
Synopsis
Synopsis
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
EDQM
0
USP
0
JP
0
Others
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
FDF
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
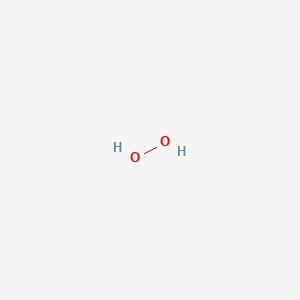

1. Hydrogen Peroxide (h2o2)
2. Hydroperoxide
3. Oxydol
4. Perhydrol
5. Peroxide, Hydrogen
6. Superoxol
1. 7722-84-1
2. Oxydol
3. Perhydrol
4. Superoxol
5. Interox
6. Hydrogen Dioxide
7. Hydroperoxide
8. Inhibine
9. Peroxaan
10. Albone
11. Hioxyl
12. Kastone
13. Albone 35
14. Albone Ds
15. Dihydrogen Dioxide
16. Hydrogen Peroxide Solution
17. T-stuff
18. Lensept
19. Peroxide
20. Elawox
21. H2o2
22. Perone
23. Albone 50
24. Albone 70
25. Perone 50
26. Albone 35cg
27. Albone 50cg
28. Albone 70cg
29. Hydrogen Peroxide (h2o2)
30. Peroxan
31. Hydrogen Peroxide, 30%
32. Hydrogen Dioxide Solution
33. Hydrogen Peroxide, 90%
34. Perone 30
35. Perone 35
36. Hydrogen Peroxide, Solution
37. Waterstofperoxyde
38. Hydrogen Peroxide, 3%
39. Wasserstoffperoxid
40. Caswell No. 486aaa
41. Peroxyde D'hydrogene
42. Perossido Di Idrogeno
43. Asepticper
44. Baquashock
45. Crystacide
46. Peroxclean
47. Ccris 1060
48. Dentasept
49. Hsdb 547
50. Hybrite
51. Metrokur
52. Mirasept
53. Oxigenal
54. Oxyfull
55. Oxysept
56. Pegasyl
57. Eskata
58. Hipox
59. Hyrogen Peroxide
60. Select Bleach
61. Hydrogen Peroxide, Solution, 3%
62. Hydrogen Peroxide Solution (dot)
63. Xtra White
64. Hydrogen Peroxide (conc > 52%)
65. Oxysept I
66. Hydrogen Peroxide Solutions
67. Hydrogen Peroxide, Solution, 30%
68. Hydrogen Peroxide, Solution, 35%
69. Lensan A
70. Odosat D
71. Adeka Super El
72. Crestal Whitestrips
73. Un2014
74. Un2015
75. Un2984
76. Epa Pesticide Chemical Code 000595
77. Anti-keim 50
78. Hydrogen Peroxide, 8% To 20%
79. Hydrogen Peroxide, 20% To 60%
80. Hydrogen Peroxide (> 52% Conc.)
81. Hydrogen Peroxide [usp]
82. Mfcd00011333
83. Nsc-19892
84. Un 2015 (>52%)
85. Bbx060an9v
86. Un 2984 (8%-20%)
87. Un 2014 (20%-52%)
88. Chebi:16240
89. Hydrogen Peroxide Solutions (over 8% But Not Over 60%)
90. Hydrogen Peroxide Solutions (over 60% But Not Over 70%)
91. Hydrogen Peroxide (usp)
92. Peo
93. Dioxidane
94. Peroxides
95. Puresept
96. Hydrogenperoxide
