
Synopsis
Synopsis
0
EU WC
0
KDMF
0
NDC API
0
VMF
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
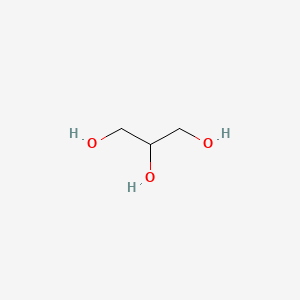

1. 1,2,3-propanetriol
2. 1,2,3-trihydroxypropane
3. Glycerin
4. Glycerine
1. Glycerin
2. 56-81-5
3. Glycerine
4. 1,2,3-propanetriol
5. Propane-1,2,3-triol
6. Glycyl Alcohol
7. Trihydroxypropane
8. Glyceritol
9. Propanetriol
10. 1,2,3-trihydroxypropane
11. Osmoglyn
12. Polyglycerine
13. Grocolene
14. Glysanin
15. Glyrol
16. Glycerin, Synthetic
17. Polyglycerol
18. Dagralax
19. Glycerinum
20. Ophthalgan
21. Vitrosupos
22. Glycerin, Anhydrous
23. Synthetic Glycerin
24. Synthetic Glycerine
25. Optim
26. Moon
27. Star
28. Incorporation Factor
29. Glycerolum
30. 90 Technical Glycerine
31. Glycerin Mist
32. Glycerin (mist)
33. 25618-55-7
34. Citifluor Af 2
35. Bulbold
36. Cristal
37. Glicerina [dcit]
38. Glycerine Mist
39. Caswell No. 469
40. Glycerin [jan]
41. Fema No. 2525
42. Propanetriol (van)
43. Glycerin Base
44. Tryhydroxypropane
45. Glycerin, Natural
46. Glycerin,anhydrous
47. Glicerol [inn-spanish]
48. Glycerolum [inn-latin]
49. Clyzerin, Wasserfrei
50. Pricerine 9091
51. Clyzerin, Wasserfrei [german]
52. Emery 916
53. Ccris 2295
54. Hsdb 492
55. Monoctanoin Component D
56. Epa Pesticide Chemical Code 063507
57. Collyrium Fresh-eye Drops
58. Ai3-00091
59. Nsc 9230
60. Glycerol [inn]
61. Brn 0635685
62. Ifp
63. Di-o-tolylphenylphosphine
64. Dynastin 7
65. Nsc-9230
66. Mfcd00004722
67. Pdc6a3c0ox
68. 1,2,3-trihydroxypropanol
69. Ins No.422
70. Chebi:17754
71. Ins-422
72. Nsc9230
73. Glycerol (inn)
74. M 314429
75. 101662-08-2
76. 144086-03-3
77. Pzn 7474853
78. Ncgc00090950-03
79. Diacylglycerol(35:0)
80. 2-propenoic Acid, Polymer With Oxirane And 1,2,3-propanetriol
81. Dsstox_cid_662
82. E-422
83. Dsstox_rid_75717
84. Dsstox_gsid_20663
85. 107283-02-3
86. 153050-05-6
87. 18803-09-3
88. 1h-thieno[3,4-d]imidazole-4-pentanamide, Hexahydro-2-oxo-n-[6-oxo-6-(2-propenylamino)hexyl]-, (3as,4
89. 2-propenoic Acid, Polymer With 2,2-bis(hydroxymethyl)-1,3-propanediol, Methyloxirane And Oxirane
90. 26403-55-4
91. Glycerol; Propane-1,2,3-triol
92. Glicerina
93. Glicerol
94. Heterochromatin-specific Nonhistone Chromosomal Protein Hp-1
95. Dag 31:3
96. Dag 35:0
97. Dag(35:0)
98. Glycerol, Acs Reagent, >=99.5%
99. Poly[oxy(2-hydroxy-1,3-propanediyl)], Alpha-hydro-omega-hydroxy-
100. Glyceol Opthalgan
101. Dg 31:3
102. Dg 35:0
103. Cas-56-81-5
104. Dg(31:3)
105. Dg(35:0)
106. Gol
107. Mackstat H 66
108. Wurcs=2.0/1,1,0/[h2h]/1/
109. Glycerin [usp:jan]
110. Rg-s
111. Einecs 200-289-5
112. Unii-pdc6a3c0ox
113. Neutracett
114. Glyceol
115. Glyzerin
116. Oelsuess
117. Glycerine Usp
118. Artifical Tears
119. D-glycerol
120. L-glycerol
121. Organic Glycerin
122. Glycerol Solution
123. Organic Glycerine
124. Glycerine (crude)
125. Polyhydric Alcohols
126. 1,3-propanetriol
127. Glycerol, Ultrapure
128. Glycerin Usp Grade
129. Glycerine 96%
130. Glycerol 85%
131. Diacylglycerol 31:3
132. Diacylglycerol 35:0
133. 8043-29-6
134. Glycerin 99.5%
135. Glycerine 96% Usp
136. 1,3-trihydroxypropane
137. 90 Technical Glycerin
138. Emery 912
139. Diacylglycerol(31:3)
140. Glycerin [hsdb]
141. Glycerin [inci]
142. Glycerol [fhfi]
143. Glycerin [ii]
144. Glycerol [mi]
145. Glycerin [vandf]
146. Dl-[1-13c]glycerol
147. Glycerinum [hpus]
148. E 422
149. Glycerin (jp17/usp)
150. Glycerin 99.5% Usp
151. Glycerine 99.7% Usp
152. Glycerol [mart.]
153. Bmse000184
154. Bmse000807
155. Bmse000856
156. Chembl692
157. Glycerin [usp-rs]
158. Glycerol [who-dd]
159. Glycerol [who-ip]
160. Molmap_000024
161. Ec 200-289-5
162. Glycerol, >=99.5%
163. Glycerol, Biochemical Grade
164. Glycerin Reagent Grade Acs
165. Wln: Q1yq1q
166. Glycerine (fragrance Grade)
167. 2-hydroxylpropane-1,3-diol
168. Glycerol, Lr, >=98%
169. Glycerol, Analytical Standard
170. 4-01-00-02751 (beilstein Handbook Reference)
171. Glycerol, Cell Culture Grade
172. Glycerol-[1,3-13c2]
173. Glycerin, Concentrated (jan)
174. Glycerol Min 98%, Anhydrous
