
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
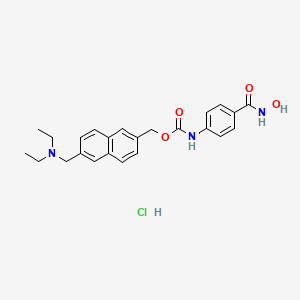

1. Itf 2357
2. Itf2357
1. 199657-29-9
2. Givinostat Hcl
3. Givinostat (hydrochloride)
4. Itf 2357
5. Z02132r2qq
6. Unii-z02132r2qq
7. Itf2357 Hydrochloride
8. Itf-2357 Hydrochloride
9. Histone Deacetylase Inhibitor
10. Itf2357
11. Itf-2357
12. Carbamic Acid, N-[4-[(hydroxyamino)carbonyl]phenyl]-, [6-[(diethylamino)methyl]-2-naphthalenyl]methyl Ester, Hydrochloride (1:1)
13. [6-(diethylaminomethyl)naphthalen-2-yl]methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate;hydrochloride
14. Carbamic Acid, N-(4-((hydroxyamino)carbonyl)phenyl)-, (6-((diethylamino)methyl)-2-naphthalenyl)methyl Ester, Hydrochloride (1:1)
15. (6-((diethylamino)methyl)naphthalen-2-yl)methyl (4-(hydroxycarbamoyl)phenyl)carbamate Hydrochloride
16. Mfcd28502062
17. {6-[(diethylamino)methyl]naphthalen-2-yl}methyl [4-(hydroxycarbamoyl)phenyl]carbamate Hydrochloride
18. Itf 2357 Hydrochloride
19. Givinostat Monohydrochloride
20. Schembl23529420
21. Givinostat Hydrochloride Anhydrous
22. Chebi:231333
23. Ex-a8024
24. Zha65729
25. Hy-14842a
26. Akos030526700
27. Cs-4728
28. Sb16937
29. As-52365
30. En300-257964
31. P14959
32. Q27294820
33. Z2327390266
34. (6-((diethylamino)methyl)naphthalen-2-yl)methyl 4-(hydroxycarbamoyl)phenylcarbamate Hydrochloride
35. [6-[(diethylamino)methyl]-2-naphthalenyl]methyl N-[4-[(hydroxyamino)carbonyl]phenyl]carbamate Hydrochloride (1:1)
36. {6-[(diethylamino)methyl]naphthalen-2-yl}methyl [4-(hydroxycarbamoyl)phenyl]carbamate--hydrogen Chloride
37. {6-[(diethylamino)methyl]naphthalen-2-yl}methyl N-[4-(hydroxycarbamoyl)phenyl]carbamate Hydrochloride
1. Givinostat
| Molecular Weight | 457.9 g/mol |
|---|---|
| Molecular Formula | C24H28ClN3O4 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 9 |
| Exact Mass | Da |
| Monoisotopic Mass | Da |
| Topological Polar Surface Area | 90.9 |
| Heavy Atom Count | 32 |
| Formal Charge | 0 |
| Complexity | 575 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Histone Deacetylase Inhibitors
Compounds that inhibit HISTONE DEACETYLASES. This class of drugs may influence gene expression by increasing the level of acetylated HISTONES in specific CHROMATIN domains. (See all compounds classified as Histone Deacetylase Inhibitors.)
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
