

Synopsis
Synopsis
0
VMF
0
South Africa

0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
Annual Reports
NA
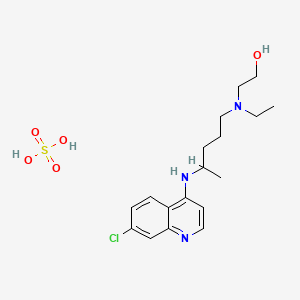

1. Hydroxychlorochin
2. Hydroxychloroquine
3. Hydroxychloroquine Sulfate (1:1) Salt
4. Oxychlorochin
5. Oxychloroquine
6. Plaquenil
1. 747-36-4
2. Hydroxychloroquine Sulphate
3. Ercoquin
4. Plaquenil
5. 2-((4-((7-chloroquinolin-4-yl)amino)pentyl)(ethyl)amino)ethanol Sulfate
6. Quensyl
7. Oxiklorin
8. Tcmdc-123987
9. Hcq Sulfate
10. Plaquenil Sulfate
11. Nsc 4375
12. Hydroxychloroquine (sulfate)
13. Oxichlorochine Sulfate
14. Nsc-4375
15. 8q2869cnvh
16. 2-[4-[(7-chloroquinolin-4-yl)amino]pentyl-ethylamino]ethanol;sulfuric Acid
17. 2-({4-[(7-chloroquinolin-4-yl)amino]pentyl}(ethyl)amino)ethan-1-ol; Sulfuric Acid
18. Plaquinol
19. Toremonil
20. 2-((4-((7-chloroquinolin-4-yl)amino)pentyl)(ethyl)amino)ethan-1-ol Sulfate
21. Dsstox_cid_27788
22. Dsstox_rid_82563
23. N4-(7-chloro-4-quinolyl)-n1-ethyl-n1-(2-hydroxyethyl)-1,4-pentanediamine Sulfate
24. Dsstox_gsid_47811
25. 2-((4-(7-chloroquinolin-4-ylamino)pentyl)(ethyl)amino)ethanol Sulfate
26. 2-((4-((7-chloroquinolin-4-yl)amino)-pentyl)(ethyl)amino)ethanol Sulfate
27. Cas-747-36-4
28. Hydroxychloroquine Sulfate [usp]
29. Ethanol, 2-((4-((7-chloro-4-quinolinyl)amino)pentyl)ethylamino)-, Sulfate (1:1)
30. Sr-05000001881
31. Ncgc00159483-02
32. Einecs 212-019-3
33. Unii-8q2869cnvh
34. Quinoric
35. Ai3-52706
36. Plaquenil (tn)
37. Ethanol, Monosulfate
38. Mfcd00078203
39. Ethanol, 2-[[4-[(7-chloro-4-quinolinyl)amino]pentyl]ethylamino]-, Sulfate (1:1)
40. Hydroxychlorquine Sulfate
41. 2-((4-((7-chloro-4-quinolinyl)amino)pentyl)ethylamino)ethanol Sulfate (1:1) (salt)
42. 2-((4-((7-chloro-4-quinolyl)amino)pentyl)ethylamino)ethanol Sulfate (1:1) (salt)
43. Chembl1690
44. Ethanol, 2-((4-((7-chloro-4-quinolyl)amino)pentyl)ethylamino)-, Sulfate (1:1) (salt)
45. Schembl41114
46. Spectrum1503978
47. 747-36-4 (sulfate)
48. Dtxsid1047811
49. Hydroxychloroquine Sulfate ,(s)
50. Hydroxychloroquine Sulfate- Bio-x
51. Ethanol, Sulfate (1:1) (salt)
52. Hms1922o12
53. Hms2093o05
54. Hms3713j22
55. Pharmakon1600-01503978
56. Bcp12823
57. Hy-b1370
58. Tox21_111707
59. Bdbm50247975
60. Ccg-39154
61. Nsc758663
62. S4430
63. Hydroxychloroquine Sulfate (jan/usp)
64. Akos015897337
65. Tox21_111707_1
66. Cs-8017
67. Hydroxychloroquine Sulfate [mi]
68. Ks-5311
69. Nsc-758663
70. Sb19140
71. (+-)-2-((4-((7-chloro-4-quinolyl)amino)pentyl)ethylamino)ethanol Sulfate (1:1) (salt)
72. 7-chloro-4-[4-[n-ethyl-n-(2-hydroxyethyl)amino]-1-methylbutylamino]quinoline Sulfate
73. Hydroxychloroquine Sulfate [jan]
74. Ncgc00095090-01
75. Ncgc00095090-02
76. Ncgc00159483-04
77. Bh164525
78. Ethanol, 2-((4-((7-chloro-4-quinolinyl)amino)pentyl)ethyl)amino-, (+-)-, Sulfate (1:1) Salt
79. Hydroxychloroquine Sulfate [mart.]
80. Hydroxychloroquine Sulfate [vandf]
81. Hydroxychloroquine Sulfate [usp-rs]
82. Hydroxychloroquine Sulfate [who-dd]
83. Db-055904
84. Ft-0603511
85. Ft-0669460
86. H1306
87. Hydroxycloroquine Sulfate, Analytical Standard
88. D02114
89. H10433
90. Hydroxychloroquine Sulfate [orange Book]
91. Hydroxychloroquine Sulfate [ep Monograph]
92. 747h364
93. A838213
