
Synopsis
Synopsis
0
USDMF
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDA Orange Book

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
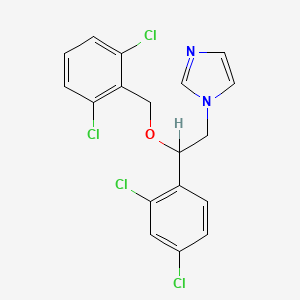

1. 1-(2,4-dichloro Beta-(2,6-dichlorobenzyloxy)phenethyl) Imidazole
2. Fazol
3. Gyno Icaden
4. Gyno-travogen
5. Icaden
6. Isoconazole Nitrate
7. R15454
8. Travogyn
1. 27523-40-6
2. Travogen
3. 1-[2-(2,4-dichlorophenyl)-2-[(2,6-dichlorophenyl)methoxy]ethyl]imidazole
4. Gri7wfr424
5. Isoconazol
6. Chebi:83667
7. R-15,454 [as Nitrate Salt]
8. Isoconazolum
9. 1h-imidazole, 1-(2-(2,4-dichlorophenyl)-2-((2,6-dichlorophenyl)methoxy)ethyl)-
10. Dsstox_cid_25447
11. Dsstox_rid_80886
12. Dsstox_gsid_45447
13. Isoconazol [inn-spanish]
14. Isoconazolum [inn-latin]
15. 1h-imidazole, 1-[2-(2,4-dichlorophenyl)-2-[(2,6-dichlorophenyl)methoxy]ethyl]-
16. Einecs 248-508-3
17. Unii-gri7wfr424
18. Isoconazole [usan:inn:ban]
19. R-15,454
20. 1-{2-[(2,6-dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1h-imidazole
21. 1-(2-((2,6-dichlorobenzyl)oxy)-2-(2,4-dichlorophenyl)ethyl)-1h-imidazole
22. Ncgc00016800-01
23. Prestwick_532
24. Cas-27523-40-6
25. Miconazole Impurity D
26. Isoconazole [mi]
27. 1-(2,4-dichloro-beta-((2,6-dichlorobenzyl)oxy)phenethyl)imidazole
28. Prestwick0_000127
29. Prestwick1_000127
30. Prestwick2_000127
31. Prestwick3_000127
32. Isoconazole (usan/inn)
33. Isoconazole [usan]
34. 1-(2-(2,4-dichlorophenyl)-2-((2,6-dichlorophenyl)methoxy)ethyl)1h-imidazole
35. Cid_3760
36. Isoconazole [mart.]
37. Bspbio_000174
38. Isoconazole [who-dd]
39. Mls002154207
40. Schembl148288
41. R15454 Free Base
42. Spbio_002113
43. Miconazole 2,6-isomer
44. Bpbio1_000192
45. Chembl1571863
46. Dtxsid7045447
47. R-15454 Free Base
48. Bdbm31771
49. Isoconazole [ep Impurity]
50. Hms1568i16
51. Hms2095i16
52. Hms2231p05
53. Hms3374m05
54. Hms3712i16
55. Isoconazole [ep Monograph]
56. Amy32539
57. Tox21_110616
58. Imidazole, 1-(2,4-dichloro-beta-((2,6-dichlorobenzyl)oxy)phenethyl)-
59. Akos015896434
60. Tox21_110616_1
61. Ccg-220127
62. Db08943
63. 1-(2-(2,4-dichlorophenyl)-2-((2,6-dichlorophenyl)methoxy)ethyl)-1h-imidazole
64. Ncgc00179642-01
65. Ncgc00179642-02
66. Ncgc00179642-03
67. Ncgc00179642-04
68. Ncgc00179642-05
69. Smr001233493
70. Miconazole Impurity D [ep Impurity]
71. Ft-0657773
72. D04624
73. A819101
74. Q262210
75. Sr-01000838847
76. Miconazole Nitrate Impurity D [ep Impurity]
77. Sr-01000838847-2
78. Brd-a93353767-001-03-5
79. 1-(2-(2,6-dichlorobenzyloxy)-2-(2,4-dichlorophenyl)ethyl)-1h-imidazole
80. 1-[2,4-dichloro-.beta.-[2,6-dichlorobenzyl)oxy]phenylethyl]imidazole
81. 1-{2-[(2,6-dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}imidazole
82. 1-(2,4-dichloro-.beta.-((2,6-dichlorobenzyl)oxy)phenethyl)imidazole
| Molecular Weight | 416.1 g/mol |
|---|---|
| Molecular Formula | C18H14Cl4N2O |
| XLogP3 | 5.3 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 6 |
| Exact Mass | 415.983074 g/mol |
| Monoisotopic Mass | 413.986024 g/mol |
| Topological Polar Surface Area | 27 Ų |
| Heavy Atom Count | 25 |
| Formal Charge | 0 |
| Complexity | 407 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 1 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Antifungal Agents
Substances that destroy fungi by suppressing their ability to grow or reproduce. They differ from FUNGICIDES, INDUSTRIAL because they defend against fungi present in human or animal tissues. (See all compounds classified as Antifungal Agents.)
D01AC05
S76 | LUXPHARMA | Pharmaceuticals Marketed in Luxembourg | Pharmaceuticals marketed in Luxembourg, as published by d'Gesondheetskeess (CNS, la caisse nationale de sante, www.cns.lu), mapped by name to structures using CompTox by R. Singh et al. (in prep.). List downloaded from https://cns.public.lu/en/legislations/textes-coordonnes/liste-med-comm.html. Dataset DOI:10.5281/zenodo.4587355
D - Dermatologicals
D01 - Antifungals for dermatological use
D01A - Antifungals for topical use
D01AC - Imidazole and triazole derivatives
D01AC05 - Isoconazole
G - Genito urinary system and sex hormones
G01 - Gynecological antiinfectives and antiseptics
G01A - Antiinfectives and antiseptics, excl. combinations with corticosteroids
G01AF - Imidazole derivatives
G01AF07 - Isoconazole

