
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
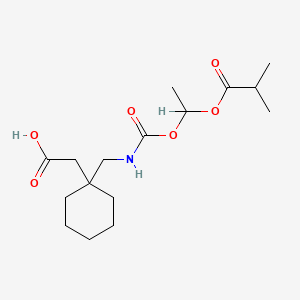

1. 1-(((alpha-isobutanoyloxyethoxy)carbonyl)aminomethyl)-1-cyclohexaneacetic Acid
2. Xp 10569
3. Xp 11084
4. Xp 11239
5. Xp 12464
6. Xp 13512
7. Xp-10569
8. Xp-11084
9. Xp-11239
10. Xp-12464
11. Xp-13512
12. Xp10569
13. Xp11084
14. Xp11239
15. Xp12464
16. Xp13512
1. 478296-72-9
2. Horizant
3. Regnite
4. Xp-13512
5. Solzira
6. Xp13512
7. Asp8825
8. Xp 13512
9. Gsk1838262
10. Asp-8825
11. 75ocl1spbq
12. Gsk-1838262
13. Chebi:68840
14. 2-(1-((((1-(isobutyryloxy)ethoxy)carbonyl)amino)methyl)cyclohexyl)acetic Acid
15. 2-[1-[[1-(2-methylpropanoyloxy)ethoxycarbonylamino]methyl]cyclohexyl]acetic Acid
16. {1-[({[1-(isobutyryloxy)ethoxy]carbonyl}amino)methyl]cyclohexyl}acetic Acid
17. (1-(((((1rs)-1-((2-methylpropanoyl)oxy)ethoxy)carbonyl)amino)methyl)cyclohexyl)acetic Acid
18. (1-{[({(1rs)-1-[isobutyryloxy]ethoxy}carbonyl) Amino]methyl}cyclohexyl)acetic Acid
19. Cyclohexaneacetic Acid, 1-((((1-(2-methyl-1-oxopropoxy)ethoxy)carbonyl)amino)methyl)-
20. Gabapentin Encarbil
21. Unii-75ocl1spbq
22. Gsk 1838262
23. Gabapentin Enacarbil [usan:inn]
24. Gabapentin-encarbil
25. Horizant (tn)
26. Asp 8825
27. Gabapentine Enacarbil
28. Gabapentina Enacarbilo
29. Gabapentinum Enacarbilum
30. Schembl25455
31. Gtpl7560
32. Chembl1628502
33. Gabapentin Encarbil [mi]
34. Dtxsid30870359
35. Gabapentin Enacarbil [inn]
36. Gabapentin Enacarbil [jan]
37. Gabapentin Enacarbil [usan]
38. Bcp12845
39. Gabapentin Enacarbil [vandf]
40. Gabapentin Enacarbil [mart.]
41. Mfcd09954124
42. Xp-053
43. Gabapentin Enacarbil [who-dd]
44. Akos025290727
45. Gabapentin Enacarbil (jan/usan/inn)
46. Cs-1698
47. Db08872
48. Pb32822
49. 1-[[[[1-(2-methyl-1-oxopropoxy)ethoxy]carbonyl]amino]methyl]cyclohexaneacetic Acid
50. Ncgc00532500-01
51. Ac-27644
52. As-35192
53. Gabapentin Enacarbil [orange Book]
54. Hy-16216
55. Db-070861
56. Ft-0753836
57. A20372
58. D09539
59. 296g729
60. Q5515257
61. {[(1-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexane Acetic Acid
62. {[(alpha-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexane Acetic Acid
63. 1-{[(alpha-isobutanovloxyethoxy)carbonyl]aminomethyl}-1-cyclohexane Acetic Acid
64. 1-{[(alpha-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexane Acetic Acid
65. 1-{[(alpha-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexaneacetic Acid
66. 2-[1-[[[1-[(2-methylpropanoyl)oxy]ethoxy]carbonylamino]methyl]cyclohexyl]acetic Acid
| Molecular Weight | 329.39 g/mol |
|---|---|
| Molecular Formula | C16H27NO6 |
| XLogP3 | 3.1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 9 |
| Exact Mass | 329.18383758 g/mol |
| Monoisotopic Mass | 329.18383758 g/mol |
| Topological Polar Surface Area | 102 Ų |
| Heavy Atom Count | 23 |
| Formal Charge | 0 |
| Complexity | 428 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 2 | |
|---|---|
| Drug Name | Horizant |
| Drug Label | HORIZANT (gabapentin enacarbil) is a prodrug of gabapentin. Gabapentin enacarbil is described as (1-{[({(1RS)-1-[(2-Methylpropanoyl)oxy]ethoxy}carbonyl)amino]methyl} cyclohexyl) acetic acid. It has a molecular formula of C16H27NO6 and a molecular wei... |
| Active Ingredient | Gabapentin enacarbil |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 600mg; 300mg |
| Market Status | Prescription |
| Company | Xenoport |
| 2 of 2 | |
|---|---|
| Drug Name | Horizant |
| Drug Label | HORIZANT (gabapentin enacarbil) is a prodrug of gabapentin. Gabapentin enacarbil is described as (1-{[({(1RS)-1-[(2-Methylpropanoyl)oxy]ethoxy}carbonyl)amino]methyl} cyclohexyl) acetic acid. It has a molecular formula of C16H27NO6 and a molecular wei... |
| Active Ingredient | Gabapentin enacarbil |
| Dosage Form | Tablet, extended release |
| Route | Oral |
| Strength | 600mg; 300mg |
| Market Status | Prescription |
| Company | Xenoport |
For the treatment of adult Restless Legs Syndrome (RLS) and postherpetic neuralgia (PHN).
FDA Label
Since gabapentin enacarbil is a prodrug of gabapentin, it's physiological effects are the same as gabapentin. Concerning PHN, gabapentin prevents allodynia and hyperalgesia.
Absorption
Gabapentin enacarbil is absorbed in the intestines by active transport through the proton-linked monocarboxylate transporter, MCT-1.
Route of Elimination
Gabapentin enacarbil is eliminated primarily in the urine (94%) and to a lesser extent in the feces (5%).
Volume of Distribution
The volume of distribution is 76L.
Clearance
Renal clearance of gabapentin is 5 to 7 L/hr.
Gabapentin enacarbil does not interact with any of the major cytochrome P450 enzymes.
The elimination half-life of gabapentin is 5.1 to 6.0 hours.
Although the exact mechanism of action of gabapentin in RLS and PHN is unknown, it is presumed to involve the descending noradrenergic system, resulting in the activation of spinal alpha2-adrenergic receptors.