94. Hydroxychloroquine Sulfate [usp Monograph]
95. Hydroxychloroquine Sulfate, >=98% (hplc), Powder
96. Sr-05000001881-1
97. Sr-05000001881-2
98. W-104413
99. Q27270879
100. Z1551900864
101. Hydroxychloroquine Sulphate 1.0 Mg/ml In Methanol (as Free Base)
102. 2-[[4-[(7-chloro-4-quinolinyl)amino]pentyl]ethylamino]ethanol Sulfate
103. Hydroxychloroquine Sulfate, Pharmaceutical Secondary Standard; Certified Reference Material
104. Hydroxychloroquine Sulfate, United States Pharmacopeia (usp) Reference Standard
105. (+/-)-2-((4-((7-chloro-4-quinolyl)amino)pentyl)ethylamino)ethanol Sulfate (1:1) (salt)
106. (+/-)-2-((4-((7-chloro-4-quinolyl)amino)pentyl)ethylamino)ethanol Sulphate (1:1) (salt)
107. 14480-75-2
108. Ethanol, 2-((4-((7-chloro-4-quinolinyl)amino)pentyl)ethyl)amino-, (+/-)-, Sulfate (1:1) Salt
109. Ethanol, 2-((4-((7-chloro-4-quinolinyl)amino)pentyl)ethyl)amino-, (+/-)-, Sulphate (1:1) Salt
| Molecular Weight | 434.0 g/mol |
|---|---|
| Molecular Formula | C18H28ClN3O5S |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 9 |
| Exact Mass | 433.1438199 g/mol |
| Monoisotopic Mass | 433.1438199 g/mol |
| Topological Polar Surface Area | 131 Ų |
| Heavy Atom Count | 28 |
| Formal Charge | 0 |
| Complexity | 413 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
| 1 of 2 | |
|---|---|
| Drug Name | Hydroxychloroquine sulfate |
| Drug Label | Hydroxychloroquine sulfate, USP is a colorless crystalline solid, soluble in water to at least 20 percent; chemically the drug is 2-[[4-[(7-Chloro-4-quinolyl) amino]pentyl] ethylamino] ethanol sulfate (1:1). Hydroxychloroquine sulfate, USP has the fo... |
| Active Ingredient | Hydroxychloroquine sulfate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg |
| Market Status | Prescription |
| Company | Sandoz; Hikma Pharms; Teva Pharms; Ipca Labs; Zydus Pharms Usa; Mylan |
| 2 of 2 | |
|---|---|
| Drug Name | Hydroxychloroquine sulfate |
| Drug Label | Hydroxychloroquine sulfate, USP is a colorless crystalline solid, soluble in water to at least 20 percent; chemically the drug is 2-[[4-[(7-Chloro-4-quinolyl) amino]pentyl] ethylamino] ethanol sulfate (1:1). Hydroxychloroquine sulfate, USP has the fo... |
| Active Ingredient | Hydroxychloroquine sulfate |
| Dosage Form | Tablet |
| Route | Oral |
| Strength | 200mg |
| Market Status | Prescription |
| Company | Sandoz; Hikma Pharms; Teva Pharms; Ipca Labs; Zydus Pharms Usa; Mylan |
Antimalarials
Agents used in the treatment of malaria. They are usually classified on the basis of their action against plasmodia at different stages in their life cycle in the human. (From AMA, Drug Evaluations Annual, 1992, p1585) (See all compounds classified as Antimalarials.)
Enzyme Inhibitors
Compounds or agents that combine with an enzyme in such a manner as to prevent the normal substrate-enzyme combination and the catalytic reaction. (See all compounds classified as Enzyme Inhibitors.)
Antirheumatic Agents
Drugs that are used to treat RHEUMATOID ARTHRITIS. (See all compounds classified as Antirheumatic Agents.)
 Click Us!
Click Us! 
GDUFA
DMF Review : Reviewed
Rev. Date : 2022-02-16
Pay. Date : 2022-02-11
DMF Number : 20692
Submission : 2007-07-17
Status : Active
Type : II

