
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
Canada
0
Australia
0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers

1. Hexabrix
2. Ioxaglate
3. Ioxaglate Meglumine
4. Ioxaglate Sodium
5. Ioxaglate, Methylglucamine
6. Ioxaglic Acid Monosodium Salt
7. Ioxaglic Acid, Calcium Salt (2:1)
8. Meglumine, Ioxaglate
9. Methylglucamine Ioxaglate
10. P-286 (contrast Media)
11. P286 (contrast Media)
1. Ioxaglate
2. 59017-64-0
3. Acido Ioxaglico
4. Acide Ioxaglique
5. Acidum Ioxaglicum
6. Hexabrix 320
7. Ioxaglic Acid (100 Mg)
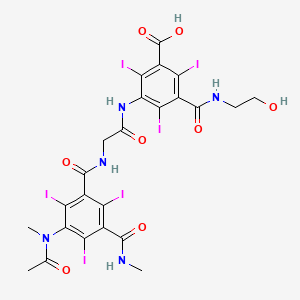
8. 3-[[2-[[3-[acetyl(methyl)amino]-2,4,6-triiodo-5-(methylcarbamoyl)benzoyl]amino]acetyl]amino]-5-(2-hydroxyethylcarbamoyl)-2,4,6-triiodobenzoic Acid
9. N-(2-hydroxyethyl)-2,4,6-triiodo-5-(2-(2,4,6-triiodo-3-(n-methylacetamido)-5-(methylcarbamoyl)benzamido)acetamido)isophthalamic Acid
10. Z40x7ei2af
11. Hexabrix 160; P 286
12. Chebi:31718
13. 3-[(n-{3-[acetyl(methyl)amino]-2,4,6-triiodo-5-(methylcarbamoyl)benzoyl}glycyl)amino]-5-[(2-hydroxyethyl)carbamoyl]-2,4,6-triiodobenzoic Acid
14. Hexabrix 160
15. Ncgc00016886-01
16. Cas-59017-64-0
17. P 286 (contrast Medium)
18. Acide Ioxaglique [inn-french]
19. Acido Ioxaglico [inn-spanish]
20. Acidum Ioxaglicum [inn-latin]
21. Einecs 261-560-1
22. Unii-z40x7ei2af
23. Ioxaglic-acid
24. Hsdb 8077
25. Ioxaglic Acid [usan:usp:inn:ban:jan]
26. Hexabrix 320 (tn)
27. Prestwick0_001062
28. Prestwick1_001062
29. Prestwick2_001062
30. Prestwick3_001062
31. Ioxaglate [vandf]
32. Dsstox_cid_3166
33. Ioxaglic Acid [mi]
34. Dsstox_rid_76899
35. Ioxaglic Acid [inn]
36. Ioxaglic Acid [jan]
37. Dsstox_gsid_23166
38. Schembl38073
39. Bspbio_001044
40. Ioxaglic Acid [usan]
41. Benzoic Acid, 3-((((3-(acetylmethylamino)-2,4,6-triiodo-5-((methylamino)carbonyl)benzoyl)amino)acetyl)amino)-5-(((2-hydroxyethyl)amino)carbonyl)-2,4,6-triiodo-
