
Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. Ar-df 26
2. Beglynor
3. Beglynora
4. Glikvidon
5. Gliquidone, Calcium Salt
6. Gliquidone, Monosodium Salt
7. Glurenor
8. Glurenorm
9. Glycvidon
1. 33342-05-1
2. Glurenorm
3. Gliquidonum
4. Ardf 26
5. Beglynor
6. Gliquidone (inn)
7. Ardf-26
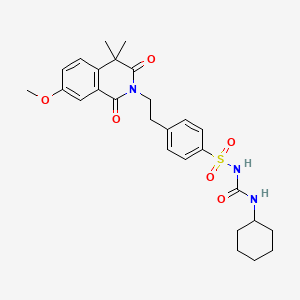
8. 1-cyclohexyl-3-[4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxoisoquinolin-2-yl)ethyl]phenyl]sulfonylurea
9. C7c2qdd75p
10. N-(cyclohexylcarbamoyl)-4-(2-(7-methoxy-4,4-dimethyl-1,3-dioxo-3,4-dihydroisoquinolin-2(1h)-yl)ethyl)benzenesulfonamide
11. Ncgc00016819-01
12. Gliquidona
13. Gliquidone [inn]
14. Cas-33342-05-1
15. Dsstox_cid_3096
16. Dsstox_rid_76873
17. Dsstox_gsid_23096
18. Gliquidonum [inn-latin]
19. 3-cyclohexyl-1-[4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-isoquinolin-2-yl)ethyl]phenyl]sulfonyl-urea
20. Gliquidona [inn-spanish]
21. Ardf 26 Se
22. Ar-df 26
23. Glurenorm (tn)
24. 1-cyclohexyl-3-({4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-1,2,3,4-tetrahydroisoquinolin-2-yl)ethyl]benzene}sulfonyl)urea
25. Smr000685793
26. Gliquidone [inn:ban]
27. Einecs 251-463-2
28. Mfcd00631870
29. Unii-c7c2qdd75p
30. Brn 1559726
31. Gliquidon
32. Gliquidone,(s)
33. Gliquidone, 98%
34. Gliquidone [mi]
35. Prestwick0_000991
36. Prestwick1_000991
37. Prestwick2_000991
38. Prestwick3_000991
39. Gliquidone [mart.]
40. Gliquidone [who-dd]
41. Oprea1_069443
42. Schembl37769
43. Bspbio_001081
44. 1-cyclohexyl-3-((p-(2-(3,4-dihydro-7-methoxy-4,4-dimethyl-1,3-dioxo-2(1h)-isoquinolyl)ethyl)phenyl)sulfonyl)urea
45. 1-cyclohexyl-3-(p-beta-(4,4-dimethyl-2-methoxy-1,3-(2h,4h)-isoquinolinedione-2-yl)phenethyl)sulfonylurea
46. Benzenesulfonamide, N-((cyclohexylamino)carbonyl)-4-(2-(3,4-dihydro-7-methoxy-4,4-dimethyl-1,3-dioxo-2(1h)-isoquinolinyl)ethyl)-
47. Mls000881123
48. Mls000881174
49. Mls002154087
50. Spbio_002972
51. Bpbio1_001191
52. Chembl383634
53. Dtxsid4023096
54. Chebi:93416
55. Hms1571g03
56. Hms2098g03
57. Hms2964j06
58. Hms3652o05
59. Hms3715g03
60. Act06290
61. Bcp11927
62. Hy-b1114
63. Zinc1482077
64. Tox21_110629
65. Bdbm50248247
66. Akos001037880
67. Tox21_110629_1
68. Ccg-220991
69. Cs-4646
70. Db01251
71. Ks-5239
72. Ncgc00016819-02
73. Ncgc00016819-03
74. Ncgc00016819-05
75. Ncgc00016819-06
76. Ncgc00179315-01
77. Ac-23356
78. N-[(cyclohexylamino)carbonyl]-4-{2-[4,4-dimethyl-7-(methyloxy)-1,3-dioxo-3,4-dihydroisoquinolin-2(1h)-yl]ethyl}benzenesulfonamide
79. N-(cyclohexylcarbamoyl)-4-(2-(7-methoxy-
80. Ab00513989
81. Ft-0630570
82. G0332
83. S3151
84. Sw197285-3
85. D02430
86. T72318
87. 4,4-dimethyl-1,3-dioxo-3,4-dihydroisoquinolin
88. Ab00513989_07
89. 342g051
90. A821736
91. Sr-01000833866
92. J-019153
93. Q5569924
94. Sr-01000833866-2
95. Brd-k80396088-001-03-9
96. 1-cyclohexyl-3-(((p-(2-(3,4-dihydro-7-methoxy-4,4-dimethyl-1,3-dioxo-2(1h)-isoquinolyl)ethyl)phenyl)sulfonyl)urea
97. 1-cyclohexyl-3-[4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-2-isoquinolinyl)ethyl]phenyl]sulfonylurea
98. 1-cyclohexyl-3-[4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-2-isoquinolyl)ethyl]phenyl]sulfonyl-urea;gliquidone
99. 3-cyclohexyl-1-({4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-1,2,3,4-tetrahydroisoquinolin-2-yl)ethyl]benzene}sulfonyl)urea
100. N-cyclohexylcarbamoyl-4-[2-(7-methoxy-4,4-dimethyl-1,3-dioxo-1,2,3,4- Tetrahydroisoquinolin-2-yl)ethyl]benzenesulfonamide
| Molecular Weight | 527.6 g/mol |
|---|---|
| Molecular Formula | C27H33N3O6S |
| XLogP3 | 4.6 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 7 |
| Exact Mass | 527.20900695 g/mol |
| Monoisotopic Mass | 527.20900695 g/mol |
| Topological Polar Surface Area | 130 Ų |
| Heavy Atom Count | 37 |
| Formal Charge | 0 |
