

Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
KDMF
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
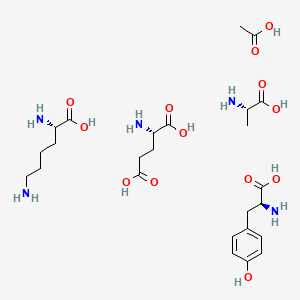

1. 5010, Tv
2. Acetate, Glatiramer
3. Copaxone
4. Glatiramer
5. Tv 5010
6. Tv-5010
7. Tv5010
1. 147245-92-9
2. Acetic Acid;(2s)-2-amino-3-(4-hydroxyphenyl)propanoic Acid;(2s)-2-aminopentanedioic Acid;(2s)-2-aminopropanoic Acid;(2s)-2,6-diaminohexanoic Acid
3. Glatirameracetat
4. Dtxsid30163637
5. Am84438
6. Ac-28732
7. L-alanine Compound With L-glutamic Acid And L-lysine And L-tyrosine And Acetic Acid (1:1:1:1:1)
8. Q418274
| Molecular Weight | 623.7 g/mol |
|---|---|
| Molecular Formula | C25H45N5O13 |
| Hydrogen Bond Donor Count | 12 |
| Hydrogen Bond Acceptor Count | 18 |
| Rotatable Bond Count | 13 |
| Exact Mass | 623.30138651 g/mol |
| Monoisotopic Mass | 623.30138651 g/mol |
| Topological Polar Surface Area | 374 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 519 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 4 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 5 |
| 1 of 2 | |
|---|---|
| Drug Name | Copaxone |
| PubMed Health | Glatiramer (Injection) |
| Drug Classes | Central Nervous System Agent, Immune Suppressant, Musculoskeletal Agent |
| Drug Label | Glatiramer acetate, the active ingredient of COPAXONE, consists of the acetate salts of synthetic polypeptides, containing four naturally occurring amino acids: L-glutamic acid, L-alanine, L-tyrosine, and L-lysine with an average molar fraction of 0.... |
| Active Ingredient | Glatiramer acetate |
| Dosage Form | Injectable |
| Route | Subcutaneous |
| Strength | 20mg/ml; 40mg/ml |
| Market Status | Prescription |
| Company | Teva Pharms Usa |
| 2 of 2 | |
|---|---|
| Drug Name | Copaxone |
| PubMed Health | Glatiramer (Injection) |
| Drug Classes | Central Nervous System Agent, Immune Suppressant, Musculoskeletal Agent |
| Drug Label | Glatiramer acetate, the active ingredient of COPAXONE, consists of the acetate salts of synthetic polypeptides, containing four naturally occurring amino acids: L-glutamic acid, L-alanine, L-tyrosine, and L-lysine with an average molar fraction of 0.... |
| Active Ingredient | Glatiramer acetate |
| Dosage Form | Injectable |
| Route | Subcutaneous |
| Strength | 20mg/ml; 40mg/ml |
| Market Status | Prescription |
| Company | Teva Pharms Usa |
Antirheumatic Agents
Drugs that are used to treat RHEUMATOID ARTHRITIS. (See all compounds classified as Antirheumatic Agents.)
Immunosuppressive Agents
Agents that suppress immune function by one of several mechanisms of action. Classical cytotoxic immunosuppressants act by inhibiting DNA synthesis. Others may act through activation of T-CELLS or by inhibiting the activation of HELPER CELLS. While immunosuppression has been brought about in the past primarily to prevent rejection of transplanted organs, new applications involving mediation of the effects of INTERLEUKINS and other CYTOKINES are emerging. (See all compounds classified as Immunosuppressive Agents.)
Adjuvants, Immunologic
Substances that augment, stimulate, activate, potentiate, or modulate the immune response at either the cellular or humoral level. The classical agents (Freund's adjuvant, BCG, Corynebacterium parvum, et al.) contain bacterial antigens. Some are endogenous (e.g., histamine, interferon, transfer factor, tuftsin, interleukin-1). Their mode of action is either non-specific, resulting in increased immune responsiveness to a wide variety of antigens, or antigen-specific, i.e., affecting a restricted type of immune response to a narrow group of antigens. The therapeutic efficacy of many biological response modifiers is related to their antigen-specific immunoadjuvanticity. (See all compounds classified as Adjuvants, Immunologic.)
L03AX13
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
L - Antineoplastic and immunomodulating agents
L03 - Immunostimulants
L03A - Immunostimulants
L03AX - Other immunostimulants
L03AX13 - Glatiramer acetate
GDUFA
DMF Review : Complete
Rev. Date : 2021-02-08
Pay. Date : 2020-12-24
DMF Number : 35493
Submission : 2020-12-25
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2014-06-05
Pay. Date : 2013-11-22
DMF Number : 27723
Submission : 2013-12-12
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 33092
Submission : 2018-09-12
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2018-03-07
Pay. Date : 2018-02-09
DMF Number : 28989
Submission : 2014-12-25
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 7609
Submission : 1988-08-02
Status : Inactive
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
 DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
DRL offers a portfolio of products & services, including APIs, CMO services, generics, biosimilars & differentiated formulations.
Date of Issue : 2022-06-17
Valid Till : 2025-07-07
Written Confirmation Number : WC-0067
Address of the Firm : Chemical Technical Operations Unit-VI, APIIC Industrial Estate, Pydibhimavaram V...
Date of Issue : 2019-09-09
Valid Till : 2022-08-04
Written Confirmation Number : WC-0038
Address of the Firm : Plot No.213, 214 & 215, I.D.A. Phase-II, Pashamylaram, Medak Dist, A.P