42. Mls002154135
43. Spbio_002962
44. Ioxaglic Acid [mart.]
45. Bpbio1_001150
46. Ioxaglic Acid (jan/usp/inn)
47. Ioxaglic Acid [usp-rs]
48. Ioxaglic Acid [who-dd]
49. Chembl1201291
50. Dtxsid5023166
51. Hms1571e06
52. Hms2098e06
53. Hms2234i18
54. Hms3371k01
55. Hms3715e06
56. Tox21_110664
57. Ioxaglic Acid [ep Monograph]
58. Ioxaglic Acid [usp Monograph]
59. Ccg-221062
60. Db09313
61. Ncgc00016886-02
62. Ncgc00016886-04
63. Smr001233442
64. Hy-106586
65. Ab00514026
66. Cs-0026109
67. Ft-0627285
68. D01761
69. Sr-01000841818
70. Q6064817
71. Sr-01000841818-2
72. W-111165
73. Brd-k79124250-001-03-0
74. [(methylamino)carbonyl]benzoyl]amino]acetyl]amino]-5-[[(2-hydroxyethyl)amino]carbonyl]-2,4,6-triiodo-
75. 3-{[n-({3-[acetyl(methyl)amino]-2,4,6-triiodo-5-[(methylamino)carbonyl]phenyl}carbonyl)glycyl]amino}-5-{[(2-hydroxyethyl)amino]carbonyl}-2,4,6-triiodobenzoic Acid
76. Benzoic Acid, 3-((((3-(acetylmethylamino)-2,4,6-triiodo-5-9((methylamino)carbonyl)benzoyl)amino)acetyl)amino)-5-(((2-hydroxyethyl)amino)carbonyl)-2,4,6-triiodo-
| Molecular Weight | 1268.9 g/mol |
|---|---|
| Molecular Formula | C24H21I6N5O8 |
| XLogP3 | 2.8 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 10 |
| Exact Mass | 1268.5658 g/mol |
| Monoisotopic Mass | 1268.5658 g/mol |
| Topological Polar Surface Area | 194 Ų |
| Heavy Atom Count | 43 |
| Formal Charge | 0 |
| Complexity | 1090 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Intravascular injection of a radiopaque diagnostic agent opacifies those vessels in the path of the flow of the contrast medium, permitting radiographic visualization of the internal structures of the human body until significant hemodilution occurs.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
Contrast Media
National Library of Medicine's Medical Subject Headings online file (MeSH, 2009)
THERAPEUTIC CATEGORY: Diagnostic aid (radiopaque medium)
O'Neil, M.J. (ed.). The Merck Index - An Encyclopedia of Chemicals, Drugs, and Biologicals. Whitehouse Station, NJ: Merck and Co., Inc., 2006., p. 881
HEXABRIX is indicated for use in pediatric angiocardiography, selective coronary arteriography with or without left ventriculography, peripheral arteriography, aortography, selective visceral arteriography, cerebral angiography, intra-arterial digital subtraction angiography, intravenous digital subtraction angiography, peripheral venography (phlebography), excretory urography, contrast enhancement of computed tomographic head imaging and body imaging, arthrography and hysterosalpingography. /Included in US product label/
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
For more Therapeutic Uses (Complete) data for Ioxaglate (7 total), please visit the HSDB record page.
/BOXED WARNING/ SEVERE ADVERSE EVENTS - INADVERTENT INTRATHECAL ADMINISTRATION: Serious adverse reactions have been reported due to the inadvertent intrathecal administration of iodinated contrast media that are not indicated for intrathecal use. These serious adverse reactions include: death, convulsions, cerebral hemorrhage, coma, paralysis, arachnoiditis, acute renal failure, cardiac arrest, seizures, rhabdomyolysis, hyperthermia, and brain edema. Special attention must be given to insure that this drug product is not administered intrathecally.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
Ionic iodinated contrast media inhibit blood coagulation, in vitro, more than nonionic contrast media. Nonetheless, it is prudent to avoid prolonged contact of blood with syringes containing ionic contrast media. Serious, rarely fatal, thromboembolic events causing myocardial infarction and stroke have been reported during angiographic procedures with both ionic and nonionic contrast media. Therefore, meticulous intravascular administration technique is necessary, particularly during angiographic procedures, to minimize thromboembolic events. Numerous factors, including length of procedure, catheter and syringe material, underlying disease state and concomitant medications may contribute to the development of thromboembolic events. For these reasons, meticulous angiographic techniques are recommended including close attention to guidewire and catheter manipulation, use of manifold systems and/ or three-way stopcocks, frequent catheter flushing with heparinized saline solutions and minimizing the length of the procedure. The use of plastic syringes in place of glass syringes has been reported to decrease but not eliminate the likelihood of in vitro clotting. Serious or fatal reactions have been associated with the administration of iodine containing radiopaque media. It is of utmost importance to be completely prepared to treat any contrast medium reaction.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
As with any contrast medium, serious neurologic sequelae, including permanent paralysis, can occur following cerebral arteriography, selective spinal arteriography and arteriography of vessels supplying the spinal cord. The injection of a contrast medium should never be made following the administration of vasopressors since they strongly potentiate neurologic effects. In patients with subarachnoid hemorrhage, a rare association between contrast administration and clinical deterioration, including convulsions and death, has been reported. Therefore, administration of intravascular iodinated contrast media in these patients should be undertaken with caution.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
A definite risk exists in the use of intravascular contrast agents in patients who are known to have multiple myeloma. In such instances anuria has developed resulting in progressive uremia, renal failure and eventually death. Although neither the contrast agent nor dehydration has separately proved to be the cause of anuria in myeloma, it has been speculated that the combination of both may be causative factors. The risk in myelomatous patients is not a contraindication to the procedure; however, partial dehydration in the preparation of these patients for the examination is not recommended since this may predispose to precipitation of myeloma protein in the renal tubules. No form of therapy, including dialysis, has been successful in reversing the effect. Myeloma, which occurs most commonly in persons over 40, should be considered before instituting intravascular administration of contrast agents.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
For more Drug Warnings (Complete) data for Ioxaglate (29 total), please visit the HSDB record page.
