDMF) empowers Furazolidone API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Furazolidone JDMF during the approval evaluation for pharmaceutical products. At the time of Furazolidone JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Furazolidone suppliers with JDMF on PharmaCompass.
Furazolidone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Furazolidone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Furazolidone GMP manufacturer or Furazolidone GMP API supplier for your needs.
A Furazolidone CoA (Certificate of Analysis) is a formal document that attests to Furazolidone's compliance with Furazolidone specifications and serves as a tool for batch-level quality control.
Furazolidone CoA mostly includes findings from lab analyses of a specific batch. For each Furazolidone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Furazolidone may be tested according to a variety of international standards, such as European Pharmacopoeia (Furazolidone EP), Furazolidone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Furazolidone USP).
