
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
VMF
0
Listed Suppliers
0
Canada
0
Australia
0
South Africa
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz

1. 123i-labeled Idoxuridine
2. 125i-labeled Idoxuridine
3. 131i-labeled Idoxuridine
4. 3h-labeled Idoxuridine
5. 5 Iodo 2' Deoxyuridine
6. 5 Iododeoxyuridine
7. 5-iodo-2'-deoxyuridine
8. 5-iododeoxyuridine
9. Allergan 211
10. Herplex Liquifilm
11. Idoxuridine, 123i Labeled
12. Idoxuridine, 123i-labeled
13. Idoxuridine, 125i Labeled
14. Idoxuridine, 125i-labeled
15. Idoxuridine, 131i Labeled
16. Idoxuridine, 131i-labeled
17. Idoxuridine, 3h Labeled
18. Idoxuridine, 3h-labeled
19. Idoxuridine, Radical Ion (+1)
20. Idoxuridine, Radical Ion (1-)
21. Iododeoxyuridine
22. Iudr
23. Kerecide
24. Liquifilm, Herplex
25. Nsc 39661
26. Nsc-39661
27. Nsc39661
28. Oftan Idu
29. Oftan-idu
30. Oftanidu
31. Sk And F-14287
32. Stoxil
1. 5-iodo-2'-deoxyuridine
2. 54-42-2
3. 5-iododeoxyuridine
4. Idoxuridin
5. Iododeoxyridine
6. Iodoxuridine
7. Joddeoxiuridin
8. 2'-deoxy-5-iodouridine
9. Herplex
10. Stoxil
11. Allergan 211
12. Iudr
13. (+)-5-iodo-2'-deoxyuridine
14. 5iudr
15. Idur
16. Idoxuridinum
17. Virudox
18. Dendrid
19. 5-iudr
20. Idu
21. Idoxuridina
22. Allergan 201
23. 5-iodouracil Deoxyriboside
24. Idossuridina [dcit]
25. Idoxene
26. Iduridin
27. Kerecid
28. 1-(2-deoxy-beta-d-ribofuranosyl)-5-iodouracil
29. Iododeoxyuridine
30. Sk&f-14287
31. Allergan-211
32. 5idu
33. 1beta-d-2'-deoxyribofuranosyl-5-iodouracil
34. Nsc-39661
35. 1-beta-d-2'-deoxyribofuranosyl-5-iodouracil
36. Sk&f 14287
37. Uridine, 2'-deoxy-5-iodo-
38. Chebi:147675
39. Nsc 39661
40. (+)-5-iodo-2-deoxyuridine
41. 1-((2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)-5-iodopyrimidine-2,4(1h,3h)-dione
42. Antizona
43. Heratil
44. Lgp81v5245
45. Skf 14287
46. Id2
47. Iduoculos
48. Idurd
49. Mfcd00134656
50. 1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodopyrimidine-2,4-dione