175. Concentrated Glycerin (jp17)
176. Glycerol 3 M Solution, 3 M
177. Glycerol, >=99% (gc)
178. Glycerin [orange Book]
179. Glycerol [ep Impurity]
180. Gtpl5195
181. Qspl 181
182. Glycerol [ep Monograph]
183. Dtxsid9020663
184. Glycerin [usp Monograph]
185. Glycerol, Ar, >=99.5%
186. Pentrioxido Sulfurico Glycerincol
187. Glycerol, >99%, Fcc, Fg
188. Glycerol, Technical Grade, 95%
189. Glycerol, Ultrapure, Hplc Grade
190. 2w97
191. Chebi:131416
192. Chebi:178017
193. Chebi:189439
194. Glycerol 85% [who-dd]
195. Glycerol, Acs Reagent, 99.5%
196. Glycerol, Molecular Biology Grade
197. Glycerolum [who-ip Latin]
198. Glycerin,anhydrous [vandf]
199. Pharmakon1600-01300020
200. Zinc895048
201. Glycerol, Vetec(tm) Reagent Grade
202. Glycerol Solution, 86-89% (t)
203. Hy-b1659
204. Str02073
205. Glycerine 912 (96% Cp/usp)
206. Tox21_111043
207. Tox21_202077
208. Tox21_300144
209. C0066
210. Dag(31:3)
211. Glycerol, Bioxtra, >=99% (gc)
212. Glycerol, Reagentplus(r), >=99%
213. Nsc759633
214. S2766
215. Stl199174
216. 2-propanol, 1,3-dihydroxy-
217. Akos000120102
218. Cs-6964
219. Db09462
220. Glycerol, Usp, 99.0-101.0%
221. Nsc-759633
222. Sb83762
223. Glycerin; Propane-1,2,3-triol
224. Glycerolglycerin; Propane-1,2,3-triol
225. Glycerol, Saj First Grade, >=98.0%
226. Ncgc00090950-01
227. Ncgc00090950-02
228. Ncgc00090950-04
229. Ncgc00090950-05
230. Ncgc00253975-01
231. Ncgc00259626-01
232. Bp-31039
233. E422
234. Glycerol, For Molecular Biology, >=99%
235. Glycerol, Jis Special Grade, >=99.0%
236. Glycerol, Vetec(tm) Reagent Grade, 99%
237. Glycerin, Meets Usp Testing Specifications
238. Ft-0626742
239. Ft-0669018
240. Ft-0697060
241. G0316
242. Glycerol, Ultrapure, Spectrophotometric Grade
243. Glycerol, Reagentplus(r), >=99.0% (gc)
244. Glycerol, Spectrophotometric Grade, >=99.5%
245. C00116
246. D00028
247. D92249
248. A831186
249. Glycerol Solution, Puriss. P.a., 86-89% (t)
250. Glycerol, Tested According To Ph.eur., Anhydrous
251. Q132501
252. Brd-k73866522-001-02-6
253. Glycerol-gelatine, For Mounting (histochemical Slides)
254. F0001-1470
255. 8dfdfcd7-1ed2-4373-845e-054f5ad00089
256. Glycerin, United States Pharmacopeia (usp) Reference Standard
257. Glycerin, Pharmaceutical Secondary Standard; Certified Reference Material
258. Glycerol, Bioultra, For Molecular Biology, Anhydrous, >=99.5% (gc)
259. Glycerol, P.a., Acs Reagent, Reag. Iso, Reag. Ph. Eur., 98.0-101.0%
260. Glycerol, Puriss. P.a., Acs Reagent, Anhydrous, Dist., >=99.5% (gc)
261. Astm(r) D6584 Glycerin Solution, 500 Mug/ml In Pyridine, Analytical Standard
262. Astm(r) D6584 Glycerin Solution, Certified Reference Material, 500 Mug/ml In Pyridine
263. Glycerol Solution, Puriss., Meets Analytical Specification Of Ph.??eur., Bp, 84-88%
264. Glycerol, Bioreagent, Suitable For Cell Culture, Suitable For Insect Cell Culture, Suitable For Electrophoresis, >=99% (gc)
265. Glycerol, Polymer-bound, Extent Of Labeling: 1-2 Mmol/g Glycerol Loading, 1 % Cross-linked With Divinylbenzene
266. Glycerol, Puriss., Meets Analytical Specification Of Ph. Eur., Bp, Usp, Fcc, E422, Anhydrous, 99.0-101.0% (alkalimetric)
| Molecular Weight | 92.09 g/mol |
|---|---|
| Molecular Formula | C3H8O3 |
| XLogP3 | -1.8 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 2 |
| Exact Mass | 92.047344113 g/mol |
| Monoisotopic Mass | 92.047344113 g/mol |
| Topological Polar Surface Area | 60.7 Ų |
| Heavy Atom Count | 6 |
| Formal Charge | 0 |
| Complexity | 25.2 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cathartics; Cryoprotective Agents; Solvents; Vehicles
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
/Glycerin is indicated/ for relief of occasional constipation.
US Natl Inst Health; DailyMed. Current Medication Information for ADULT GLYCERIN LAXATIVE (glycerin) suppository (February 2010). Available from, as of July 18, 2011: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f44d5cca-c28d-4f37-92db-510f6605be90
