
Synopsis
Synopsis
0
API Suppliers
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
EDQM
0
USP
0
JP
0
Others
0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
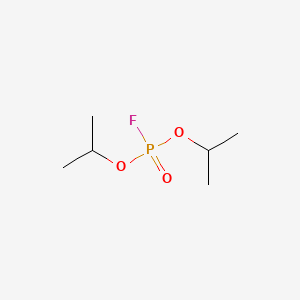

1. Bis(1-methylethyl) Phosphorofluoridate
2. Dfp
3. Di Isopropylphosphorofluoridate
4. Di-isopropylphosphorofluoridate
5. Diisopropylfluorophosphate
6. Diisopropylphosphofluoridate
7. Dyflos
8. Floropryl
9. Fluorostigmine
10. Fluostigmine
11. Isoflurophate
1. Isoflurophate
2. Fluostigmine
3. Diisopropyl Phosphorofluoridate
4. 55-91-4
5. Diisopropylfluorophosphate
6. Isofluorphate
7. Dyflos
8. Floropryl
9. Neoglaucit
10. Diflupyl
11. Isoflurophosphate
12. Diflurophate
13. Isopropyl Fluophosphate
14. Isofluorophate
15. Fluorodiisopropyl Phosphate
16. Diisopropoxyphosphoryl Fluoride
17. Diisopropyl Fluorophosphonate
18. Diisopropyl Phosphofluoridate
19. Isopropyl Phosphorofluoridate
20. Diisopropylphosphorofluoridate
21. Diisopropylfluorfosfat
22. Diisopropylfluorophosphoric Acid Ester
23. O,o-diisopropyl Fluorophosphate
24. Pf-3
25. O,o'-diisopropyl Phosphoryl Fluoride
26. Phosphorofluoridic Acid, Bis(1-methylethyl) Ester
27. Rcra Waste Number P043
28. Tl 466
29. Fluophosphoric Acid, Diisopropyl Ester
30. Ea 1152
31. Diisopropylfluorphosphorsaeureester
32. Phosphorofluoridic Acid, Diisopropyl Ester
33. T-1703
34. Bis(propan-2-yl) Fluorophosphate
35. Chembl1025
36. 12uhw9r67n
37. Fluorostigmine
38. Chebi:17941
39. Nsc-727370
40. Ncgc00168783-01
41. Fluoropryl
42. Fluropryl
43. Isoflurophate [usp]
44. Diisopropylphosphofluoridate
45. Difp
46. Diisopropylfluorfosfat [czech]
47. Dfp (pesticide)
48. Diisopropyl Fluoridophosphate
49. Disiopropyl Fluorophosphonate
50. Floropryl (tn)
51. Fluorophosphoric Acid, Diisopropyl Ester
52. Hsdb 2133
53. Diisopropylfluorphosphorsaeureester [german]
54. Einecs 200-247-6
55. Rcra Waste No. P043
56. Brn 1723307
57. Unii-12uhw9r67n
58. 2-[fluoro(propan-2-yloxy)phosphoryl]oxypropane
59. Dipropan-2-yl Phosphorofluoridoate
60. Mfcd00008873
61. Phosphorofluoridic Acid Diisopropyl Ester
62. Dyflos [mart.]
63. Isoflurophate [mi]
64. Di-isopropyl Fluorophosphate
65. Dsstox_cid_20667
66. Dsstox_rid_79530
67. Isoflurophate [hsdb]
68. Dsstox_gsid_40667
69. Schembl63841
70. Isoflurophate [vandf]
71. Diisopropyl Fluoridophosphate #
72. Isoflurophate [who-dd]
73. Dtxsid1040667
74. Bis(propan-2-yl) Fluorophosphonate
75. Isoflurophate [orange Book]
76. Zinc8214587
77. Isoflurophate [usp Impurity]
78. Tox21_112643
79. Bdbm50022772
80. Geo-01118
81. Hsci1_000366
82. Pdsp1_000466
83. Pdsp2_000464
84. Akos005258363
85. O,o''-diisopropyl Phosphoryl Fluoride
86. Db00677
87. Nsc 727370
88. Cas-55-91-4
89. Db-052820
90. Diisopropylfluorophosphate, >=97.0% (gc)
91. C00202
92. D00043
93. Q384993
| Molecular Weight | 184.15 g/mol |
|---|---|
| Molecular Formula | C6H14FO3P |
| XLogP3 | 1.2 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 4 |
| Exact Mass | 184.06645946 g/mol |
| Monoisotopic Mass | 184.06645946 g/mol |
| Topological Polar Surface Area | 35.5 Ų |
| Heavy Atom Count | 11 |
| Formal Charge | 0 |
| Complexity | 144 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
Cholinesterase Inhibitors; Miotics; Parasympathomimetics; Protease Inhibitors
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
OCCASIONALLY BY PHYSICIANS (OPHTHALMOLOGISTS) FOR TREATMENT OF GLAUCOMA.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-299
IT IS USED TOPICALLY IN TREATMENT OF PRIMARY OPEN-ANGLE GLAUCOMA, BUT ONLY WHEN SHORT-ACTING MIOTICS HAVE FAILED. ... ALSO USED IN TREATMENT OF APHAKIC GLAUCOMA & ACCOMMODATIVE ESOTROPIA. WITHIN A DAY INTRAOCULAR TENSION DROPS, & ... MAY REMAIN DEPRESSED FOR A ... WK. MIOSIS LASTS 2 TO 4 WK.