51. Dsstox_cid_25238
52. Dsstox_rid_80769
53. Dsstox_gsid_45238
54. Idoxuridinum [inn-latin]
55. Idoxuridina [inn-spanish]
56. Idossuridina
57. 5-iododesoxyuridine
58. 5-iodo-deoxyuridine
59. Ccris 2827
60. Sr-05000001510
61. Einecs 200-207-8
62. Brn 0030397
63. Unii-lgp81v5245
64. Ai3-50861
65. Hsdb 7479
66. Cas-54-42-2
67. Ncgc00016243-01
68. Dendrid (tn)
69. Uracil, 5-iodo-1-(2-deoxy-beta-d-ribofuranosyl)-
70. Idoxuridine [usan:usp:inn:ban:jan]
71. 5-iodo-2'deoxyuridine
72. Herplex (tn)
73. Idoxuridine [mi]
74. Prestwick3_000018
75. Idoxuridine [inn]
76. Idoxuridine [jan]
77. Idoxuridine [hsdb]
78. Chembl788
79. Epitope Id:138107
80. Idoxuridine [vandf]
81. 5-i-2'-durd
82. Schembl3683
83. 5-iodo-2 Inverted Exclamation Marka-deoxyuridine
84. Idoxuridine [mart.]
85. Bspbio_000055
86. Idoxuridine [usp-rs]
87. Idoxuridine [who-dd]
88. Idoxuridine [who-ip]
89. 4-24-00-01235 (beilstein Handbook Reference)
90. Mls002154175
91. 5-iodo-2-deoxyuridine
92. Bpbio1_000061
93. Idoxuridine (jp17/usp/inn)
94. Dtxsid2045238
95. Schembl15850897
96. 2'-desoxy-5-iodouridine
97. Idoxuridine [orange Book]
98. Hms2090m10
99. Hms2095c17
100. Hms2230e10
101. Hms3712c17
102. Idoxuridine [ep Monograph]
103. Idoxuridine [usp Impurity]
104. Idoxuridine [usp Monograph]
105. Bcp29428
106. Hy-b0307
107. Idoxuridinum [who-ip Latin]
108. Zinc3834173
109. 5-iudr; Idu; Idurd; Idoxuridine
110. Tox21_110324
111. Bdbm50370388
112. S1883
113. Akos015920431
114. Tox21_110324_1
115. (+)-5-iodo-2'-deoxyuridine, 98%
116. Ac-8219
117. Ccg-220018
118. Db00249
119. Smp2_000072
120. Ncgc00179673-01
121. Ncgc00179673-03
122. Ncgc00179673-05
123. As-13469
124. Smr001233472
125. Idoxuridine 100 Microg/ml In Acetonitrile
126. Db-009133
127. 5-iodo-2'-deoxyuridine, >=99% (hplc)
128. I0258
129. Sw198484-2
130. 1-(2-deoxy-ss -d-ribofuranosyl)-5-iodouracil
131. D00342
132. 134i656
133. Q409765
134. J-009907
135. J-700180
136. Sr-05000001510-1
137. Sr-05000001510-3
138. Sr-05000001510-4
139. Brd-k76634210-001-14-7
140. Idoxuridine, European Pharmacopoeia (ep) Reference Standard
141. Idoxuridine, United States Pharmacopeia (usp) Reference Standard
142. 1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-iodo-1,2,3,4-tetrahydropyrimidine-2,4-dione
143. 1-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl]-5-iodo-pyrimidine-2,4-dione
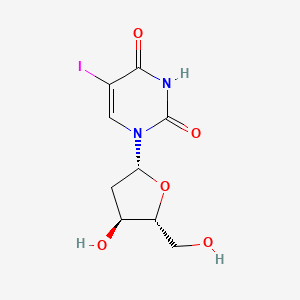
| Molecular Weight | 354.10 g/mol |
|---|---|
| Molecular Formula | C9H11IN2O5 |
| XLogP3 | -1 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Exact Mass | 353.97127 g/mol |
| Monoisotopic Mass | 353.97127 g/mol |
| Topological Polar Surface Area | 99.1 Ų |
| Heavy Atom Count | 17 |
| Formal Charge | 0 |
| Complexity | 386 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 3 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
| 1 of 4 | |
|---|---|
| Drug Name | Dendrid |
| PubMed Health | Idoxuridine (Into the eye) |
| Drug Classes | Antiviral |
| Drug Label | Dendrid (idoxuridine) is an antiviral chemotherapeutic agent prepared in a sterile buffered isotonic solution. The active ingredient is represented by the chemical structure:Established name:IdoxuridineChemical name:Uridine, 2-deoxy-5-iodo-Each... |
| Active Ingredient | Idoxuridine |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Alcon |
| 2 of 4 | |
|---|---|
| Drug Name | Herplex |
| Active Ingredient | Idoxuridine |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Allergan |
| 3 of 4 | |
|---|---|
| Drug Name | Dendrid |
| PubMed Health | Idoxuridine (Into the eye) |
| Drug Classes | Antiviral |
| Drug Label | Dendrid (idoxuridine) is an antiviral chemotherapeutic agent prepared in a sterile buffered isotonic solution. The active ingredient is represented by the chemical structure:Established name:IdoxuridineChemical name:Uridine, 2-deoxy-5-iodo-Each... |
| Active Ingredient | Idoxuridine |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Alcon |
| 4 of 4 | |
|---|---|
| Drug Name | Herplex |
| Active Ingredient | Idoxuridine |
| Dosage Form | Solution/drops |
| Route | Ophthalmic |
| Strength | 0.1% |
| Market Status | Prescription |
| Company | Allergan |
Antiviral
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. 13th Edition, Whitehouse Station, NJ: Merck and Co., Inc., 2001., p. 878
Idoxuridine is indicated in the treatment of keratitis caused by herpes simplex virus (HSV). /Included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Idoxuridine is indicated in the treatment of keratitis caused by vaccinia virus. /Not included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Idoxuridine is used in the treatment of keratoconjunctivitis caused by herpes simplex virus (HSV). /Not included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Idoxuridine has no effect on accumulated scarring, vascularization, or progressive loss of vision that may result from the infection. It also has no effect on corneal inflammation that may follow HSV keratitis when the virus is absent, nor on adenoviral keratoconjunctivitis. /Not included in US product label/
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Patients sensitive to iodine or iodine-containing preparations may be sensitive to this medication also.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
The following side/adverse effects have been selected on the basis of their potential clinical significance: Incidence less frequent /include/ Hypersensitivity (itching, redness, swelling, pain, or other sign of irritation not present before therapy), or increased sensitivity of eyes to light; Incidence rare: Corneal clouding (blurring, dimming, or haziness of vision).