/Glycerin/ is indicated as a/ lubricant /to/ relieve dryness of the eyes and prevent further irritation.
US Natl Inst Health; DailyMed. Current Medication Information for SOOTHE (glycerin and propylene glycol) solution/ drops (August 2010). Available from, as of July 18, 2011: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=fef002ea-c4bd-4486-a42e-1bbd9d73d28d
Glycerin ophthalmic solution may be used topically to reduce superficial corneal edema resulting from disease to facilitate opthalmoscopic and gonioscopic examination.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1773
For more Therapeutic Uses (Complete) data for GLYCERIN (7 total), please visit the HSDB record page.
For rectal use only. May cause rectal discomfort or a burning sensation.
US Natl Inst Health; DailyMed. Current Medication Information for ADULT GLYCERIN LAXATIVE (glycerin) suppository (February 2010). Available from, as of July 18, 2011: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f44d5cca-c28d-4f37-92db-510f6605be90
Do not use for more than one per day; for a period of longer than one week unless directed by a doctor; laxative products when abdominal pain, nausea, or vomiting are present unless directed by a doctor; if seal under product lid is damaged, missing or broken.
US Natl Inst Health; DailyMed. Current Medication Information for ADULT GLYCERIN LAXATIVE (glycerin) suppository (February 2010). Available from, as of July 18, 2011: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f44d5cca-c28d-4f37-92db-510f6605be90
If you have rectal bleeding or fail to have a bowel movement after using a laxative. This may indicate a serious condition.
US Natl Inst Health; DailyMed. Current Medication Information for ADULT GLYCERIN LAXATIVE (glycerin) suppository (February 2010). Available from, as of July 18, 2011: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=f44d5cca-c28d-4f37-92db-510f6605be90
Adverse effects occur rarely following rectal administration of glycerin or sorbitol. Glycerin may produce rectal discomfort, irritation, burning or griping, cramping pain and tenesmus. Hyperemia of the rectal mucosa with minimal amounts of hemorrhage and mucus discharge may also occur. These adverse effects occur less frequently following rectal administration of sorbitol.
American Society of Health-System Pharmacists 2011; Drug Information 2011. Bethesda, MD. 2011
For more Drug Warnings (Complete) data for GLYCERIN (11 total), please visit the HSDB record page.
It is used as a solvent, emollient, pharmaceutical agent, and sweetening agent.
Glycerin is commonly classified as an osmotic laxative but may act additionally or alternatively through its local irritant effects; it may also have lubricating and fecal softening actions. Glycerin suppositories usually work within 15 to 30 minutes.
Cryoprotective Agents
Substances that provide protection against the harmful effects of freezing temperatures. (See all compounds classified as Cryoprotective Agents.)
Solvents
Liquids that dissolve other substances (solutes), generally solids, without any change in chemical composition, as, water containing sugar. (Grant and Hackh's Chemical Dictionary, 5th ed) (See all compounds classified as Solvents.)
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AG - Enemas
A06AG04 - Glycerol
A - Alimentary tract and metabolism
A06 - Drugs for constipation
A06A - Drugs for constipation
A06AX - Other drugs for constipation
A06AX01 - Glycerol
Absorption
Well absorbed orally, poorly absorbed rectally. Studies in humans and animals indicate glycerol is rapidly absorbed in the intestine and the stomach
Route of Elimination
Approx 7-14% of dose is excreted unchanged in the urine within 2.5 hr.
Volume of Distribution
Glycerin is distributed throughout the blood. Although glycerin generally does not appear in ocular fluids, it may enter the orbital sac when the eye is inflamed, with a consequent decrease in osmotic effect.