97. Waterstofperoxyde [dutch]
98. Accel Concentrate
99. Wasserstoffperoxid [german]
100. Astri-uc
101. Htp [peroxide]
102. High Test Peroxide
103. Cix
104. Peroxyde D'hydrogene [french]
105. Teat Dip Hp 5
106. Perossido Di Idrogeno [italian]
107. Pre Milk Hp 0.5
108. Pre Milk Hp 1.0
109. Hydrogen Peroxide 35%
110. Einecs 231-765-0
111. Nsc 19892
112. Unii-bbx060an9v
113. Pre Milk Hp 0.5 10
114. Whitespeed
115. A Peroxide
116. Magic Bleaching
117. Clarigel Gold
118. Lase Peroxide
119. Quasar Brite
120. Opalescence Xtra
121. Whiteness Hp
122. Deslime Lp
123. Hydrogen Peroxide (conc >52%)
124. Hydrogen Per Oxide
125. Dihydrogen Peroxide
126. Dihydrogen(peroxide)
127. Oxyfull (tn)
128. Hooh
129. Hydrogen Hydroperoxide
130. Nite White Excel 2
131. Oxydol (jp17)
132. Hydrogen Peroxide 50%
133. Hydrogen-peroxide
134. Oxydol [jan]
135. Hydrogen Peroxide, 50%
136. Hydrogen Peroxide (8ci)
137. Molmap_000025
138. Bis(hydridooxygen)(o--o)
139. Ec 231-765-0
140. 30 %hydrogen Peroxide(aq)
141. 8007-30-5
142. Chembl71595
143. Hydrogen Peroxide 32 Wt. %
144. Oxteril? 350 Spray
145. Hydrogen Peroxide [ii]
146. Hydrogen Peroxide [mi]
147. [oh(oh)]
148. Hydrogen Peroxide [fcc]
149. Dtxsid2020715
150. Hydrogen Peroxide [hsdb]
151. Hydrogen Peroxide [iarc]
152. Hydrogen Peroxide [inci]
153. Chebi:25940
154. Hydrogen Peroxide [vandf]
155. Hydrogen Peroxide [mart.]
156. Hydrogen Peroxide [who-dd]
157. Hydrogen Peroxide (h2o2) (9ci)
158. Nsc19892
159. Wln: H2 O2 90%
160. Hydrogen Peroxide [topical Solution]
161. Akos015856794
162. Hydrogen Peroxide [green Book]
163. Db11091
164. Hydrogen Peroxide [orange Book]
165. Hydrogen Peroxide [usp Impurity]
166. Qtl1_000041
167. Hydrogen Peroxide, Aqueous Solutions With >40% But Not >60% Hydrogen Peroxide (stabilized As Necessary) [un2014] [oxidizer]
168. Hydrogen Peroxide, Aqueous Solutions With Not <20% But Not >40% Hydrogen Peroxide (stabilized As Necessary) [un2014] [oxidizer]
169. Hydrogen Peroxide, Aqueous Solutions With Not <8% But <20% Hydrogen Peroxide (stabilized As Necessary) [un2984] [oxidizer]
170. Hydrogen Peroxide, Stabilized Or Hydrogen Peroxide Aqueous Solutions, Stabilized With >60% Hydrogen Peroxide [un2015] [oxidizer]
