
Synopsis
Synopsis
0
EU WC
0
KDMF
0
VMF
0
Australia
DRUG PRODUCT COMPOSITIONS
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Big Plasma Glucagon
2. Glucagon-like-immunoreactivity
3. Gut Glucagon-like Immunoreactants
1. Glucagonum
2. Glucagone
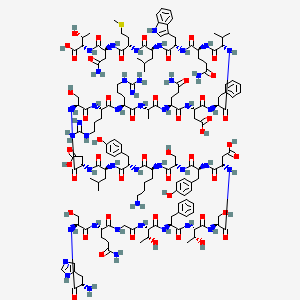
3. His-ser-glu(nh2)-gly-thr-phe-thr-ser-asp-tyr-ser-lys-tyr-leu-asp-ser-arg-arg-ala-glu(nh2)-asp-phe-val-glu(nh2)-trp-leu-met-asp(nh2)-thr
4. Glucagon-like-immunoreactivity
5. Glucaton
6. Bovine Glucagon
7. Glukagon Novo
8. Glucagon, Pig
9. Glucagon (dog)
10. Glucagon (pig)
11. Glucagon (ox)
12. Big Plasma Glucagon
13. Glucagone [dcit]
14. Glucagonum [inn-latin]
15. Glucagon (xenopus Laevis)
16. Glucagon (saimiri Sciureus)
17. Unii-76la80ig2g
18. 76la80ig2g
19. Glucagon (mesocricetus Auratus)
20. Glucagon, Porcine, For Bioassay
21. Glucagon [usp:inn:ban:jan]
22. Schembl15268863
23. Gut Glucagon-like Immunoreactants
24. Hsdb 3337
25. Dtxsid101016809
26. Glucagon, Porcine, For Immunoassay
27. Hsqgtftsdyskyldsrraqdfvqwlmnt
28. Einecs 232-708-2
29. Ncgc00167140-01
30. Glucagon, Acetate Salt, >=97.0% (hplc)
31. Human Glucagon, European Pharmacopoeia (ep) Reference Standard
32. His-ser-gln-gly-thr-phe-thr-ser-asp-tyr-ser-lys-tyr-leu-asp-ser-arg-arg-ala-gln-asp-phe-val-gln-trp-leu-met-asn-thr
33. L-histidyl-l-seryl-l-glutaminylglycyl-l-threonyl-l-phenylalanyl-l-threonyl-l-seryl-l-alpha-aspartyl-l-tyrosyl-l-seryl-l-lysyl-l-tyrosyl-l-leucyl-l-alpha-aspartyl-l-seryl-l-arginyl-l-arginyl-l-alanyl-l-glutaminyl-l-alpha-aspartyl-l-phenylalanyl-l-valyl-l-glutaminyl-l-tryptophyl-l-leucyl-l-methionyl-l-asparaginyl-l-threonine
34. L-threonine, L-histidyl-l-seryl-l-glutaminylglycyl-l-threonyl-l-phenylalanyl-l-threonyl-l-seryl-l-.alpha.-aspartyl-l-tyrosyl-l-seryl-l-lysyl-l-tyrosyl-l-leucyl-l-.alpha.-aspartyl-l-seryl-l-arginyl-l-argin Yl-l-alanyl-l-glutaminyl-l-.alpha.-aspartyl-l-phenylalanyl-l-valyl-l-glutaminyl-l-tryptophyl-l-leucyl-l-methionyl-l-asparaginyl-
35. L-threonine, L-histidyl-l-seryl-l-glutaminylglycyl-l-threonyl-l-phenylalanyl-l-threonyl-l-seryl-l-alpha-aspartyl-l-tyrosyl-l-seryl-l-lysyl-l-tyrosyl-l-leucyl-l-alpha-aspartyl-l-seryl-l-arginyl-l-arginyl-l-alanyl-l-glytaminyl-l-alpha-aspartyl-l-phenylalanyl-l-valyl-l-glutaminyl-l-tryptophyl-l-leucyl-l-methionyl-l-asparaginyl-
| Molecular Weight | 3482.7 g/mol |
|---|---|
| Molecular Formula | C153H225N43O49S |
| XLogP3 | -16.9 |
| Hydrogen Bond Donor Count | 55 |
| Hydrogen Bond Acceptor Count | 55 |
| Rotatable Bond Count | 115 |
| Exact Mass | 3481.6190567 g/mol |
| Monoisotopic Mass | 3480.6157019 g/mol |
| Topological Polar Surface Area | 1560 Ų |
| Heavy Atom Count | 246 |
| Formal Charge | 0 |
| Complexity | 8160 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 31 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Gastrointestinal Agents; Protein Synthesis Inhibitors
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
Glucagon is used in the treatment of lower esophageal obstruction due to foreign bodies, including food boluses. /NOT included in US product labeling/
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1440
Glucagon may be of use in treating myocardial depression due to calcium channel blocking agents in those patients in whom conventional therapies have been ineffective. /NOT included in US product labeling/
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1440