Data from studies in humans and animals indicate glycerol is rapidly absorbed in the intestine and the stomach, distributed over the extracellular space and excreted.
United Nations Environment Programme: Screening Information Data Sheets on Glycerol (56-81-5) (March 2002) Available from, as of July 14, 2009: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
After hydrolysis of glycerol esters in the intestine, glycerol is readily absorbed.
WHO/FAO: Expert Committee on Food Additives. Summary of Toxicological Data of Certain Food Additives Series 48: Aliphatic acyclic diols, triols, and related substances (56-81-5) (2002). Available from, as of July 14, 2011: https://www.inchem.org/pages/jecfa.html
Following rectal administration, glycerin and sorbitol are poorly absorbed; colonic evacuation of glycerin rectal suppositories or enemas occurs within 15-60 minutes, while colonic evacuation of oral sorbitol occurs within 24-48 hours.
American Society of Health-System Pharmacists 2011; Drug Information 2011. Bethesda, MD. 2011
Following absorbption from GI tract, glycerin is distributed throughout the blood. Although glycerin glycerin generally does not appear in ocular fluids, it may enter the orbital sac when the eye is inflamed, with a consequent decrease in osmotic effect.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1773
For more Absorption, Distribution and Excretion (Complete) data for GLYCERIN (7 total), please visit the HSDB record page.
Glycerin is a substrate for synthesis of triacylglycerols and of phospholipids in the liver and adipose tissue. When fat metabolized as a source of energy, glycerol and fatty acids are released into the bloodstream. Circulating glycerin does not glycate proteins and does not lead to the formation of advanced glycation endproducts (AGEs). In some organisms, the glycerin component can enter the glycolysis pathway directly to provide a substrate for energy or glucose production. Glycerol must be converted to their intermediate glyceraldehyde 3-phosphate before being used in glycolysis or gluconeogenesis. Glycerol metabolism is regulated by the enzymes glycerol kinase, (cytosolic) NAD+-dependent G3P dehydrogenase and (mitochondrial) FAD-linked G3P dehydrogenase.
Glycerol is phosphorylated to alpha-glycerophosphate by glycerol kinase predominantly in the liver (80-90%) and kidneys (10-20%) and incorporated in the standard metabolic pathways to form glucose and glycogen. Glycerol kinase is also found in intestinal mucosa, brown adipose tissue, lymphatic tissue, lung and pancreas. Glycerol may also be combined with free fatty acids in the liver to form triglycerides (lipogenesis) which are distributed to the adipose tissues. The turnover rate is directly proportional to plasma glycerol levels.
United Nations Environment Programme: Screening Information Data Sheets on Glycerol (56-81-5) (March 2002) Available from, as of July 14, 2009: https://www.chem.unep.ch/irptc/sids/OECDSIDS/sidspub.html
Glycerol is endogenous in the human body. It enters the glycolytic pathway after its conversion in the liver to glycerol-3-phosphate by glycerol kinase. Glycerol-3-phosphate is then oxidized by glycerol-3-phosphate dehydrogenase to yield dihydroxyacetone phosphate, which is then isomerized to glyceral-dehyde-3-phosphate, eventually yielding pyruvic acid.
WHO/FAO: Expert Committee on Food Additives. Summary of Toxicological Data of Certain Food Additives Series 48: Aliphatic acyclic diols, triols, and related substances (56-81-5) (2002). Available from, as of July 14, 2011: https://www.inchem.org/pages/jecfa.html
Glycerol esters are hydrolyzed to glycerol and the corresponding carboxylic acids. The hydrolysis is catalysed by intestinal lipase, which attacks the ester bonds at carbons 1 and 3. The ester bond at carbon 2 is more resistant to hydrolysis, possibly because of its stereochemistry and steric hindrance. The beta-monoglyceride can, however, spontaneously isomerise to the alpha-form (3-acylglycerol), permitting further hydrolysis to yield glycerol.
