

Synopsis
Synopsis
0
USDMF
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
FDF
0
FDA Orange Book

0
Europe

0
Australia

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
EDQM
0
USP
0
JP
0
Others
0
US Patents
0
US Exclusivities
0
Health Canada Patents
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
US Medicaid
NA
Annual Reports
NA
Finished Drug Prices
NA
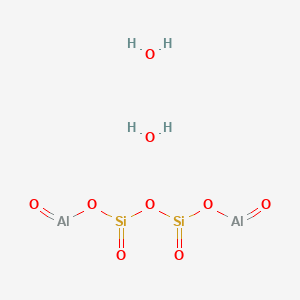

1. Kaolinite
1. 1332-58-7
2. China Clay
3. Argilla
4. Porcelain Clay
5. Bolus Alba
6. White Bole
7. Kaolin(superfine)
8. Glomax
9. Light Kaolin
10. Hydrated Aluminum Silicate
11. Kaolin Clay Slurry
12. Bentone
13. Alphagloss
14. Altowhites
15. Alfaplate
16. Alphacoat
17. Altowhite
18. Apsilex
19. Argiflex
20. Comalco
21. Electros
22. Emathlite
23. Hydrite
24. Kaopaous
25. Langford
26. Mcnamee
27. Parclay
28. Vanclay
29. Fitrol
30. Arcilla Blanca
31. Oxo-oxoalumanyloxy-[oxo(oxoalumanyloxy)silyl]oxysilane;dihydrate
32. Argilla Alba
33. Comalco Kaolin
34. Osmo Kaolin
35. Kaolin Colloidal
36. Beta Coat
37. Astra-glaze
38. Snow Tex
39. Asp Ultrafine
40. Kao-gel
41. Century Hc
42. Kaophills-2
43. Aa Kaolin
44. Clay (kaolin)
45. Acidic White Clays
46. Bol Blanc
47. Asp (mineral)
48. Burgess 10
49. Airflo V 8
50. Amazon 88
51. Amazon 90
52. Argirec B 22
53. Argirec Kn 15
54. Bilt Plate 156
55. Fitrol Desiccite 25
56. Clays, White, Acidic
57. Amazon Kaolin 855d
58. Kaolin [usp:jan]
59. Cb 1 (clay)
60. Cb 2 (clay)
61. Clay 347
62. Aluminum Silicate Hydroxide
63. Unii-24h4nwx5co
64. Metakaolin
65. Kaolinum
66. Clay Nanopowder
67. Clay Slurry
68. Kaolin Nanoclay
69. Nanoclay Powder
70. Ccris 9288
71. Hsdb 630
72. Perlite Clay Mix
73. Clay Nanoparticles
74. Epa Pesticide Chemical Code 100104
75. Expanded Perlite Powder
76. Perlite Nanoclay Powder
77. Halloysite Clay Nanotubes
78. Aluminium Silicate Dihydrate
79. Aluminum Silicate (hydrated)
80. Nanoclay Montmorillonite 20a
81. Expanded Perlite Nanoparticles
82. Montmorillonite Bentonite Clay
83. Halloysite Nanotubes Properties
84. Chembl3833365
85. Chebi:140503
86. Nanoclay Modified Asphalt Materials
87. Expanded Perlite Powder / Nanopowder
88. Shelsite 30b Montmorillonite Nanoparticles
89. Ft-0627569
90. J-521617
91. 95077-05-7
| Molecular Weight | 258.16 g/mol |
|---|---|
| Molecular Formula | Al2H4O9Si2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 9 |
| Rotatable Bond Count | 4 |
| Exact Mass | 257.902462 g/mol |
| Monoisotopic Mass | 257.902462 g/mol |
| Topological Polar Surface Area | 98 Ų |
| Heavy Atom Count | 13 |
| Formal Charge | 0 |
| Complexity | 167 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 3 |
Antidiarrheals; Pharmaceutic Aids
National Library of Medicine's Medical Subject Headings online file (MeSH, 1999)
/Kaolin/ either alone or as...mixt with pectin, is used medicinally as an adsorbent...of value chiefly in treatment of diarrhea caused by agents capable of being adsorbed, as, for example, diarrhea of food poisoning or dysentery.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 751
Kaolin has...been used in treatment of chronic ulcerative colitis, but it is doubtful whether any adsorptive capacity is retained by time prepn reaches colon.