Glucagon administered in large intravenous doses is used to treat the cardiotoxic effects, specifically bradycardia and hypotension, in overdoses of beta-adrenergic blocking agents. Glucagon may be used with the proterenol or dobutamine. Supplemental potassium may be necessary for treated patients since glucagon tends to reduce serum potassium. /NOT included in US product labeling/
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1440
For more Therapeutic Uses (Complete) data for GLUCAGON (19 total), please visit the HSDB record page.
...EFFECTIVE ONLY WHEN ADMIN PARENTERALLY. ITS HYPERGLYCEMIC EFFECT IS...OF RELATIVELY BRIEF DURATION. .../SUPPLEMENTARY CARBOHYDRATES SHOULD BE GIVEN AS SOON AS POSSIBLE AFTER PATIENT RESPONDS/. AN ADDITIONAL SUGAR SOURCE IS ESPECIALLY IMPORTANT IN JUVENILES...
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 1045
Since glucagon is a protein, the possibility of hypersensitivity reactions should be considered.
McEvoy, G.K. (ed.). American Hospital Formulary Service--Drug Information 94. Bethesda, MD: American Society of Hospital Pharmacists, Inc. 1994 (Plus Supplements)., p. 2076
Side/Adverse Effects: Those indicating need for medical attention only if they continue or are bothersome: Nausea or vomiting - incidence is generally dependent upon dose and (with intravenous use) the rate of injection; these effects may be diminished by slower intravenous administration.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1441
Glucagon should not be used to treat birth asphyxia or hypoglycemia in premature infants or in infants who have had intrauterine growth retardation.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1440
Glucagon has been used as an aid in the diagnosis of insulinoma and pheochromocytoma; however, USP advisory panels do not generally recommend this use because of questions about safety.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1440
Glucagon is indicated as a diagnostic aid in radiologic exams to temporarily inhibit the movement of the gastrointestinal tract and to treat severe hypoglycemia.
FDA Label
Baqsimi is indicated for the treatment of severe hypoglycaemia in adults, adolescents, and children aged 4 years and over with diabetes mellitus.
Ogluo is indicated for the treatment of severe hypoglycaemia in adults, adolescents, and children aged 2 years and over with diabetes mellitus.
Treatment of hypoglycaemia
Glucagon is indicated as a diagnostic aid in radiologic exams to temporarily inhibit the movement of the gastrointestinal tract and severe hypoglycemia. Glucagon raises blood sugar through activation of hepatic glucagon receptors, stimulating glycogenolysis and the release of glucose. Glucagon has a short duration of action. Glucagon may cause hyperglycemia in diabetic patients.
H04AA01
H04AA01
H - Systemic hormonal preparations, excl. sex hormones and insulins
H04 - Pancreatic hormones
H04A - Glycogenolytic hormones
H04AA - Glycogenolytic hormones
H04AA01 - Glucagon
Absorption
A 1mg intravenous dose of glucagon reaches a Cmax of 7.9ng/mL with a Tmax of 20 minutes. An intramuscular dose reaches a Cmax of 6.9ng/mL with a Tmax of 13 minutes. A 3mg dose of glucagon nasal powder reaches a Cmax of 6130pg/mL with a Tmax of 15 minutes.
Route of Elimination
Elimination of glucagon is not fully characterized in literature, however the kidney and liver appear to contribute significantly in animal models. The liver and kidney are responsible for approximately 30% of glucagon elimination each.
Volume of Distribution