Osol, A. (ed.). Remington's Pharmaceutical Sciences. 16th ed. Easton, Pennsylvania: Mack Publishing Co., 1980., p. 840
IRREVERSIBLE ANTICHOLINESTERASE ... ISOFLUROPHATE ... /IS/ ONLY OF HISTORICAL INTEREST IN TREATMENT OF MYASTHENIA GRAVIS ... UNSATISFACTORY BECAUSE OF ... TOXICITY.
American Medical Association, AMA Department of Drugs, AMA Drug Evaluations. 3rd ed. Littleton, Massachusetts: PSG Publishing Co., Inc., 1977., p. 1018
For more Therapeutic Uses (Complete) data for DIISOPROPYL FLUOROPHOSPHATE (10 total), please visit the HSDB record page.
... Should be used cautiously in patients with bronchial asthma, bradycardia, or hypotension. An increase in blood pressure may occur occasionally due to a nicotinic effect on sympathetic ganglia.
American Medical Association, AMA Department of Drugs. AMA Drug Evaluations. 5th ed. Chicago: American Medical Association, 1983., p. 449
Because of their cataractogenic properties & other toxicity, /diisopropyl fluorophosphate/ should be reserved for patients refractory to short-acting miotics, epinephrine, beta-blocking drugs, & possibly, carbonic anhydrase inhibitors. /Long-acting miotics, including floropryl/
American Medical Association, AMA Department of Drugs. AMA Drug Evaluations. 5th ed. Chicago: American Medical Association, 1983., p. 447
For use in the eye to treat certain types of glaucoma and other eye conditions, such as accommodative esotropia.
Isoflurophate is used as ocular drops in the treatment of chronic glaucoma. Isoflurophate is an organophosphorus compound that acts as an irreversible cholinesterase inhibitor. As such, it displays parasympathomimetic effects. Isoflurophate is used in the eye to treat certain types of glaucoma and other eye conditions, such as accommodative esotropia. They may also be used in the diagnosis of certain eye conditions, such as accommodative esotropia. Isoflurophate damages the acetylcholinesterase enzyme and is therefore irreversible, however, pralidoxime can displace organophosphates such as isoflurophate from acetylcholinesterase, but only if administered before isoflurophate damages (alkylates) the enzyme.
Cholinesterase Inhibitors
Drugs that inhibit cholinesterases. The neurotransmitter ACETYLCHOLINE is rapidly hydrolyzed, and thereby inactivated, by cholinesterases. When cholinesterases are inhibited, the action of endogenously released acetylcholine at cholinergic synapses is potentiated. Cholinesterase inhibitors are widely used clinically for their potentiation of cholinergic inputs to the gastrointestinal tract and urinary bladder, the eye, and skeletal muscles; they are also used for their effects on the heart and the central nervous system. (See all compounds classified as Cholinesterase Inhibitors.)
Protease Inhibitors
Compounds which inhibit or antagonize biosynthesis or actions of proteases (ENDOPEPTIDASES). (See all compounds classified as Protease Inhibitors.)
S - Sensory organs
S01 - Ophthalmologicals
S01E - Antiglaucoma preparations and miotics
S01EB - Parasympathomimetics
S01EB07 - Fluostigmine
WHEN SMALL AMOUNTS (0.1 MG/KG) OF DFP WERE ADMIN TO GUINEA PIGS, DFP WAS PREFERENTIALLY BOUND BY SERUM & BY THE LUNGS. ADMIN OF LARGE AMOUNTS (3-6 MG/KG) PRODUCED UNIFORM BODY DISTRIBUTION & ACCUMULATION IN KIDNEYS & LIVER. DFP WAS CONVERTED IN PART TO DIISOPROPYL PHOSPHATE (DP). BOTH DFP & DP WERE EXCRETED MAINLY IN THE URINE. SOME APPEARED IN BILE.
Menzie, C.M. Metabolism of Pesticides. U.S. Department of the Interior, Bureau of Sport Fisheries and Wildlife, Publication 127. Washington, DC: U.S. Government Printing Office, 1969., p. 142
TRANSPLACENTAL PASSAGE OF MORE POLAR INSECTICIDES, DFP ... HAS BEEN INFERRED FROM FETAL CHOLINESTERASE INHIBITION.