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
FDA Pregnancy Risk Category: C /RISK CANNOT BE RULED OUT. Adequate, well controlled human studies are lacking, and animal studies have shown risk to the fetus or are lacking as well. There is a chance of fetal harm if the drug is given during pregnancy; but the potential benefits may outweigh the potential risk./
Briggs, G.G, R.K. Freeman, S.J. Yaffe. A Reference Guide to Fetal and Neonatal Risk. Drugs in Pregnancy and Lactation. 4th ed. Baltimore, MD: Williams & Wilkins 1994., p. 435
The ... toxicity of topical applications of 30% idoxuridine in dimethyl sulfoxide, dimethyl sulfoxide alone, or saline in 96 recurrent and 39 first episodes of genital herpes simplex virus (HSV) infection were compared. ...Complications in patients given idoxuridine in dimethyl sulfoxide included local burning, generalized contact dermatitis, and vulvar carcinoma in situ. Thirty percent idoxuridine in dimethyl sulfoxide has no effect on clinical manifestations of genital HSV infection and may be hazardous.
PMID:7047788 Silvestri DL et al; JAMA 248 (8): 953-9 (1982)
For use in keratoconjunctivitis and keratitis caused by herpes simplex virus.
In chemical structure idoxuridine closely approximates the configuration of thymidine, one of the four building blocks of DNA (the genetic material of the Herpes virus). As a result, idoxuridine is able to replace thymidine in the enzymatic step of viral replication or "growth". The consequent production of faulty DNA results in a pseudostructure which cannot infect or destroy tissue. In short, by pre-empting a vital building block in the genetic material of the Herpes simplex virus, Herplex-D topical solution destroys the infective and destructive capacity of the viral material. The virus infected cell may only be attacked during the period of active synthesis of DNA. This occurs early in the development of the Herpes simplex lesion, but at different times in different cells. Therefore, ideally, the affected area should remain saturated with the antiviral agent.
Antiviral Agents
Agents used in the prophylaxis or therapy of VIRUS DISEASES. Some of the ways they may act include preventing viral replication by inhibiting viral DNA polymerase; binding to specific cell-surface receptors and inhibiting viral penetration or uncoating; inhibiting viral protein synthesis; or blocking late stages of virus assembly. (See all compounds classified as Antiviral Agents.)
Nucleic Acid Synthesis Inhibitors
Compounds that inhibit cell production of DNA or RNA. (See all compounds classified as Nucleic Acid Synthesis Inhibitors.)
D - Dermatologicals
D06 - Antibiotics and chemotherapeutics for dermatological use
D06B - Chemotherapeutics for topical use
D06BB - Antivirals
D06BB01 - Idoxuridine
J - Antiinfectives for systemic use
J05 - Antivirals for systemic use
J05A - Direct acting antivirals
J05AB - Nucleosides and nucleotides excl. reverse transcriptase inhibitors
J05AB02 - Idoxuridine
S - Sensory organs
S01 - Ophthalmologicals
S01A - Antiinfectives
S01AD - Antivirals
S01AD01 - Idoxuridine
Absorption
Systemic absorption is unlikely following ocular administration even when nasolacrimal secretions are swallowed, since vidarabine is rapidly deaminated in the gastrointestinal tract.
Idoxuridine penetrates the cornea poorly and therefore is ineffective in the treatment of iritis or deep stromal infections.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Idoxuridine crosses the placenta. Studies in humans have not been done.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
It is not known whether idoxuridine is distributed into breast milk. However, problems in humans have not been documented.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
A reproducible microbiologic assay of microgram quantities of idoxuridine (IDU) in serum, urine, or cerebrospinal fluid is presented. The antiviral assay is not interfered with by type-specific antibody or interferon. During slow intravenous infusions of idox-uridine (4 mg/min) in patients with suspected diagnoses of Herpesvirus hominis encephalitis, the rate of inactivation and/or removal of drug exceeded its administration. During several rapid infusions of idoxuridine (50 mg/min) significant quantities of the drug were found in serum, urine, and cerebrospinal fluid. Idoxuridine is not significantly bound to serum proteins and is not deiodinated in fresh serum or urine in vitro to inactive products (iodouracil, uracil, iodide). It is rapidly excreted into the urine. Inactivation of IDU occurs in tissues. This antiviral assay of IDU in body fluids should be applicable to other viruses and potential antiviral agents.