The volume of distribution of glucagon is 0.25L/kg. The apparent volume of distribution is 885L.
Clearance
A 1mg intravenous dose of glucagon has a clearance of 13.5mL/min/kg.
Because of its polypeptide nature, glucagon is destroyed in the GI tract, and therefore must be administered parenterally.
McEvoy, G.K. (ed.). American Hospital Formulary Service--Drug Information 94. Bethesda, MD: American Society of Hospital Pharmacists, Inc. 1994 (Plus Supplements)., p. 2075
Glucagon is a protein and so it is metabolized into smaller polypeptides and amino acids in the liver, kidney, and plasma.
The half life of glucagon is 26 minutes for an intramuscular dose. The half life of glucagon nasal powder is approximately 35 minutes. The half life of glucagon by a subcutaneous auto-injector or pre-filled syringe is 32 minutes.
Glucagon has a plasma half-life of about 3-10 minutes.
McEvoy, G.K. (ed.). American Hospital Formulary Service--Drug Information 94. Bethesda, MD: American Society of Hospital Pharmacists, Inc. 1994 (Plus Supplements)., p. 2075
Glucagon binds to the glucagon receptor activating Gs and Gq. This activation activates adenylate cyclase, which increases intracellular cyclic AMP and activates protein kinase A. Activating Gq activates phospholipase C, increases production of inositol 1,4,5-triphosphate, and releases intracellular calcium. Protein kinase A phosphorylates glycogen phosphorylase kinase, which phosphorylates glycogen phosphorylase, which phosphorylates glycogen, causing its breakdown. Glucagon also relaxes smooth muscle of the stomach, duodenum, small bowel, and colon.
Glucagon increases the blood glucose concentration by mobilizing hepatic glycogen and thus is effective only when hepatic glycogen is available. Patients with reduced glycogen stores (eg, starvation, adrenal insufficiency, alcoholic hypoglycemia) cannot respond to glucagon.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 1045
Glucagon produces extra hepatic effects that are independent of its hyperglycemic action. Although the exact mechanism(s) of action has not been conclusively determined, glucagon produces relaxation of smooth muscle of the stomach, duodenum, small intestine, and colon. The drug has also been shown to inhibit gastric and pancreatic secretions.
McEvoy, G.K. (ed.). American Hospital Formulary Service--Drug Information 94. Bethesda, MD: American Society of Hospital Pharmacists, Inc. 1994 (Plus Supplements)., p. 2075
Promotes hepatic glycogenolysis and gluconeogenesis. Stimulates adenylate cyclase to produce increased cyclic-AMP, which is involved in a series of enzymatic activities. The resultant effects are increased concentrations of plasma glucose, a relaxant effect on smooth musculature, and an inotropic myocardial effect. Hepatic stores of glycogen are necessary for glucagon to elicit an antihypoglycemic effect.
USP Convention. USPDI-Drug Information for the Health Care Professional. 14th ed. Volume I. Rockville, MD: United States Pharmacopeial Convention, Inc., 1994. (Plus Updates)., p. 1441
 Bachem Group is a public, innovation-driven company specializing in the development and manufacturing of peptides and oligonucleotides.
Bachem Group is a public, innovation-driven company specializing in the development and manufacturing of peptides and oligonucleotides.
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2015-05-07
Pay. Date : 2015-04-27
DMF Number : 18277
Submission : 2005-04-12
Status : Active
Type : II