The Chemical Society. Foreign Compound Metabolism in Mammals Volume 3. London: The Chemical Society, 1975., p. 633
... Its high lipid solubility, low molecular weight, and volatility facilitate inhalation and transdermal absorption. DFP also readily penetrates the central nervous system.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 167
The effects of skin metabolism on percutaneous penetration of drugs with high lipophilicity were studied in vitro using rat skin pretreated with and without an esterase inhibitor, isoflurophate (diisopropylphosphofluoridate; DFP). Without diisopropylphosphofluoridate, about 96% of the total penetrated amount appeared as metabolized p-hydroxybenzoic acid in the receptor fluid after application of butylparaben, whereas about 30% penetrated as intact form after application of propylparaben. On the other hand, metabolized p-hydroxybenzoic acid was not detected in the receptor fluid under pretreatment with DFP. DFP significantly decreased the total amount that penetrated after application of butylparaben, but it did not significantly affect that of propylparaben. It was concluded that skin metabolism directly affected the total amount that penetrated in the case of highly lipophilic drugs, and that the higher the metabolic rate to hydrophilic drugs, the greater the amount that penetrated the skin.
PMID:9188062 Bando H et al; J Pharm Sci 86 (Jun): 759-761 (1997)
HEN EGG-WHITE LYSOZYME WAS ALLOWED TO REACT WITH AN EXCESS OF DFP @ 25 C & AT PH VALUES RANGING FROM 9.5 TO 11.0. ANALYSIS INDICATED THAT ALKYLPHOSPHORYLATION OF TYROSYL HYDROXYL GROUPS HAD OCCURRED & ... SOME OTHER AMINO ACID RESIDUES HAD ALSO BEEN PHOSPHORYLATED. SIMILAR RESULTS ... OBTAINED WITH STEM BROMELAIN, TAKA-AMYLASE A, & PAPAIN.
Menzie, C. M. Metabolism of Pesticides, An Update. U.S. Department of the Interior, Fish, Wild-life Service, Special Scientific Report - Wildlife No. 184, Washington, DC: U.S. Government Printing Office, l974., p. 152
... DIISOPROPYL FLUOROPHOSPHONATE, UNDERGOES DEFLUORINATION IN BOTH MAMMALS & BACTERIA.
Parke, D. V. The Biochemistry of Foreign Compounds. Oxford: Pergamon Press, 1968., p. 126
DFP is rapidly metabolized to diisopropylphosphate & is excreted, mainly in the urine. Less than 1% is eliminated by bile & lung within the first 2 hr.
Hayes, Wayland J., Jr. Pesticides Studied in Man. Baltimore/London: Williams and Wilkins, 1982., p. 332
WHEN SMALL AMOUNTS (0.1 MG/KG) OF DFP WERE ADMIN TO GUINEA PIGS, DFP WAS ... CONVERTED IN PART TO DIISOPROPYL PHOSPHATE (DP) ... DFP WAS DEGRADED BY PSEUDOMONAS MELOPHTHORA BUT NO METABOLITES WERE IDENTIFIED. BLUEGILL (LEPOMIS MACROCHIRUS RAFINESQUE) & CHANNEL CATFISH (ICTALURUS PUNCTALUS WALBAUM) CONVERTED DFP TO DIISOPROPYL PHOSPHATE.
Menzie, C.M. Metabolism of Pesticides. U.S. Department of the Interior, Bureau of Sport Fisheries and Wildlife, Publication 127. Washington, DC: U.S. Government Printing Office, 1969., p. 142
An enzyme in Escherichia coli hydrolyzes isofluorophate. A superficially similar but distinctly different enzyme is found in squid nerve. The results of this study suggest that while several tissues of the squid contain only this second kind of DFP hydrolyzing enzyme, termed squid type DFPase, many other sources incl E coli contain a mixt of squid type DFPase (the name not strictly indicative of source) and the other DFP hydrolyzing enzyme, now termed Mazur type DFPase.
Hoskin FC et al; Two Enzymes for the Detoxication of Organophosphorus Compounds--Sources, Similarities, and Significance: Fundam Appl Toxicol 4 (2, Pt 2): S165-72 (1984)
Following iv injection of tritium-labeled DFP, the concn in arterial serum declined in two exponential phases with t1/2 of about 7 & 200 minutes, respectively, reflecting, first, fast accumulation of the compound & its metabolites in the tissue &, second, elimination.
Hayes, Wayland J., Jr. Pesticides Studied in Man. Baltimore/London: Williams and Wilkins, 1982., p. 332
The mechanism of isoflurophate's action involves the irreversible inhibition of cholinesterase.