This medicinal product is for diagnostic use only in adults and children as a low-osmolality medium,.
This drug allows for the visualization of important organs and structures in the body. It binds to tissues, allowing the blockage of X-rays and diagnostic visualization in various soft tissues and body cavities. The joint spaces in addition to the uterus and fallopian tubes may be visualized by the direct injection of the contrast medium into the region to be studied.
Contrast Media
Substances used to allow enhanced visualization of tissues. (See all compounds classified as Contrast Media.)
V - Various
V08 - Contrast media
V08A - X-ray contrast media, iodinated
V08AB - Watersoluble, nephrotropic, low osmolar x-ray contrast media
V08AB03 - Ioxaglic acid
Absorption
Following the intravascular route of injection, Ioxaglic acid is rapidly transported through the circulatory system to the kidneys. The pharmacokinetics of radiopaque contrast media given by the IV route are described by a two-compartment model with a rapid alpha phase for drug distribution and a slow beta phase for the elimination of the drug. Following the intravenous administration of 50 mL of ioxaglic acid in 10 healthy volunteers, the mean peak plasma concentration occurred at two (1-3) minutes, reaching a concentration of 2.1 (1.8-2.8) mg/mL. Approximately 50 percent of the intravenously administered dose was recovered in the urine at two hours, and 90% percent was recovered at the 24 hour time point.
Route of Elimination
Excreted unchanged in the urine The liver and small intestine provide the major alternate route of excretion. In patients with severe renal impairment, the excretion of this contrast medium through the gallbladder and into the small intestine sharply increases.
Volume of Distribution
Ioxaglate salts cross the placental barrier in humans and are excreted unchanged in human milk.
Clearance
245 ml/kg
Following intravascular injection, HEXABRIX is rapidly transported through the circulatory system to the kidneys and is excreted unchanged in the urine. The pharmacokinetics of intravascularly administered radiopaque contrast media are usually best described by a two compartment model with a rapid alpha phase for drug distribution and a slower beta phase for drug elimination.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
Following the intravenous administration of 50 mL of HEXABRIX in 10 normal volunteers, the mean peak plasma concentration occurred at two (1-3) minutes, reaching a concentration of 2.1 (1.8-2.8) mg/mL. Approximately 50 (42-67) percent of the intravenously administered dose was recovered in the urine at two hours, and 90 (68-105) percent was recovered at 24 hours.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
Following intravenous injection, HEXABRIX is rapidly excreted by the kidneys. HEXABRIX may be visualized in the renal parenchyma one minute following bolus injection. Maximum radiographic density in the calyces and pelves occurs in most instances within 7 to 12 minutes after injection. In patients with severe renal impairment, contrast visualization may be substantially delayed.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
In brain scanning, contrast media do not accumulate in normal brain tissue due to the blood brain barrier (BBB). The increase in x-ray attenuation usually seen in normal tissue following contrast medium injection is due to the presence of the contrast medium in the blood pool. Disruption in the BBB, such as occurs in malignant tumors of the brain, allows accumulation of contrast medium within the interstitial tumor tissue; adjacent normal brain tissue does not contain the contrast medium.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
For more Absorption, Distribution and Excretion (Complete) data for Ioxaglate (8 total), please visit the HSDB record page.
Excreted unchanged.
Hexabrix 320 is rapidly eliminated by the kidneys with a half-life of about 90 minutes
In 10 patients with normal renal function, the alpha and beta half-lives of HEXABRIX were 12 (4-17) and 92 (61-140) minutes, respectively.