WHO/FAO: Expert Committee on Food Additives. Summary of Toxicological Data of Certain Food Additives Series 48: Aliphatic acyclic diols, triols, and related substances (56-81-5) (2002). Available from, as of July 14, 2011: https://www.inchem.org/pages/jecfa.html
Glycerol, pyruvic acid, and lactic acid are endogenous in humans. Glycerol and pyruvic acid are metabolized completely and are not excreted. ... Glycerol is metabolized via the glycolytic pathway after it has been converted in the liver to glycerol-3-phosphate.
WHO/FAO: Expert Committee on Food Additives. Summary of Toxicological Data of Certain Food Additives Series 48: Aliphatic acyclic diols, triols, and related substances (56-81-5) (2002). Available from, as of July 14, 2011: https://www.inchem.org/pages/jecfa.html
For more Metabolism/Metabolites (Complete) data for GLYCERIN (6 total), please visit the HSDB record page.
30 - 45 minutes
Elimination half-life of glycerin is about 30-40 min.
McEvoy, G.K. (ed.). American Hospital Formulary Service - Drug Information 93. Bethesda, MD: American Society of Hospital Pharmacists, Inc., 1993 (Plus Supplements, 1993)., p. 1773
When administered rectally, glycerin exerts a hygroscopic and/or local irritant action, drawing water from the tissues into the feces and reflexively stimulating evacuation. Glycerin decreases intraocular pressure by creating an osmotic gradient between the blood and intraocular fluid, causing fluid to move out of the aqueous and vitreous humors into the bloodstream.
Glycerin (glycerol) and sorbitol are hyperosmotic laxatives.
American Society of Health-System Pharmacists 2011; Drug Information 2011. Bethesda, MD. 2011
When administered rectally, glycerin and sorbitol exert a hygroscopic and/or local irritant action, drawing water from the tissues into the feces and reflexly stimulating evacuation. The extent to which the simple physical distention of the rectum and the hygroscopic and/or local irritant actions are responsible for the laxative effects of some of these drugs is not known. Only extremely high oral doses of sorbitol (25 g daily) or glycerin exert laxative action.
American Society of Health-System Pharmacists 2011; Drug Information 2011. Bethesda, MD. 2011
/Glycerin/ decreases intraocular pressure by creating an osmotic gradient between the blood and intraocular fluid, causing fluid to move out of the aqueous and vitreous humors into the bloodstream.
Physicians Desk Reference for Ophthalmic Medicines 38th ed. Thomson Reuters. Montvale, NJ 2010.
The physicochemical effects of a series of alkanols, alkanediols and glycerol on erythrocyte shape and hemolysis at 4 and 20 degrees C were examined. We calculated the dielectric constant of the incubation medium, Ds, and the dielectric constant of the erythrocyte membrane Dm in the presence of organic solutes. The ratio Ds/Dm = -38.48 at 20 degrees C defines the normal biconcave shape in a medium without hemolytic agents. A decrease in Ds/Dm favors externalization or internalization with consequent hemolysis. Alkanols and alkanediols convert biconcave erythrocytes into echinocytes, which is accompanied by an increase in the projected surface area. Glycerol converts biconcave erythrocytes into stomatocytes, which was accompanied by a marginal decrease in the projected surface area. Progressive externalization in alkanols and alkanediols or internalization in glycerol resulted in a decrease in the projected surface area and the formation of smooth spheres. The degree of shape change induced was related to the degree of hemolysis and the ratio Ds/Dm. A decrease in temperature reduced both the degree of shape change and hemolysis. .../Thus/ physicochemical toxicity may be a result of a temperature dependent hydrophobic interaction between the organic solutes and the membrane and is best interpreted by the ability of the solutes to change Ds and Dm.
PMID:8634318 Bakaltcheva IB et al; Biochim Biophys Acta 1280 (1): 73-80 (1996)