Date of Issue : 2022-08-04
Valid Till : 2025-03-04
Written Confirmation Number : WC-0427A3-4
Address of the Firm : Plot No.49 & 50, J.N. Pharma City, Parawada (M), Visakhapatnam - 531019, Andhra ...

Date of Issue : 2019-06-26
Valid Till : 2022-06-25
Written Confirmation Number : WC-0014
Address of the Firm : Mekaguda (V) kothur (M) Mahabood nagar Dist, Telangana State India

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Copaxone Depot, a long-acting glatiramer acetate designed to be administered as an intramuscular injection, is being investigated as a once-monthly injection for the treatment of relapsing forms of multiple sclerosis (RMS).
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: WhisperJECT
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Mapi Pharma
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable August 07, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Mapi Pharma
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : Copaxone Depot, a long-acting glatiramer acetate designed to be administered as an intramuscular injection, is being investigated as a once-monthly injection for the treatment of relapsing forms of multiple sclerosis (RMS).
Brand Name : WhisperJECT
Molecule Type : Small molecule
Upfront Cash : Not Applicable
August 07, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Copaxone (glatiramer acetate) its ability to induce the peripheral Th2 cells and their subsequent crossing of the blood barrier allows for the reduction of further inflammation within the CNS.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Oncology Brand Name: Copaxone
Study Phase: Phase IProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 03, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Oncology
Highest Development Status : Phase I
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Kinimmune Publishes Results from A Phase I “Window of Opportunity” Trial in Patients with Soli...
Details : Copaxone (glatiramer acetate) its ability to induce the peripheral Th2 cells and their subsequent crossing of the blood barrier allows for the reduction of further inflammation within the CNS.
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 03, 2023

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The study evaluating a long-acting glatiramer acetate (copaxone) injection, GA Depot 40 mg once every four weeks, met the primary endpoint, significantly reducing the annualized relapse rate (ARR) by 30.1 percent compared to placebo.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable September 21, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : The study evaluating a long-acting glatiramer acetate (copaxone) injection, GA Depot 40 mg once every four weeks, met the primary endpoint, significantly reducing the annualized relapse rate (ARR) by 30.1 percent compared to placebo.
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
September 21, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
At AAN, company will be presenting 5 year data from Phase IIa study in RRMS. Also, the results seeN in the ongoing Phase II study for GA Depot in primary progressive MS (PPMS) patients, to be presented at the meeting as plan to initiate a Phase-3 for PPMS at the end of 2022.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: GA Depot
Study Phase: Phase IIIProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 31, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : At AAN, company will be presenting 5 year data from Phase IIa study in RRMS. Also, the results seeN in the ongoing Phase II study for GA Depot in primary progressive MS (PPMS) patients, to be presented at the meeting as plan to initiate a Phase-3 for PPM...
Brand Name : GA Depot
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 31, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
GA Depot is a long-acting injection version of the approved Copaxone® (Glatiramer Acetate) designed to be administered as an intramuscular injection once every four weeks, is being evaluated for treatment of relapsing forms of multiple sclerosis.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable March 30, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Details : GA Depot is a long-acting injection version of the approved Copaxone® (Glatiramer Acetate) designed to be administered as an intramuscular injection once every four weeks, is being evaluated for treatment of relapsing forms of multiple sclerosis.
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
March 30, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
The label update of COPAXONE® (Glatiramer Acetate) follows the review of clinical and non-clinical evidence, including latest data from the COBRA study that investigated safety outcomes in infants breastfed by mothers with MS undergoing GA treatment during the first 18 months.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Immunology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable February 10, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Immunology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
COPAXONE® Label Now Indicates Its Use in Breastfeeding Mothers with Relapsing Multiple Sclerosis
Details : The label update of COPAXONE® (Glatiramer Acetate) follows the review of clinical and non-clinical evidence, including latest data from the COBRA study that investigated safety outcomes in infants breastfed by mothers with MS undergoing GA treatment dur...
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
February 10, 2022