NDC Package Code : 45542-1161
Start Marketing Date : 2018-08-31
End Marketing Date : 2026-05-09
Dosage Form (Strength) : POWDER (40kg/40kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Samoh Pharmaceutical Co., Ltd.
Registration Date : 2021-03-10
Registration Number : 20210310-209-J-787
Manufacturer Name : EUROAPI Hungary Ltd.
Manufacturer Address : To utca 1-5, Budapest, 1045, Hungary

| Available Reg Filing : ASMF, RU |

GDUFA
DMF Review : Reviewed
Rev. Date : 2016-02-26
Pay. Date : 2016-01-21
DMF Number : 30014
Submission : 2015-11-24
Status : Active
Type : II

Certificate Number : CEP 2017-176 - Rev 02
Issue Date : 2024-01-03
Type : Chemical
Substance Number : 2849
Status : Valid

Registration Number : 305MF10090
Registrant's Address : Koivu-Mankkaan tie 6A, FI-02200 Espoo, Finland
Initial Date of Registration : 2023-08-02
Latest Date of Registration : --

NDC Package Code : 12780-2771
Start Marketing Date : 2016-01-12
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT
 Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Date of Issue : 2023-02-14
Valid Till : 2024-02-26
Written Confirmation Number : WC-0494
Address of the Firm :
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.

GDUFA
DMF Review : Reviewed
Rev. Date : 2015-09-11
Pay. Date : 2015-09-08
DMF Number : 8619
Submission : 1990-06-27
Status : Active
Type : II

Certificate Number : CEP 2017-297 - Rev 03
Issue Date : 2024-08-28
Type : Chemical
Substance Number : 2849
Status : Valid

NDC Package Code : 68108-0336
Start Marketing Date : 2012-12-02
End Marketing Date : 2025-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

| Available Reg Filing : CA, CN |

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 34940
Submission : 2022-01-26
Status : Active
Type : II

Certificate Number : R0-CEP 2021-317 - Rev 00
Issue Date : 2023-02-16
Type : Chemical
Substance Number : 2849
Status : Valid


GDUFA
DMF Review : Reviewed
Rev. Date : 2020-06-22
Pay. Date : 2020-06-11
DMF Number : 30825
Submission : 2016-08-01
Status : Active
Type : II

Certificate Number : R0-CEP 2022-022 - Rev 00
Issue Date : 2023-06-26
Type : Chemical
Substance Number : 2849
Status : Valid

Registration Number : 305MF10077
Registrant's Address : No. 4 HUAZHONG ROAD, CHONGQING (CHANGSHOU) CHEMICAL INDUSTRIAL PARK
Initial Date of Registration : 2023-07-05
Latest Date of Registration : --