PMID:4331799 Full text: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC332927 Lerner A et al; J Clin Invest 51 (1): 45-9 (1972)
Idoxuridine is rapidly inactivated by deaminases or nucleotidases.
Idoxuridine is rapidly inactivated by deaminases or nucleotidases.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.
Idoxuridine acts as an antiviral agent by inhibiting viral replication by substituting itself for thymidine in viral DNA. This in turn inhibits thymidylate phosphorylase and viral DNA polymerases from properly functioning. The effect of Idoxuridine results in the inability of the virus to reproduce or to infect/destroy tissue.
Idoxuridine, which closely resembles thymidine, inhibits thymidylic phosphorylase and specific DNA polymerases, which are necessary for the incorporation of thymidine into viral DNA. Idoxuridine is incorporated in place of thymidine into viral DNA, resulting in faulty DNA and the inability to infect or destroy tissue or to reproduce. Idoxuridine is incorporated into mammalian DNA as well.
Thomson/Micromedex. Drug Information for the Health Care Professional. Volume 1, Greenwood Village, CO. 2006.

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
37
PharmaCompass offers a list of Idoxuridine API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Idoxuridine manufacturer or Idoxuridine supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Idoxuridine manufacturer or Idoxuridine supplier.
PharmaCompass also assists you with knowing the Idoxuridine API Price utilized in the formulation of products. Idoxuridine API Price is not always fixed or binding as the Idoxuridine Price is obtained through a variety of data sources. The Idoxuridine Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Idoxuridine manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Idoxuridine, including repackagers and relabelers. The FDA regulates Idoxuridine manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Idoxuridine API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Idoxuridine supplier is an individual or a company that provides Idoxuridine active pharmaceutical ingredient (API) or Idoxuridine finished formulations upon request. The Idoxuridine suppliers may include Idoxuridine API manufacturers, exporters, distributors and traders.
click here to find a list of Idoxuridine suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Idoxuridine DMF (Drug Master File) is a document detailing the whole manufacturing process of Idoxuridine active pharmaceutical ingredient (API) in detail. Different forms of Idoxuridine DMFs exist exist since differing nations have different regulations, such as Idoxuridine USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Idoxuridine DMF submitted to regulatory agencies in the US is known as a USDMF. Idoxuridine USDMF includes data on Idoxuridine's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Idoxuridine USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Idoxuridine suppliers with USDMF on PharmaCompass.
National Drug Code is a comprehensive database maintained by the FDA that contains information on all drugs marketed in the US. This directory includes information about finished drug products, unfinished drug products, and compounded drug products, including those containing Idoxuridine as an active pharmaceutical ingredient (API).
The FDA updates the NDC directory daily. The NDC numbers for Idoxuridine API and other APIs are published in this directory by the FDA.
The NDC unfinished drugs database includes product listing information submitted for all unfinished drugs, such as active pharmaceutical ingredients (APIs), drugs intended for further processing and bulk drug substances for compounding.
Pharmaceutical companies that manufacture Idoxuridine as an active pharmaceutical ingredient (API) must furnish the FDA with an updated record of all drugs that they produce, prepare, propagate, compound, or process for commercial distribution in the US at their facilities.
The NDC directory also contains data on finished compounded human drug products that contain Idoxuridine and are produced by outsourcing facilities. While these outsourcing facilities are not mandated to assign a Idoxuridine NDC to their finished compounded human drug products, they may choose to do so.
click here to find a list of Idoxuridine suppliers with NDC on PharmaCompass.
Idoxuridine Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Idoxuridine GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Idoxuridine GMP manufacturer or Idoxuridine GMP API supplier for your needs.
A Idoxuridine CoA (Certificate of Analysis) is a formal document that attests to Idoxuridine's compliance with Idoxuridine specifications and serves as a tool for batch-level quality control.
Idoxuridine CoA mostly includes findings from lab analyses of a specific batch. For each Idoxuridine CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Idoxuridine may be tested according to a variety of international standards, such as European Pharmacopoeia (Idoxuridine EP), Idoxuridine JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Idoxuridine USP).