171. Ft-0627133
172. H1222
173. Hydrogen Peroxide In Aqueous Solution
174. C00027
175. D00008
176. Hydrogen Peroxide Solution, 3%, For Microbiology
177. Hydrogen Peroxide Solution, For Ultratrace Analysis
178. Q171877
179. Q1088474
180. Hydrogen Peroxide Solution, Extra Pure, 30.0-32.0%
181. Hydrogen Peroxide Solution, Purum P.a., >=30% (rt)
182. Hydrogen Peroxide Solution, Purum P.a., >=35% (rt)
183. Hydrogen Peroxide Solution, >=30%, For Trace Analysis
184. Hydrogen Peroxide Solution, Saj First Grade, >=30.0%
185. Hydrogen Peroxide Solution, Tested According To Ph.eur.
186. Hydrogen Peroxide Solution, 50 Wt. % In H2o, Stabilized
187. Hydrogen Peroxide Solution, Jis Special Grade, 30.0-35.5%
188. Hydrogen Peroxide Solution, 30 % (w/w) In H2o, Contains Stabilizer
189. Hydrogen Peroxide Solution, Contains Inhibitor, 35 Wt. % In H2o
190. Hydrogen Peroxide Solution, P.a., Acs Reagent, Reag. Iso, 30.0%
191. Hydrogen Peroxide 30%, Meets The Analytical Specifications Of Ph. Eur., Stabilized
192. Hydrogen Peroxide Solution, 30% (w/w), Puriss. P.a., Reag. Iso, Reag. Ph. Eur.
193. Hydrogen Peroxide Solution, Ar, Contains Sodium Pyrophosphate As Stabilizer, >=30 % (w/v)
194. Hydrogen Peroxide Solution, Contains ~200 Ppm Acetanilide As Stabilizer, 3 Wt. % In H2o
195. Hydrogen Peroxide Solution, Contains Inhibitor, 30 Wt. % In H2o, Acs Reagent
196. Hydrogen Peroxide Solution, Lr, Contains Sodium Pyrophosphate As Stabilizer, >=30 % (w/v)
197. Hydrogen Peroxide Solution, Lr, Contains Sodium Pyrophosphate As Stabilizer, >=50 % (w/v)
198. Hydrogen Peroxide Solution, Meets Analytical Specification Of Ph. Nord., 34.5-36.5%
199. Hydrogen Peroxide Solution, Puriss. P.a., Acs Reagent, Not Stabilized, >=30% (rt)
200. Hydrogen Peroxide Solution, Semiconductor Grade Mos Puranal(tm) (honeywell 17937), >=30%
201. Hydrogen Peroxide Solution, Semiconductor Grade Puranal(tm) (honeywell 17948), >=30%
202. Hydrogen Peroxide Solution, Semiconductor Grade Ulsi Puranal(tm) (honeywell 17024), >=30%
203. Hydrogen Peroxide Solution, Semiconductor Grade Vlsi Puranal(tm) (honeywell 17606), >=30%
204. Hydrogen Peroxide Solution, Contains Inhibitor, 30 Wt. % In H2o, Meets Usp Testing Specifications
205. Hydrogen Peroxide Solution, Contains Potassium Stannate As Inhibitor, 30-32 Wt. % In Water, Semiconductor Grade, 99.999% Trace Metals Basis
206. Hydrogen Peroxide, Aqueous Solutions With >40% But Not >60% Hydrogen Peroxide (stabilized As Necessary)
207. Hydrogen Peroxide, Aqueous Solutions With >40% But Not >60% Hydrogen Peroxide (stabilized As Necessary) [un2014] [oxidizer]
208. Hydrogen Peroxide, Aqueous Solutions With Not <20% But Not >40% Hydrogen Peroxide (stabilized As Necessary)
209. Hydrogen Peroxide, Aqueous Solutions With Not <20% But Not >40% Hydrogen Peroxide (stabilized As Necessary) [un2014] [oxidizer]
210. Hydrogen Peroxide, Aqueous Solutions With Not <8% But <20% Hydrogen Peroxide (stabilized As Necessary)
211. Hydrogen Peroxide, Aqueous Solutions With Not <8% But <20% Hydrogen Peroxide (stabilized As Necessary) [un2984] [oxidizer]
212. Hydrogen Peroxide, Stabilized Or Hydrogen Peroxide Aqueous Solutions, Stabilized With >60% Hydrogen Peroxide
213. Hydrogen Peroxide, Stabilized Or Hydrogen Peroxide Aqueous Solutions, Stabilized With >60% Hydrogen Peroxide [un2015] [oxidizer]
| Molecular Weight | 34.015 g/mol |
|---|---|
| Molecular Formula | H2O2 |
| XLogP3 | -0.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 0 |
| Exact Mass | 34.005479302 g/mol |
| Monoisotopic Mass | 34.005479302 g/mol |
| Topological Polar Surface Area | 40.5 Ų |
| Heavy Atom Count | 2 |
| Formal Charge | 0 |
| Complexity | 0 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Anti-Infective Agents, Local; Oxidants
National Library of Medicine's Medical Subject Headings. Hydrogen peroxide. Online file (MeSH, 2017). Available from, as of October 2, 2017: https://www.nlm.nih.gov/mesh/2017/mesh_browser/MBrowser.html
/CLINICAL TRIALS/ ClinicalTrials.gov is a registry and results database of publicly and privately supported clinical studies of human participants conducted around the world. The Web site is maintained by the National Library of Medicine (NLM) and the National Institutes of Health (NIH). Each ClinicalTrials.gov record presents summary information about a study protocol and includes the following: Disease or condition; Intervention (for example, the medical product, behavior, or procedure being studied); Title, description, and design of the study; Requirements for participation (eligibility criteria); Locations where the study is being conducted; Contact information for the study locations; and Links to relevant information on other health Web sites, such as NLM's MedlinePlus for patient health information and PubMed for citations and abstracts for scholarly articles in the field of medicine. Hydrogen peroxide is included in the database.