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 16080
Submission : 2002-08-01
Status : Active
Type : II


GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 26941
Submission : 2013-02-28
Status : Active
Type : IV



GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 32997
Submission : 2018-10-26
Status : Active
Type : IV


GDUFA
DMF Review : Reviewed
Rev. Date : 2025-02-03
Pay. Date : 2024-10-09
DMF Number : 40171
Submission : 2024-10-16
Status : Active
Type : II

Certificate Number : CEP 2024-360 - Rev 00
Issue Date : 2025-10-10
Type : TSE
Substance Number : 496
Status : Valid

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9284
Submission : 1991-08-12
Status : Inactive
Type : II




Registration Number : 217MF10844
Registrant's Address : 1-2-6 Awajicho, Chuo-ku, Osaka City, Osaka Prefecture
Initial Date of Registration : 2005-11-01
Latest Date of Registration :


 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Approved
Registration Country : Sweden
Brand Name : Karbamid/Glycerol Aco
Dosage Form : Cream
Dosage Strength : 20mg/g;200mg/g
Packaging :
Approval Date : 03-05-2021
Application Number : 2.02E+13
Regulatory Info : Approved
Registration Country : Sweden

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Suspended
Registration Country : Spain
Brand Name : Glycilax Children
Dosage Form : Rectal Solution
Dosage Strength : 2.25G
Packaging :
Approval Date : 21-11-2000
Application Number : 63455
Regulatory Info : Suspended
Registration Country : Spain

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Authorized
Registration Country : Spain
Brand Name : Glycilax Children Suppositories
Dosage Form : Suppository
Dosage Strength :
Packaging :
Approval Date : 20-03-2012
Application Number : 75643
Regulatory Info : Authorized
Registration Country : Spain

 Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Boost your online visibility by uploading your products, APIs, FDFs, intermediates, excipients, and services for free on PharmaCompass.
Rank higher among suppliers and expand your reach across the internet efficiently and cost-effectively.
Regulatory Info : Withdrawn
Registration Country : Malta
Glycerol Percent Weight/Weight
Brand Name : Glycerin Suppositories Bp
Dosage Form : Suppository
Dosage Strength : 4G
Packaging :
Approval Date : 2006-10-31
Application Number :
Regulatory Info : Withdrawn
Registration Country : Malta

Regulatory Info :
Registration Country : Italy
Brand Name : Glycerol 10% Sodium Chloride 0.9%
Dosage Form :
Dosage Strength : 1 Bag 500 Ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info :
Registration Country : Italy
Brand Name : Glycerol 10% Sodium Chloride 0.9%
Dosage Form :
Dosage Strength : 1 Bottle 250 Ml
Packaging :
Approval Date :
Application Number :
Regulatory Info :
Registration Country : Italy