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
Patients were eligible for the study if they were diagnosed with relapsing MS (RMS), gave a live birth, were breastfeeding under GA treatment (COPAXONE® 20 or 40 mg/mL) or breastfeeding under no DMT treatment.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable October 13, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
New Safety Data on Treatment with COPAXONE® (glatiramer acetate) of Breastfeeding Mothers who Liv...
Details : Patients were eligible for the study if they were diagnosed with relapsing MS (RMS), gave a live birth, were breastfeeding under GA treatment (COPAXONE® 20 or 40 mg/mL) or breastfeeding under no DMT treatment.
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
October 13, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
A non-interventional analysis of the study found no evidence of safety concerns in infants born to mothers with multiple sclerosis (MS) who were breastfeeding and receiving glatiramer acetate (GA) treatment.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable June 22, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
New Safety Data on Treatment of Breastfeeding Mothers with COPAXONE® (glatiramer acetate) Who Are...
Details : A non-interventional analysis of the study found no evidence of safety concerns in infants born to mothers with multiple sclerosis (MS) who were breastfeeding and receiving glatiramer acetate (GA) treatment.
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
June 22, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
GA Depot is a long-acting version of Teva Pharmaceutical’s Copaxone (glatiramer acetate). Mapi Pharma is working with the CRO PRA Health Sciences for its Phase III trial investigating glatiramer acetate depot (GA Depot) for relapsing forms of multiple sclerosis (RMS).
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Neurology Brand Name: Copaxone
Study Phase: ApprovedProduct Type: Small molecule
Sponsor: PRA Health Sciences
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable January 20, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Neurology
Highest Development Status : Approved
Partner/Sponsor/Collaborator : PRA Health Sciences
Deal Size : Not Applicable
Deal Type : Not Applicable
Mapi Pharma Engages PRA Health Sciences as CRO for Phase III Multiple Sclerosis Trial
Details : GA Depot is a long-acting version of Teva Pharmaceutical’s Copaxone (glatiramer acetate). Mapi Pharma is working with the CRO PRA Health Sciences for its Phase III trial investigating glatiramer acetate depot (GA Depot) for relapsing forms of multiple ...
Brand Name : Copaxone
Molecule Type : Small molecule
Upfront Cash : Not Applicable
January 20, 2021

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Details:
GA Depot is a long acting injection version of the approved Glatiramer Acetate products (Copaxone® and its generic versions) for treating multiple sclerosis (RMS), designed to be administered as an intramuscular injection once every four weeks.
Lead Product(s): Glatiramer Acetate
Therapeutic Area: Immunology Brand Name: GA Depot
Study Phase: Phase IIIProduct Type: Large molecule
Sponsor: Not Applicable
Deal Size: Not Applicable Upfront Cash: Not Applicable
Deal Type: Not Applicable December 21, 2020

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Lead Product(s) : Glatiramer Acetate
Therapeutic Area : Immunology
Highest Development Status : Phase III
Partner/Sponsor/Collaborator : Not Applicable
Deal Size : Not Applicable
Deal Type : Not Applicable
Mapi Pharma Provides Enrollment Update in Phase III GA Depot Clinical Trial
Details : GA Depot is a long acting injection version of the approved Glatiramer Acetate products (Copaxone® and its generic versions) for treating multiple sclerosis (RMS), designed to be administered as an intramuscular injection once every four weeks.
Brand Name : GA Depot
Molecule Type : Large molecule
Upfront Cash : Not Applicable
December 21, 2020

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?
