
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
API
0
FDF
0
Europe

0
Canada

0
Australia

0
South Africa

DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
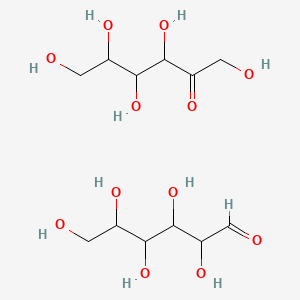

1. Invert Sugar
1. 8013-17-0
2. Invert Sugar
3. Hs 500
4. 2,3,4,5,6-pentahydroxyhexanal;1,3,4,5,6-pentahydroxyhexan-2-one
5. Sugar,invert
6. Schembl20361633
7. Mfcd00148911
8. 1,3,4,5,6-pentahydroxyhexan-2-one; 2,3,4,5,6-pentahydroxyhexanal
9. 2,3,4,5,6-pentahydroxyhexanal Compound With 1,3,4,5,6-pentahydroxyhexan-2-one (1:1)
| Molecular Weight | 360.31 g/mol |
|---|---|
| Molecular Formula | C12H24O12 |
| Hydrogen Bond Donor Count | 10 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 10 |
| Exact Mass | 360.12677620 g/mol |
| Monoisotopic Mass | 360.12677620 g/mol |
| Topological Polar Surface Area | 236 Ų |
| Heavy Atom Count | 24 |
| Formal Charge | 0 |
| Complexity | 285 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 7 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 2 |
Cariogenic Agents
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
SOLN CONTAINING 25% TO 50% DEXTROSE ARE USED IN PARENTERAL HYPERALIMENTATION. THESE MAY BE ADMIN SLOWLY TO PROVIDE AS MUCH AS 3,000-4,000 CALORIES DAILY, & MUST BE GIVEN VIA THE SUPERIOR VENA CAVA OR OTHER EQUALLY LARGE VEIN. /DEXTROSE/
American Medical Association, Council on Drugs. AMA Drug Evaluations. 2nd ed. Acton, Mass.: Publishing Sciences Group, Inc., 1973., p. 181
SOLN CONTAINING 50% TO 75% DEXTROSE DECR THE FREQUENCY OF DIALYSIS IN PATIENTS WITH RENAL FAILURE. /DEXTROSE/
American Medical Association, Council on Drugs. AMA Drug Evaluations. 2nd ed. Acton, Mass.: Publishing Sciences Group, Inc., 1973., p. 181
IF THE INDIVIDUAL PATIENT'S CAPACITY TO UTILIZE DEXTROSE IS EXCEEDED, GLYCOSURIA & DIURESIS WILL OCCUR. /DEXTROSE/
American Medical Association, Council on Drugs. AMA Drug Evaluations. 2nd ed. Acton, Mass.: Publishing Sciences Group, Inc., 1973., p. 181
10% SOLN OF INVERT SUGAR BUFFERED TO PH 6.8 WAS FOUND TO CAUSE SIGNIFICANTLY LOWER INCIDENCE OF THROMBOPHLEBITIS THAN SOLN OF SAME STRENGTH WITH PH BETWEEN 3.5 AND 5.4 FOLLOWING INTRAVENOUS INJECTION OF 500 ML QUANTITIES IN 76 PATIENTS.
Blacow, N. W. (ed.). Martindale: The Extra Pharmacopoeia. 26th ed. London: The Pharmaceutical Press, 1972., p. 81
DEXTROSE INJECTION SHOULD NOT BE USED AS A DILUENT FOR BLOOD BECAUSE IT CAUSES CLUMPING OF RED BLOOD CELLS &, POSSIBLY, HEMOLYSIS. THIS CAN BE AVOIDED BY USING 5% DEXTROSE IN 0.2% OR 0.11% SODIUM CHLORIDE INJECTION. /DEXTROSE/
American Medical Association, Council on Drugs. AMA Drug Evaluations. 2nd ed. Acton, Mass.: Publishing Sciences Group, Inc., 1973., p. 181
SUBCUTANEOUS /ADMIN OF INVERT SUGAR/: GENERALLY DEEMED INADVISABLE.
American Medical Association, Council on Drugs. AMA Drug Evaluations. 2nd ed. Acton, Mass.: Publishing Sciences Group, Inc., 1973., p. 181
For more Drug Warnings (Complete) data for INVERT SUGAR (7 total), please visit the HSDB record page.
Invert sugar presents a large variety of uses. It can be used therapeutically for parenteral hyperalimentation or to be used as an excipient with a known effect. Invert sugar is also approved for its use in food products as a humectant, crystallization modifier, and liquid and nutritive sweetener.
Intravenous administration of invert sugar has been proven to be favorable. These reports indicate a temporary rise, within physiological levels, of lactate, pyruvate, and insulin in the blood plasma whereas the level of free fatty acids was declined.
Cariogenic Agents
Substances that promote DENTAL CARIES. (See all compounds classified as Cariogenic Agents.)
Sweetening Agents
Substances that sweeten food, beverages, medications, etc., such as sugar, saccharine or other low-calorie synthetic products. (From Random House Unabridged Dictionary, 2d ed) (See all compounds classified as Sweetening Agents.)
C - Cardiovascular system
C05 - Vasoprotectives
C05B - Antivaricose therapy
C05BB - Sclerosing agents for local injection
C05BB03 - Invert sugar
Absorption
It has been reported that administration of 10-15% of intravenous invert sugar solution is rapidly absorbed in the intestine and distributed in blood without exceeding the renal threshold. The absorption of invert sugar happens mainly as monosaccharides. Glucose is absorbed into the portal vein by the transporter GLUT2 while fructose is absorbed by the transporter GLUT5.
Route of Elimination
Reports have indicated that after intravenous administration of invert sugar present a reduced content or carbohydrates in urine when compared to the intravenous administration of glucose. This response indicates a reduced diuresis of invert sugar. It has also been reported a reduced diuresis in the presence of invert diuresis.
LESS THAN 2% OF SUGAR IS EXCRETED IN URINE WHEN 1 L OF 10% INVERT SUGAR SOLN IS INFUSED IN 1 HR. WHEN GIVEN OVER A LONGER PERIOD OF TIME INVERT SUGAR IS COMPLETELY UTILIZED AND NONE IS EXCRETED IN URINE.
American Hospital Formulary Service. Volumes I and II. Washington, DC: American Society of Hospital Pharmacists, to 1984., p. 40:20
The metabolism of invert sugar and other fructose-glucose sweeteners is not meaningfully different. Once absorbed, the fructose monosaccharides are taken up by the liver and bypass a key step regulatory step in glycolysis. Both of the monosaccharides first step metabolism is marked by its phosphorylation to glucose-6-phosphate by glucokinase or fructose-1-phosphate by fructokinase. After this step, the glycolysis continues its pathway until the obtention of pyruvic acid. The metabolism of fructose is more rapid than the one of glucose and thus, it is possible to administer at the same speed than a glucose dose correspondent to 55% of the invert sugar dose.
The glucose half-life following intravenous administration of invert sugar is of approximately 30 min. In the same report, the half-life of fructose after the intravenous administration is reported to be 16 min.
ABOUT THIS PAGE


LOOKING FOR A SUPPLIER?