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Allowed
Registration Country : Switzerland
Anise Oil; Eucalyptus Oil; Levomenthol; Dry Ethanolic Extract of Polygala Senega Root; Glycerol; Licorice Juice
Brand Name : Chest Bronchial pastilles
Dosage Form : Lozenge
Dosage Strength :
Packaging :
Approval Date : 11/07/2001
Application Number : 54887
Regulatory Info : Allowed
Registration Country : Switzerland

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Allowed
Registration Country : Switzerland
Brand Name : Amavita Glycolax
Dosage Form : Suppository
Dosage Strength : 2250mg
Packaging :
Approval Date : 17/01/2017
Application Number : 66488
Regulatory Info : Allowed
Registration Country : Switzerland

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Authorized
Registration Country : Spain
Brand Name : Rovi Glycerin Suppositories For Infants
Dosage Form : Suppository
Dosage Strength :
Packaging :
Approval Date : 29-09-2023
Application Number : 89066
Regulatory Info : Authorized
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
Regulatory Info : Authorized
Registration Country : Spain
Brand Name : Glycerin Suppositories Cuve Adults
Dosage Form : Suppository
Dosage Strength :
Packaging :
Approval Date : 01-01-1980
Application Number : 53833
Regulatory Info : Authorized
Registration Country : Spain

Portfolio PDF
Product Web Link
Virtual Booth
Digital Content
Website
Corporate PDF
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
29
PharmaCompass offers a list of Glycerine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Glycerine manufacturer or Glycerine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Glycerine manufacturer or Glycerine supplier.
PharmaCompass also assists you with knowing the Glycerine API Price utilized in the formulation of products. Glycerine API Price is not always fixed or binding as the Glycerine Price is obtained through a variety of data sources. The Glycerine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Glycerine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Glycerine, including repackagers and relabelers. The FDA regulates Glycerine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Glycerine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Glycerine manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Glycerine supplier is an individual or a company that provides Glycerine active pharmaceutical ingredient (API) or Glycerine finished formulations upon request. The Glycerine suppliers may include Glycerine API manufacturers, exporters, distributors and traders.
click here to find a list of Glycerine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Glycerine DMF (Drug Master File) is a document detailing the whole manufacturing process of Glycerine active pharmaceutical ingredient (API) in detail. Different forms of Glycerine DMFs exist exist since differing nations have different regulations, such as Glycerine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Glycerine DMF submitted to regulatory agencies in the US is known as a USDMF. Glycerine USDMF includes data on Glycerine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Glycerine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Glycerine suppliers with USDMF on PharmaCompass.
The Pharmaceuticals and Medical Devices Agency (PMDA) established the Japan Drug Master File (JDMF), also known as the Master File (MF), to permit Japanese and foreign manufacturers of drug substances, intermediates, excipients, raw materials, and packaging materials (‘Products’) to voluntarily register confidential information about the production and management of their products in Japan.
The Glycerine Drug Master File in Japan (Glycerine JDMF) empowers Glycerine API manufacturers to present comprehensive information (e.g., production methods, data, etc.) to the review authority, i.e., PMDA (Pharmaceuticals & Medical Devices Agency).
PMDA reviews the Glycerine JDMF during the approval evaluation for pharmaceutical products. At the time of Glycerine JDMF registration, PMDA checks if the format is accurate, if the necessary items have been included (application), and if data has been attached.
click here to find a list of Glycerine suppliers with JDMF on PharmaCompass.
A Glycerine CEP of the European Pharmacopoeia monograph is often referred to as a Glycerine Certificate of Suitability (COS). The purpose of a Glycerine CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Glycerine EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Glycerine to their clients by showing that a Glycerine CEP has been issued for it. The manufacturer submits a Glycerine CEP (COS) as part of the market authorization procedure, and it takes on the role of a Glycerine CEP holder for the record. Additionally, the data presented in the Glycerine CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Glycerine DMF.
A Glycerine CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Glycerine CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Glycerine suppliers with CEP (COS) on PharmaCompass.
Glycerine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Glycerine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Glycerine GMP manufacturer or Glycerine GMP API supplier for your needs.
A Glycerine CoA (Certificate of Analysis) is a formal document that attests to Glycerine's compliance with Glycerine specifications and serves as a tool for batch-level quality control.
Glycerine CoA mostly includes findings from lab analyses of a specific batch. For each Glycerine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Glycerine may be tested according to a variety of international standards, such as European Pharmacopoeia (Glycerine EP), Glycerine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Glycerine USP).