NIH/NLM; ClinicalTrials.Gov. Available from, as of October 2, 2017: https://clinicaltrials.gov/
Antiseptic; disinfectant.
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Cambridge, UK: Royal Society of Chemistry, 2013., p. 889
Hydrogen peroxide 3% topical solution is used to cleanse minor cuts or skin abrasions. More potent solutions (e.g., 20-30%) have been used as a hair bleach.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
For more Therapeutic Uses (Complete) data for Hydrogen peroxide (14 total), please visit the HSDB record page.
The US Food and Drug Administration (FDA) has alerted consumers that high strength hydrogen peroxide preparations (e.g., 35% food grade hydrogen peroxide) should not be used for any medicinal purpose. These preparations are being promoted on websites illegally for various medicinal purposes (e.g., AIDS, cancer, emphysema, other life-threatening conditions) without any proven clinical value, and such uses are dangerous. Hydrogen peroxide 35% is not approved by the FDA for any purpose. Ingestion of such preparations can cause serious harm or death. Oral ingestion of hydrogen peroxide can result in GI irritation and ulceration. IV administration of hydrogen peroxide can result in inflammation at the injection site, gas embolism, and life-threatening allergic reactions.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Concentrated solutions (20-30% or more) of hydrogen peroxide are strongly irritating to skin or mucous membranes and should be handled cautiously.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Indicated to be used as a disinfectant and sterilizer.
Treatment of seborrhoeic keratosis
Treatment of common warts (verrucae vulgaris)
Hydrogen peroxide exhibits antimicrobial properties against most forms of microorganisms, including dormant forms with known high resistance profiles, such as bacterial spores and protozoal cysts. It acts as an oxidative biocide to generate free radical species to induce DNA, protein and membrane lipid damage via oxidation.
Anti-Infective Agents, Local
Substances used on humans and other animals that destroy harmful microorganisms or inhibit their activity. They are distinguished from DISINFECTANTS, which are used on inanimate objects. (See all compounds classified as Anti-Infective Agents, Local.)
Oxidants
Electron-accepting molecules in chemical reactions in which electrons are transferred from one molecule to another (OXIDATION-REDUCTION). (See all compounds classified as Oxidants.)
A - Alimentary tract and metabolism
A01 - Stomatological preparations
A01A - Stomatological preparations
A01AB - Antiinfectives and antiseptics for local oral treatment
A01AB02 - Hydrogen peroxide
D - Dermatologicals
D08 - Antiseptics and disinfectants
D08A - Antiseptics and disinfectants
D08AX - Other antiseptics and disinfectants
D08AX01 - Hydrogen peroxide
D - Dermatologicals
D11 - Other dermatological preparations
D11A - Other dermatological preparations
D11AX - Other dermatologicals
D11AX25 - Hydrogen peroxide
S - Sensory organs
S02 - Otologicals
S02A - Antiinfectives
S02AA - Antiinfectives
S02AA06 - Hydrogen peroxide
Absorption
It is reported that hydrogen peroxide is decomposed before absorption in the intestine. Solutions of hydrogen peroxide displays poor penetration when applied to tissue.
Volume of Distribution
Target organs affected by hydrogen peroxide include the lungs, intestine, thymus, liver, and kidney.
This in vivo study determined the kinetics of 3% hydrogen peroxide in a bleaching gel within the first hour. The material used in this study was 3% hydrogen peroxide gel and the study involved 10 subjects who met the inclusion and exclusion criteria. Each subject wore the tray with gel six different times on separate days. Evaluation of the remaining amount of hydrogen peroxide was calculated by the method stated in US Pharmacopoeia. The study results indicate that the mean percentage of hydrogen peroxide recovered for 5, 10, 20, 30, 45 and 60 minutes was 61, 56, 49, 44, 38 and 32, respectively. The amount of hydrogen peroxide in the saliva sample after one hour was 0.42 mg. Excluding the first 10 minutes, the kinetics of hydrogen peroxide in the tray and teeth sample was exponential.