GDUFA
DMF Review : Reviewed
Rev. Date : 2023-12-20
Pay. Date : 2023-11-29
DMF Number : 39197
Submission : 2023-12-01
Status : Active
Type : II

NDC Package Code : 59651-882
Start Marketing Date : 2023-11-29
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (50kg/50kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2019-05-10
Pay. Date : 2019-02-21
DMF Number : 33386
Submission : 2019-03-29
Status : Active
Type : II

NDC Package Code : 70966-0011
Start Marketing Date : 2011-12-13
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : Reviewed
Rev. Date : 2018-11-21
Pay. Date : 2018-11-02
DMF Number : 33296
Submission : 2018-11-02
Status : Active
Type : II

Date of Issue : 2023-12-28
Valid Till : 2026-12-28
Written Confirmation Number : WC-0310
Address of the Firm :

NDC Package Code : 66039-945
Start Marketing Date : 2018-11-02
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41053
Submission : 2024-12-30
Status : Active
Type : II



Date of Issue : 2022-04-04
Valid Till : 2025-01-13
Written Confirmation Number : WC-0331
Address of the Firm :



 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : Complete
Rev. Date : 2018-11-21
Pay. Date : 2018-11-02
DMF Number : 33296
Submission : 2018-11-02
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2023-12-20
Pay. Date : 2023-11-29
DMF Number : 39197
Submission : 2023-12-01
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2019-05-10
Pay. Date : 2019-02-21
DMF Number : 33386
Submission : 2019-03-29
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 41053
Submission : 2024-12-30
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]About the Company : Atman Pharma Private Limited is a fully integrated pharmaceutical company that has distinguished itself as a leader in Bulk Drugs (API) marketing both domestically in India and ove...
About the Company : Founded in 1986 by Mr. P.V. Ramaprasad Reddy, Mr. K. Nityananda Reddy and a small group of highly committed professionals, Aurobindo Pharma was born off a vision. The company comme...

About the Company : Dasami Lab Private Limited incorporated with MCA on 24 July 2015. The Dasami Lab Private Limited is listed in the class of pvtltd company and classified as Non Govt Company. This c...