Passive avoidance retention and cortical (3)H-quinuclidinyl benzilate (QNB) binding were exam in rats that were chronically treated with isofluorophate. Retention of a passive avoidance response was significantly lower when compared to vehicle-treated controls. Passive avoidance retention decr from 93% in control animals to 68% in DFP-treated rats. QNB binding studies revealed the density of muscarinic receptors in cortical homogenates was significantly reduced from 0.95 +/- 0.04 pmol/mg protein in controls to 0.72 +/- 0.04 pmol/mg protein in DFP-treated rats. Based on data that DFP causes a redn in cholinergic receptors, this study supports the hypothesis that central cholinergic receptors are assoc with mechanisms involved in memory storage.
Gardner R et al; A Possible Mechanism for Diisopropylfluorophosphate-Induced Memory Loss in Rats. Pharmacol Biochem Behav 21 (1): 43-6 (1984)
The organophosphorus inhibitors, such as DFP, serve as true hemisubstrates, since the resultant conjugate with the active center serine phosphorylated or phosphonylated is extremely stable. ... If the alkyl groups in the phosphorylated enzyme are ethyl or methyl, a significant degree of spontaneous regeneration of active enzyme requires several hours. Secondary (as in DFP) or tertiary alkyl groups enhance the stability of the phosphorylated enzyme, & significant regeneration of active enzyme is not observed. Hence, the return of acetylcholinesterase activity depends on synthesis of new enzyme.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 163
The characteristic pharmacological effects of the anticholinesterase agents are due primarily to the prevention of hydrolysis of acetylcholine by acetylcholinesterase at sites of cholinergic transmission. The transmitter thus accumulates, & the action of acetylcholine that is liberated by cholinergic impulses that leak from the nerve ending is enhanced. With most of the organophosphorus agents, such as DFP, virtually all the acute effects of moderate doses are attributable to this action. For example, the characteristic miosis that follows local application of DFP to the eye is not observed after chronic postganglionic denervation of the eye because there is no effective source of endogenous acetylcholine.
Hardman, J.G., L.E. Limbird, P.B. Molinoff, R.W. Ruddon, A.G. Goodman (eds.). Goodman and Gilman's The Pharmacological Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill, 1996., p. 165
Rats were treated with DFP using 1 or 2 mg/kg acutely, or with 1 mg/kg daily for 4, 14 or 28 days. Tremors, chewing movements and hind-limb abduction induced by DFP incr in a steeply dose-dependent manner. Tremor occurred in a complex spectrum of slow to intense fast types. Except for chewing, tolerance developed for these parameters, but at different rates. After acute treatment striatal dopamine (DA) and dihydroxyphenylacetic acid (DOPAC) levels were altered and the DOPAC/DA ratios were consistently incr within about the first 2 hr, suggesting an incr turnover of DA. It is suggested that the changes in DA metab arose secondarily to an elevation of brain acetylcholine following cholinesterase inhibition. A prolonged change in the levels or turnover of DA could be responsible for incr of postsynaptic DA receptor density previously found, which might then partly mediate the behavioral tolerance to DFP.
Fernando JC et al; Effect on Striatal Dopamine Metabolism and Differential Motor Behavioral Tolerance Following Chronic Cholinesterase Inhibition with diisopropylfluorophosphate; Pharmacol Biochem Behav 20 (6): 951-7 (1984)
For more Mechanism of Action (Complete) data for DIISOPROPYL FLUOROPHOSPHATE (10 total), please visit the HSDB record page.
ABOUT THIS PAGE
24
PharmaCompass offers a list of Isoflurophate API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Isoflurophate manufacturer or Isoflurophate supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Isoflurophate manufacturer or Isoflurophate supplier.
PharmaCompass also assists you with knowing the Isoflurophate API Price utilized in the formulation of products. Isoflurophate API Price is not always fixed or binding as the Isoflurophate Price is obtained through a variety of data sources. The Isoflurophate Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Isoflurophate manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Isoflurophate, including repackagers and relabelers. The FDA regulates Isoflurophate manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Isoflurophate API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Isoflurophate supplier is an individual or a company that provides Isoflurophate active pharmaceutical ingredient (API) or Isoflurophate finished formulations upon request. The Isoflurophate suppliers may include Isoflurophate API manufacturers, exporters, distributors and traders.
Isoflurophate Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Isoflurophate GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Isoflurophate GMP manufacturer or Isoflurophate GMP API supplier for your needs.
A Isoflurophate CoA (Certificate of Analysis) is a formal document that attests to Isoflurophate's compliance with Isoflurophate specifications and serves as a tool for batch-level quality control.
Isoflurophate CoA mostly includes findings from lab analyses of a specific batch. For each Isoflurophate CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Isoflurophate may be tested according to a variety of international standards, such as European Pharmacopoeia (Isoflurophate EP), Isoflurophate JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Isoflurophate USP).