Registration Number : 231MF10022
Registrant's Address : Hauptstrasse 144,4416 Bubendorf Switzerland
Initial Date of Registration : 2019-01-29
Latest Date of Registration :

NDC Package Code : 55463-0009
Start Marketing Date : 2005-03-10
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT
 Bachem Group is a public, innovation-driven company specializing in the development and manufacturing of peptides and oligonucleotides.
Bachem Group is a public, innovation-driven company specializing in the development and manufacturing of peptides and oligonucleotides.

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 32703
Submission : 2018-06-08
Status : Active
Type : II

Registration Number : 218MF10708
Registrant's Address : Hauptstrasse 144,4416 Bubendorf Switzerland
Initial Date of Registration : 2006-08-11
Latest Date of Registration :

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26326
Submission : 2012-08-21
Status : Active
Type : II

NDC Package Code : 41524-0005
Start Marketing Date : 2012-09-06
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 36017
Submission : 2022-12-22
Status : Active
Type : II

NDC Package Code : 66558-0195
Start Marketing Date : 2022-10-01
End Marketing Date : 2026-12-31
Dosage Form (Strength) : POWDER (1g/g)
Marketing Category : BULK INGREDIENT


USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
USDMF, CEP/COS, JDMF, EU-WC, NDC, KDMF, VMF, Others
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 5546
Submission : 1984-09-20
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 2961
Submission : 1977-06-07
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 3738
Submission : 1980-03-14
Status : Inactive
Type : II



Certificate Number : R1-CEP 2005-083 - Rev 03
Issue Date : 2021-08-27
Type : TSE
Substance Number :
Status : Valid


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

Patents & EXCLUSIVITIES
REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
97
PharmaCompass offers a list of Glucagon API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glucagon manufacturer or Glucagon supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Glucagon manufacturer or Glucagon supplier.
A Glucagon manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glucagon, including repackagers and relabelers. The FDA regulates Glucagon manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glucagon API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Glucagon manufacturers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PhamaCompass.
A Glucagon supplier is an individual or a company that provides Glucagon active pharmaceutical ingredient (API) or Glucagon finished formulations upon request. The Glucagon suppliers may include Glucagon API manufacturers, exporters, distributors and traders.
click here to find a list of Glucagon suppliers with USDMF, JDMF, KDMF, CEP, GMP, and COA related information on PharmaCompass.
A Glucagon DMF (Drug Master File) is a document detailing the whole manufacturing process of Glucagon active pharmaceutical ingredient (API) in detail. Different forms of Glucagon DMFs exist exist since differing nations have different regulations, such as Glucagon USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Glucagon DMF submitted to regulatory agencies in the US is known as a USDMF. Glucagon USDMF includes data on Glucagon's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Glucagon USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Glucagon suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Glucagon Drug Master File in Japan (Glucagon JDMF) empowers Glucagon API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Glucagon JDMF during the approval evaluation for pharmaceutical products. At the time of Glucagon JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Glucagon suppliers with JDMF on PharmaCompass.
A Glucagon CEP of the European Pharmacopoeia monograph is often referred to as a Glucagon Certificate of Suitability (COS). The purpose of a Glucagon CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Glucagon EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Glucagon to their clients by showing that a Glucagon CEP has been issued for it. The manufacturer submits a Glucagon CEP (COS) as part of the market authorization procedure, and it takes on the role of a Glucagon CEP holder for the record. Additionally, the data presented in the Glucagon CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Glucagon DMF.
A Glucagon CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Glucagon CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Glucagon suppliers with CEP (COS) on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Glucagon as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Glucagon API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Glucagon as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Glucagon and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Glucagon NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Glucagon suppliers with NDC on PharmaCompass.
Glucagon Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glucagon GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, and more, enabling you to easily find the right Glucagon GMP manufacturer or Glucagon GMP API supplier for your needs.
A Glucagon CoA (Certificate of Analysis) is a formal document that attests to Glucagon's compliance with Glucagon specifications and serves as a tool for batch-level quality control.
Glucagon CoA mostly includes findings from lab analyses of a specific batch. For each Glucagon CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glucagon may be tested according to a variety of international standards, such as European Pharmacopoeia (Glucagon EP), Glucagon JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glucagon USP).