Osol, A. and J.E. Hoover, et al. (eds.). Remington's Pharmaceutical Sciences. 15th ed. Easton, Pennsylvania: Mack Publishing Co., 1975., p. 751
VET: adsorbent, protective antidiarrheal. A physical adsorbent with hydrophilic capacity. Topical skin & GI protectant. It incr bulk & slows .. Passage of feces. Has/ ability to absorb toxins...topically & intrauterine in addn to GI.
Rossoff, I.S. Handbook of Veterinary Drugs. New York: Springer Publishing Company, 1974., p. 297
For more Therapeutic Uses (Complete) data for KAOLIN (8 total), please visit the HSDB record page.
... Formulation changes ... have occurred with Kaopectate ... The current Kaopectate marketed is not the formulation ... with Kaolin and Pectin. In the late 1980's or early '90s, the manufacturer reformulated the product to contain attapulgite. Then in 2003, the sponsor reformulated the product again, this time to include bismuth subsalicylate as the active ingredient. ...
FDA Safety Drug Page. Drug Topics April 19, 2004. Available from the website www.fda.gov/cder/drug/mederrors/kaopectate.pdf as of July 25, 2007
If/...kaolin-containing product must be used...should be given...2 hr before lincomycin. If this...proves impractical, lincomycin may be administered by injection or a nondiarrhea-producing antibiotic should be substituted.
Evaluations of Drug Interactions. 2nd ed. and supplements. Washington, DC: American Pharmaceutical Assn., 1976, 1978., p. 136
Inert except for dangers of obstruction, perforation, & granuloma formation.
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-95
The use of antidiarrheal mixtures containing opioids or poorly absorbed antibacterial agents with adsorbents and protectants (most commonly, kaolin and pectin) and antispasmodic agents is unwarranted, since additional benefits beyond those afforded by the single effective agent are questionable and the patient is subjected to the added expense and the combined adverse effects of the individual ingredients.
American Medical Association, Council on Drugs. AMA Drug Evaluations Annual 1994. Chicago, IL: American Medical Association, 1994., p. 935
Practically non-toxic: probable oral lethal dose (human) above 15 g/kg; more than 1 quart (2.2 lb) for 70 kg person (150 lb).
Gosselin, R.E., R.P. Smith, H.C. Hodge. Clinical Toxicology of Commercial Products. 5th ed. Baltimore: Williams and Wilkins, 1984., p. II-95
Antidiarrheals
Miscellaneous agents found useful in the symptomatic treatment of diarrhea. They have no effect on the agent(s) that cause diarrhea, but merely alleviate the condition. (See all compounds classified as Antidiarrheals.)
A - Alimentary tract and metabolism
A07 - Antidiarrheals, intestinal antiinflammatory/antiinfective agents
A07B - Intestinal adsorbents
A07BC - Other intestinal adsorbents
A07BC02 - Kaolin
Kaolin and pectin are not absorbed following oral administration.
McEvoy, G.K. (ed.). American Hospital Formulary Service--Drug Information 94. Bethesda, MD: American Society of Hospital Pharmacists, Inc. 1994 (Plus Supplements)., p. 1879

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
About the Company : Nitika Pharmaceutical Specialities was found in 1991 as a small manufacturing unit has grown today into a dynamic production center for a range of products finding applications in ...