NDC Package Code : 67680-003
Start Marketing Date : 2017-11-21
End Marketing Date : 2024-12-31
Dosage Form (Strength) : POWDER (1kg/kg)
Marketing Category : BULK INGREDIENT

Registrant Name : Daeshin Pharmaceutical Co., Ltd.
Registration Date : 2021-05-11
Registration Number : 20210511-209-J-984
Manufacturer Name : Chongqing Kangle Pharmaceutical Co.,Ltd.
Manufacturer Address : No.4, Huazhong Road, Chongqing(Changshou) Chemical and Industry Park, China

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
GDUFA
DMF Review : Complete
Rev. Date : 2022-02-16
Pay. Date : 2022-02-11
DMF Number : 20692
Submission : 2007-07-17
Status : Active
Type : II
GDUFA
DMF Review : Complete
Rev. Date : 2016-02-26
Pay. Date : 2016-01-21
DMF Number : 30014
Submission : 2015-11-24
Status : Active
Type : II
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
GDUFA
DMF Review : Complete
Rev. Date : 2015-09-11
Pay. Date : 2015-09-08
DMF Number : 8619
Submission : 1990-06-27
Status : Active
Type : II
GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 8487
Submission : 1990-03-22
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 12762
Submission : 1997-11-12
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21567
Submission : 2008-04-25
Status : Active
Type : II

GDUFA
DMF Review : Complete
Rev. Date : 2013-07-31
Pay. Date : 2013-07-17
DMF Number : 14087
Submission : 1999-04-17
Status : Active
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 9490
Submission : 1991-12-06
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 21115
Submission : 2008-02-06
Status : Inactive
Type : II

GDUFA
DMF Review : N/A
Rev. Date :
Pay. Date :
DMF Number : 14977
Submission : 2000-08-01
Status : Active
Type : II

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
About the Company : EUROAPI is the market leader in small molecule APIs with projected sales of about €1 billion in 2022. With around 200 APIs, it has one of the largest portfolios in the market. Th...
About the Company : Fermion is fully owned subsidiary of Orion Corporation & headquartered in Espoo, Finland. Together with Orion we are a fully integrated CDMO & offer services covering both APIs & F...
About the Company : Established in 1997, Temad Co. is one of the largest producers of APIs in Iran & an innovative manufacturer of narcotic and non-narcotic products of world-class quality in the Midd...
About the Company : Jai Radhe Sales was founded in 1999 as an out-of-the-box distribution firm specializing in the global supply of high-quality pharmaceutical ingredients. The firm provides complete ...
About the Company : HRV Global is a leading global manufacturer, seller & exporter of a wide range of APIs, advanced intermediates, pellets, food grade chemicals, food additives & food ingredients. It...
 Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
Tagoor's product development expertise, backed by our comprehensive understanding of the processes, helps us offer high-quality APIs.
About the Company : Tagoor Laboratories, established in 2018, is a part of the Tagoor Group. It specializes in providing APIs, advanced intermediates and key starting materials for critical and high-g...
About the Company : Porton Pharma Solutions Ltd. was founded in 2005, & the company's stock was successfully listed in Shenzhen Stock Exchange in 2014. Our R&D, manufacturing & operation facilities ar...
 SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
SCI Pharmtech offers high-quality, cost-effective APIs, advanced intermediates, & custom products with global expertise and precision.
About the Company : SCI Pharmtech Inc. offers APIs, advanced intermediates, and custom products, focusing on quick development and cost-effective production. Our research labs, pilot plants, and produ...
About the Company : Mangalam Drugs & Organics Limited started manufacturing APIs and intermediates in 1977 in Gujarat, India. It has a multi-product manufacturing facility and an in-house R&D lab reco...

About the Company : Symed Labs Ltd is a leading Hyderabad based manufacturer of Active Pharmaceutical Ingredients (APIs) and an end-to-end solution provider for the pharmaceutical industry for chemist...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
DRUG PRODUCT COMPOSITIONS

Related Excipient Companies

Excipients by Applications
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
We have 8 companies offering Hydroxychloroquine Sulfate
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