US Natl Inst Health; DailyMed. Current Medication Information for HEXABRIX (ioxaglate meglumine and ioxaglate sodium) injection (December 2010). Available from, as of June 25, 2012: https://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=d9a16f54-c83a-4527-b6fe-11c71a875a41
Ioxaglic acid is an iodine-containing, low osmolality contrast agent. Ioxaglate is a molecule that consists of six iodine atoms and achieves water solubility by ionization; thus, it is a 3.0 ratio ionic agent. By far the most successful and widely used contrast agents in use today are the iodinated contrast media, which were introduced into clinical practice in the 1950s. Approximately 75 million doses of iodinated contrast agents are administered worldwide each year. The iodinated contrast agents fall into 4 groups. Each group has unique chemical, physical, and biologic properties. These various contrast agents are required to meet the demands of a broad variety of imaging modalities. All agents share a similar function groupa tri-iodinated benzene ring. Iodine plays an imperative role in the attenuation of x-ray signal. The atomic radius of a covalently bonded iodine atom is about 133 picometers, falling within the same range of the wavelengths of x-rays: 10 to 10,000 picometers. Therefore, x-rays are easily attenuated by the iodine atoms. In addition, iodine atoms covalently bonded to a benzene ring have 2 main advantages: (1) 3 large atoms located in such close proximity increase the effective molecular size, attenuating longer-wavelength x-rays, and (2) covalent bonding to a stable organic functional group (for example, benzene) decrease the risk of toxic adverse effects from free iodide molecules. Ionization is an important characteristic to note in contrast media preparations. Compounds are classified as either ionic or nonionic. The ionic compounds dissociate in aqueous solution and therefore have a higher osmolality. The anion is the radiopaque portion of the molecule, however, both the anion and cation are osmotically active. Compared with nonionic media such as ioxaglic acid, ionic contrast media have more adverse hemodynamic effects, especially in patients with heart disease. The nonionic media are water soluble but do not dissociate in solution. Nonionic agents possess lower osmolality because there are fewer particles in solution. The nonionic contrast agents are more hydrophilic than ionic agents, resulting in lower osmolality, reduced binding to plasma proteins, decreased tissue binding, and a decreased tendency to cross cell membranes. The low-osmolality of ioxaglic acid reduces the risk of adverse events. The occurrence of adverse reactions is more common after the use of high-osmolarity agents: about 15% with a high-osmolarity agent versus 3% with a low-osmolarity agent. The use of high-osmolarity agents has decreased significantly in recent years. Most adverse effects and adverse reactions to this group of drugs are multifactorial and are likely due to a combination of direct cellular toxicity, the ionic state (for example, ionic vs nonionic), or the osmolarity of the injected contrast drug.
Global Sales Information

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
42
PharmaCompass offers a list of Ioxaglic Acid API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Ioxaglic Acid manufacturer or Ioxaglic Acid supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Ioxaglic Acid manufacturer or Ioxaglic Acid supplier.
PharmaCompass also assists you with knowing the Ioxaglic Acid API Price utilized in the formulation of products. Ioxaglic Acid API Price is not always fixed or binding as the Ioxaglic Acid Price is obtained through a variety of data sources. The Ioxaglic Acid Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Ioxaglic Acid manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Ioxaglic Acid, including repackagers and relabelers. The FDA regulates Ioxaglic Acid manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Ioxaglic Acid API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Ioxaglic Acid supplier is an individual or a company that provides Ioxaglic Acid active pharmaceutical ingredient (API) or Ioxaglic Acid finished formulations upon request. The Ioxaglic Acid suppliers may include Ioxaglic Acid API manufacturers, exporters, distributors and traders.
click here to find a list of Ioxaglic Acid suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Ioxaglic Acid DMF (Drug Master File) is a document detailing the whole manufacturing process of Ioxaglic Acid active pharmaceutical ingredient (API) in detail. Different forms of Ioxaglic Acid DMFs exist exist since differing nations have different regulations, such as Ioxaglic Acid USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Ioxaglic Acid DMF submitted to regulatory agencies in the US is known as a USDMF. Ioxaglic Acid USDMF includes data on Ioxaglic Acid's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Ioxaglic Acid USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Ioxaglic Acid suppliers with USDMF on PharmaCompass.
Ioxaglic Acid Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Ioxaglic Acid GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Ioxaglic Acid GMP manufacturer or Ioxaglic Acid GMP API supplier for your needs.
A Ioxaglic Acid CoA (Certificate of Analysis) is a formal document that attests to Ioxaglic Acid's compliance with Ioxaglic Acid specifications and serves as a tool for batch-level quality control.
Ioxaglic Acid CoA mostly includes findings from lab analyses of a specific batch. For each Ioxaglic Acid CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Ioxaglic Acid may be tested according to a variety of international standards, such as European Pharmacopoeia (Ioxaglic Acid EP), Ioxaglic Acid JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Ioxaglic Acid USP).