 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
API Imports and Exports
| Importing Country | Total Quantity (KGS) |
Average Price (USD/KGS) |
Number of Transactions |
|---|
Upgrade, download data, analyse, strategize, subscribe with us
Related Excipient Companies

Dosage Form : Tablet
Grade : Topical, Oral
Dosage Form : Tablet
Grade : Topical, Oral
Dosage Form : Suspension
Grade : Oral, Topical
Application : Thickeners and Stabilizers
Excipient Details : Pectin is used as a thickening and stabilizing agent in the production of pharmaceutical suspensions and creams.
Dosage Form : Suspension
Grade : Oral
Dosage Form : Tablet
Grade : Oral
Dosage Form : Cream / Lotion / Ointment
Grade : Topical
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Orodispersible Tablet
Grade : Oral
Dosage Form : Softgels
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Suspension
Grade : Oral
Application : Thickeners and Stabilizers
Excipient Details : Blanose CMC 7MF is used as a thickener and viscosity modifier in liquid formulations such as suspensions.
Pharmacopoeia Ref : EP, USP/NF
Technical Specs : N/A
Ingredient(s) : Sodium Carboxymethyl Cellulose Excipient
Dosage Form : Tablet
Grade : Oral
Application : Emulsifying Agents
Excipient Details : HDK N20 Pharma is used as a pharmaceutical emulsifying agent in tablets, capsules, syrups, and solutions.
Dosage Form : Syrup
Grade : Oral
Application : Taste Masking
Excipient Details : Sodium Saccharin (Q-0300, 15-191201) is used as an artificial sweetener in tablets, syrup, suspensions, liquids, and mouthwashes.
Pharmacopoeia Ref : USP/NF, JP, Kosher, ISO, Halal...
Technical Specs : 15% MOISTURE, Min. 98% Purity
Ingredient(s) : Sodium Saccharin Excipient
Dosage Form : Ophthalmic Solution
Grade : Topical, Parenteral
Dosage Form : Tablet
Grade : Oral
Brand Name : Titanium dioxide PRETIOX ...
Application : Coating Systems & Additives
Excipient Details : Titanium dioxide Pretiox AV01FG is used as a coloring and coating agent in oral solid dosage forms such as capsules, tablets, granules, and pellets.
Pharmacopoeia Ref : Fami-QS, Kosher, Halal, OHSAS ...
Technical Specs : Ti 59.95% and O 40.05%
Ingredient(s) : Titanium Dioxide
Dosage Form : Tablet
Grade : Oral
Dosage Form : Suspension
Grade : Topical, Oral
Dosage Form : Syrup
Grade : Topical, Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Suspension
Grade : Oral and Topical
Brand Name : Magnesium aluminium silic...
Application : Thickeners and Stabilizers
Excipient Details : Magnesium aluminium silicate is used as opacifying agent, anticaking agent, viscosity agent, suspending agents.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Magnesium aluminium silicate Excipient
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Not Available
Application : Emulsifying Agents
Excipient Details : Glidant; Emulsion Stabilizer; Anti-caking Agent.
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Cream / Lotion / Ointment
Grade : Topical and Oral
Application : Topical
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Oral
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Softgel Capsule
Grade : Not Available
Application : Soft Gelatin
Dosage Form : Syrup
Grade : Not Available
Application : Taste Masking
Excipient Details : Act as a bulk sweetener and vehicle in liquid dosage forms and as a humectant in semi solids.
Pharmacopoeia Ref : EP/USP, USP/FCC
Technical Specs : Not Available
Ingredient(s) : Sorbitol liquid crystallizing
Dosage Form : Syrup
Grade : Not Available
Brand Name : NEOSORB® 70/02 SB
Application : Taste Masking
Excipient Details : Act as a bulk sweetener and vehicle in liquid dosage forms and as a humectant in semi solids.
Pharmacopoeia Ref : EP/USP
Technical Specs : Not Available
Ingredient(s) : Sorbitol liquid crystallizing
Dosage Form : Injectable / Parenteral
Grade : Not Available
Brand Name : NEOSORB® BIOPHARMA