API/FDF Prices: Book a Demo to explore the features and consider upgrading later
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Global Sales Information

Market Place

REF. STANDARDS & IMPURITIES

ABOUT THIS PAGE
16
PharmaCompass offers a list of Isoconazole API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Isoconazole manufacturer or Isoconazole supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Isoconazole manufacturer or Isoconazole supplier.
PharmaCompass also assists you with knowing the Isoconazole API Price utilized in the formulation of products. Isoconazole API Price is not always fixed or binding as the Isoconazole Price is obtained through a variety of data sources. The Isoconazole Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Isoconazole manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Isoconazole, including repackagers and relabelers. The FDA regulates Isoconazole manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Isoconazole API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Isoconazole supplier is an individual or a company that provides Isoconazole active pharmaceutical ingredient (API) or Isoconazole finished formulations upon request. The Isoconazole suppliers may include Isoconazole API manufacturers, exporters, distributors and traders.
click here to find a list of Isoconazole suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Isoconazole CEP of the European Pharmacopoeia monograph is often referred to as a Isoconazole Certificate of Suitability (COS). The purpose of a Isoconazole CEP is to show that the European Pharmacopoeia monograph adequately controls the purity of Isoconazole EP produced by a given manufacturer. Suppliers of raw materials can prove the suitability of Isoconazole to their clients by showing that a Isoconazole CEP has been issued for it. The manufacturer submits a Isoconazole CEP (COS) as part of the market authorization procedure, and it takes on the role of a Isoconazole CEP holder for the record. Additionally, the data presented in the Isoconazole CEP (COS) is managed confidentially and offers a centralized system acknowledged by numerous nations, exactly like the Isoconazole DMF.
A Isoconazole CEP (COS) is recognised by all 36 nations that make up the European Pharmacopoeia Convention. Isoconazole CEPs may be accepted in nations that are not members of the Ph. Eur. at the discretion of the authorities there.
click here to find a list of Isoconazole suppliers with CEP (COS) on PharmaCompass.
Isoconazole Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Isoconazole GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Isoconazole GMP manufacturer or Isoconazole GMP API supplier for your needs.
A Isoconazole CoA (Certificate of Analysis) is a formal document that attests to Isoconazole's compliance with Isoconazole specifications and serves as a tool for batch-level quality control.
Isoconazole CoA mostly includes findings from lab analyses of a specific batch. For each Isoconazole CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Isoconazole may be tested according to a variety of international standards, such as European Pharmacopoeia (Isoconazole EP), Isoconazole JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Isoconazole USP).