PMID:12760694 Al-Qunaian TA et al; Oper Dent 28 (3): 236-41 (2003)
Hydrogen peroxide is reduced by glutathione peroxidase, which is an endogenous enzyme in human tissue. It is rapidly decomposed to oxygen and water when in contact with catalase, an enzyme found in blood and most tissues.
The production of free hydroxyl radicals in the Fenton reaction is thought to be the basis of biocidal actions of hydrogen peroxide. Free radicals eventually lead to oxidative damage proteins and membrane lipids _in vivo_. The oxidizing radical as the ferryl radical induces DNA oxidation.
Hydrogen peroxide topical solution is a weak antibacterial agent, a wound cleanser, and a deodorant. The pharmacologic activity of the drug depends on the release of nascent oxygen which has a powerful oxidizing effect that destroys some microorganisms and chemically alters many organic substances. When hydrogen peroxide topical solution comes in contact with tissues that contain the enzyme catalase, the solution releases oxygen which exerts antibacterial action; the mechanical effect of effervescence loosens tissue debris and pus. The release of nascent oxygen and effervescence is more rapid on wounds, denuded areas, and mucous membranes than on unbroken skin. The presence of reactive organic material such as pus and blood diminishes the efficiency of hydrogen peroxide. The antibacterial activity of hydrogen peroxide is relatively weak and slow and the drug exhibits poor tissue and wound penetration. Hydrogen peroxide's mechanical effect of effervescence and resultant removal of tissue debris is probably a more effective means of reducing the bacterial content of wounds, denuded areas, and mucous membranes than actual antibacterial activity. The drug also appears to have a styptic effect when applied topically to minor wounds. Concentrated solutions of hydrogen peroxide have a bleaching effect on hair and may injure tissue.
American Society of Health-System Pharmacists 2017; Drug Information 2017. Bethesda, MD. 2017
Increases in the levels of reactive oxygen species (ROS) are correlated with a decrease in calcineurin (CN) activity under oxidative or neuropathological conditions. However, the molecular mechanism underlying this ROS-mediated CN inactivation remains unclear. Here, we describe a mechanism for the inactivation of CN by hydrogen peroxide. The treatment of mouse primary cortical neuron cells with Abeta(1-42) peptide and hydrogen peroxide triggered the proteolytic cleavage of CN and decreased its enzymatic activity. In addition, hydrogen peroxide was found to cleave CN in different types of cells. Calcium influx was not involved in CN inactivation during hydrogen peroxide-mediated cleavage, but CN cleavage was partially blocked by chloroquine, indicating that an unidentified lysosomal protease is probably involved in its hydrogen peroxide-mediated cleavage. Treatment with hydrogen peroxide triggered CN cleavage at a specific sequence within its catalytic domain, and the cleaved form of CN had no enzymatic ability to dephosphorylate nuclear factor in activated T cells. Thus, our findings suggest a molecular mechanism by which hydrogen peroxide inactivates CN by proteolysis in ROS-related diseases.
PMID:17217415 Lee JE et al; J Neurochem 100 (6): 1703-12 (2007)
Matrix metalloproteinase-2 (MMP-2) is well known to proteolyse both extracellular and intracellular proteins. Reactive oxygen species activate MMP-2 at both transcriptional and post-translational levels, thus MMP-2 activation is considered an early event in oxidative stress injury. Although hydrogen peroxide is widely used to trigger oxidative stress-induced cell death, the type of cell death (apoptosis vs. necrosis) in cardiomyocytes is still controversial depending on the concentration used and the exposure time. We ... investigated the mode of cell death in neonatal rat cardiomyocytes induced by different concentrations (50-500 uM) of hydrogen peroxide at various time intervals after exposure and determined whether MMP-2 is implicated in hydrogen peroxide-induced cardiomyocyte death. Treating cardiomyocytes with hydrogen peroxide led to elevated MMP-2 level/activity with maximal effects seen at 200 uM. Hydrogen peroxide caused necrotic cell death by disrupting the plasmalemma as evidenced by the release of lactate dehydrogenase in a concentration- and time-dependent manner as well as the necrotic cleavage of PARP-1. The absence of both caspase-3 cleavage/activation and apoptotic cleavage of PARP-1 illustrated the weak contribution of apoptosis. Pre-treatment with selective MMP inhibitors did not protect against hydrogen peroxide-induced necrosis. In conclusion hydrogen peroxide increases MMP-2 level/activity in cardiomyocytes and induces necrotic cell death, however, the later effect is MMP-2 independent.