| Complexity | 948 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Used in the treatment of diabetes mellitus type 2.
Gliquidone is an anti-diabetic drug in the sulfonylurea class. In patients with diabetes mellitus, there is a deficiency or absence of a hormone manufactured by the pancreas called insulin. Insulin is the main hormone responsible for the control of sugar in the blood. Gliquidone is an antidiabetic medication which is used in those patients with adult maturity onset or non-insulin dependent diabetes (NIDDM). It works by lowering blood sugar levels by stimulating the production and release of insulin from the pancreas. It also promotes the movement of sugar from the blood into the cells in the body which need it.
Hypoglycemic Agents
Substances which lower blood glucose levels. (See all compounds classified as Hypoglycemic Agents.)
A10BB08
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
A - Alimentary tract and metabolism
A10 - Drugs used in diabetes
A10B - Blood glucose lowering drugs, excl. insulins
A10BB - Sulfonylureas
A10BB08 - Gliquidone
The mean terminal half-life was approximately 8 hours (range 5.7-9.4 hours)
The mechanism of action of gliquidone in lowering blood glucose appears to be dependent on stimulating the release of insulin from functioning pancreatic beta cells, and increasing sensitivity of peripheral tissues to insulin. Gliquidone likely binds to ATP-sensitive potassium channel receptors on the pancreatic cell surface, reducing potassium conductance and causing depolarization of the membrane. Membrane depolarization stimulates calcium ion influx through voltage-sensitive calcium channels. This increase in intracellular calcium ion concentration induces the secretion of insulin.
Global Sales Information

Market Place

ABOUT THIS PAGE
10
PharmaCompass offers a list of Gliquidone API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Gliquidone manufacturer or Gliquidone supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Gliquidone manufacturer or Gliquidone supplier.
PharmaCompass also assists you with knowing the Gliquidone API Price utilized in the formulation of products. Gliquidone API Price is not always fixed or binding as the Gliquidone Price is obtained through a variety of data sources. The Gliquidone Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Gliquidone manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Gliquidone, including repackagers and relabelers. The FDA regulates Gliquidone manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Gliquidone API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Gliquidone manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Gliquidone supplier is an individual or a company that provides Gliquidone active pharmaceutical ingredient (API) or Gliquidone finished formulations upon request. The Gliquidone suppliers may include Gliquidone API manufacturers, exporters, distributors and traders.
click here to find a list of Gliquidone suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
Gliquidone Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Gliquidone GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Gliquidone GMP manufacturer or Gliquidone GMP API supplier for your needs.
A Gliquidone CoA (Certificate of Analysis) is a formal document that attests to Gliquidone's compliance with Gliquidone specifications and serves as a tool for batch-level quality control.
Gliquidone CoA mostly includes findings from lab analyses of a specific batch. For each Gliquidone CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Gliquidone may be tested according to a variety of international standards, such as European Pharmacopoeia (Gliquidone EP), Gliquidone JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Gliquidone USP).