About the Company : Glenmark was founded with a vision to emerge as a leading integrated research-based, global pharmaceutical company. Our branded generics business has a significant presence in mark...

About the Company : Hetero is a research based global pharmaceutical company focused on development, manufacturing and marketing of Active Pharmaceutical Ingredients (APIs), Intermediate Chemicals & F...

About the Company : Maithri Drugs is one of India's fast-growing pharmaceutical companies. Maithri's strategic focus is on active pharma ingredients (APIs). The company is widely recognized for its ex...

About the Company : Rakshit is an integrated API manufacturing company. Established in 2000. Today Rakshit has emerged a dependable and strong manufacturer of key APIs & intermediates for global marke...

About the Company : Zhejiang Bamboo Pharmaceuticals is a new, innovative, green, high-grade pharmaceutical intermediate, API and oral solid formulation pharmaceutical manufacturer which was establishe...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
0.0
<10
0.0
10,864.3
API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|---|---|---|
| ISRAEL | 0.00 | 0.0 | <10 |
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
34
PharmaCompass offers a list of Gabapentin Enacarbil API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Gabapentin Enacarbil manufacturer or Gabapentin Enacarbil supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Gabapentin Enacarbil manufacturer or Gabapentin Enacarbil supplier.
PharmaCompass also assists you with knowing the Gabapentin Enacarbil API Price utilized in the formulation of products. Gabapentin Enacarbil API Price is not always fixed or binding as the Gabapentin Enacarbil Price is obtained through a variety of data sources. The Gabapentin Enacarbil Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Gabapentin Enacarbil manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Gabapentin Enacarbil, including repackagers and relabelers. The FDA regulates Gabapentin Enacarbil manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Gabapentin Enacarbil API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Gabapentin Enacarbil manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Gabapentin Enacarbil supplier is an individual or a company that provides Gabapentin Enacarbil active pharmaceutical ingredient (API) or Gabapentin Enacarbil finished formulations upon request. The Gabapentin Enacarbil suppliers may include Gabapentin Enacarbil API manufacturers, exporters, distributors and traders.
click here to find a list of Gabapentin Enacarbil suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Gabapentin Enacarbil DMF (Drug Master File) is a document detailing the whole manufacturing process of Gabapentin Enacarbil active pharmaceutical ingredient (API) in detail. Different forms of Gabapentin Enacarbil DMFs exist exist since differing nations have different regulations, such as Gabapentin Enacarbil USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Gabapentin Enacarbil DMF submitted to regulatory agencies in the US is known as a USDMF. Gabapentin Enacarbil USDMF includes data on Gabapentin Enacarbil's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Gabapentin Enacarbil USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Gabapentin Enacarbil suppliers with USDMF on PharmaCompass.
A Gabapentin Enacarbil written confirmation (Gabapentin Enacarbil WC) is an official document issued by a regulatory agency to a Gabapentin Enacarbil manufacturer, verifying that the manufacturing facility of a Gabapentin Enacarbil active pharmaceutical ingredient (API) adheres to the Good Manufacturing Practices (GMP) regulations of the importing country. When exporting Gabapentin Enacarbil APIs or Gabapentin Enacarbil finished pharmaceutical products to another nation, regulatory agencies frequently require a Gabapentin Enacarbil WC (written confirmation) as part of the regulatory process.
click here to find a list of Gabapentin Enacarbil suppliers with Written Confirmation (WC) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Gabapentin Enacarbil as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Gabapentin Enacarbil API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Gabapentin Enacarbil as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Gabapentin Enacarbil and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Gabapentin Enacarbil NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Gabapentin Enacarbil suppliers with NDC on PharmaCompass.
Gabapentin Enacarbil Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Gabapentin Enacarbil GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Gabapentin Enacarbil GMP manufacturer or Gabapentin Enacarbil GMP API supplier for your needs.
A Gabapentin Enacarbil CoA (Certificate of Analysis) is a formal document that attests to Gabapentin Enacarbil's compliance with Gabapentin Enacarbil specifications and serves as a tool for batch-level quality control.
Gabapentin Enacarbil CoA mostly includes findings from lab analyses of a specific batch. For each Gabapentin Enacarbil CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Gabapentin Enacarbil may be tested according to a variety of international standards, such as European Pharmacopoeia (Gabapentin Enacarbil EP), Gabapentin Enacarbil JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Gabapentin Enacarbil USP).