PMID:23665313 Ali MA et al; Toxicol In Vitro 27 (6): 1686-92 (2013)

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Excipients by Applications
Market Place

Patents & EXCLUSIVITIES
ABOUT THIS PAGE
83
PharmaCompass offers a list of Hydrogen Peroxide API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Hydrogen Peroxide manufacturer or Hydrogen Peroxide supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Hydrogen Peroxide manufacturer or Hydrogen Peroxide supplier.
PharmaCompass also assists you with knowing the Hydrogen Peroxide API Price utilized in the formulation of products. Hydrogen Peroxide API Price is not always fixed or binding as the Hydrogen Peroxide Price is obtained through a variety of data sources. The Hydrogen Peroxide Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Hydrogen Peroxide manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Hydrogen Peroxide, including repackagers and relabelers. The FDA regulates Hydrogen Peroxide manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Hydrogen Peroxide API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
click here to find a list of Hydrogen Peroxide manufacturers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PhamaCompass.
A Hydrogen Peroxide supplier is an individual or a company that provides Hydrogen Peroxide active pharmaceutical ingredient (API) or Hydrogen Peroxide finished formulations upon request. The Hydrogen Peroxide suppliers may include Hydrogen Peroxide API manufacturers, exporters, distributors and traders.
click here to find a list of Hydrogen Peroxide suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Hydrogen Peroxide DMF (Drug Master File) is a document detailing the whole manufacturing process of Hydrogen Peroxide active pharmaceutical ingredient (API) in detail. Different forms of Hydrogen Peroxide DMFs exist exist since differing nations have different regulations, such as Hydrogen Peroxide USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Hydrogen Peroxide DMF submitted to regulatory agencies in the US is known as a USDMF. Hydrogen Peroxide USDMF includes data on Hydrogen Peroxide's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Hydrogen Peroxide USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Hydrogen Peroxide suppliers with USDMF on PharmaCompass.
A Hydrogen Peroxide CEP of the European Pharmacopoeia monograph is often referred to as a Hydrogen Peroxide Certificate of Suitability (COS). The purpose of a Hydrogen Peroxide CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Hydrogen Peroxide EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Hydrogen Peroxide to their clients by showing that a Hydrogen Peroxide CEP has been issued for it. The manufacturer submits a Hydrogen Peroxide CEP (COS) as part of the market authorization procedure, and it takes on the role of a Hydrogen Peroxide CEP holder for the record. Additionally, the data presented in the Hydrogen Peroxide CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Hydrogen Peroxide DMF.
A Hydrogen Peroxide CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Hydrogen Peroxide CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Hydrogen Peroxide suppliers with CEP (COS) on PharmaCompass.
Hydrogen Peroxide Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Hydrogen Peroxide GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Hydrogen Peroxide GMP manufacturer or Hydrogen Peroxide GMP API supplier for your needs.
A Hydrogen Peroxide CoA (Certificate of Analysis) is a formal document that attests to Hydrogen Peroxide's compliance with Hydrogen Peroxide specifications and serves as a tool for batch-level quality control.
Hydrogen Peroxide CoA mostly includes findings from lab analyses of a specific batch. For each Hydrogen Peroxide CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Hydrogen Peroxide may be tested according to a variety of international standards, such as European Pharmacopoeia (Hydrogen Peroxide EP), Hydrogen Peroxide JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Hydrogen Peroxide USP).