Application : Parenteral
Excipient Details : Suitable for use in biopharmaceutical manufacturing and as an excipient for injectable dosage forms
Pharmacopoeia Ref : USP/EP/JP
Technical Specs : Not Available
Ingredient(s) : Sorbitol, low endotoxin
Dosage Form : Tablet
Grade : Not Available
Dosage Form : Injectable / Parenteral
Grade : Not Available
Application : Parenteral
Excipient Details : A pyrogen-free sorbitol used as a carbohydrate source and osmotic diuretic agent in large volume parenteral injectables.
Dosage Form : Capsule
Grade : Not Available
Brand Name : POLYSORB® 85/70/00
Application : Film Formers & Plasticizers
Excipient Details : It is a plasticizer in soft capsules and orally dissolving films. It can be used in pharmaceutical dosage forms.
Pharmacopoeia Ref : EP/USP
Technical Specs : Not Available
Ingredient(s) : Sorbitol liquid, partially dehydrated
Dosage Form : Tablet
Grade : Not Available
Brand Name : ReadiLYCOAT® D CLEAR 110...
Application : Film Formers & Plasticizers
Excipient Details : A natural inert polymer and ready-to-use coating system for fast aqueous film coating saves up to 50% or more time.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Hydroxypropyl Pea Starch, Sorbitol, Stearic Acid
Dosage Form : Tablet
Grade : Not Available
Brand Name : ReadiLYCOAT® D CLEAR 110...
Application : Film Formers & Plasticizers
Excipient Details : A natural inert polymer and ready-to-use coating system for fast aqueous film coating saves up to 50% or more time.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Hydroxypropyl Pea Starch, Sorbitol, Stearic Acid
Dosage Form : Tablet
Grade : Not Available
Brand Name : ReadiLYCOAT® D WHITE 010...
Application : Film Formers & Plasticizers
Excipient Details : A natural inert polymer and ready-to-use coating system for fast aqueous film coating saves up to 50% or more time.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Hydroxypropyl Pea Starch, Sorbitol, Titanium Dioxide, Stearic Acid
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Syrup
Grade : Not Available
Application : Taste Masking
Pharmacopoeia Ref : Not Available
Technical Specs : Pharma Grade/Food Grade
Ingredient(s) : Sorbitol
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Tablet
Grade : Oral
Application : Taste Masking
Excipient Details : HiCel CE15 offers a superior mouthfeel with less chalkiness and gritness in chewable tablets and orally disintegrating tablets (ODTs).
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Guar Gum Excipient
Dosage Form : Emulsion
Grade : Oral
Application : Rheology Modifiers
Excipient Details : HiCel MCG is use in oral suspensions as a stabilizer & emulsion prepared with high shear forces. It is a good binder for tablets & excellent stabilizer as well as thickener.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Carboxymethyl cellulose sodium
Dosage Form : Tablet
Grade : Oral
Application : Fillers, Diluents & Binders
Excipient Details : HiCel SMCC has unique combination which result in optimum compaction & superior flow properties.
Pharmacopoeia Ref : Not Available
Technical Specs : Not Available
Ingredient(s) : Microcrystalline Cellulose
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Dosage Form : Solution
Grade : Oral, Topical
Application : Solubilizers
Excipient Details : Hydrosol 50 is used as a solubilizer and emulsifying agent in oral and topical liquid and semi-solid dosage forms.
Pharmacopoeia Ref : USP/NF
Technical Specs : N/A
Ingredient(s) : Polyoxyl 40 Hydrogenated Castor Oil
 FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]
FULL SCREEN VIEW Click here to open all results in a new tab [this preview display 10 results]Excipients by Applications
Dosage Form : Cream / Lotion / Ointment, Ophthalmic Solution
Grade : Topical, Parenteral
Category : Solubilizers, Topical
Dosage Form : Cream / Lotion / Ointment, Emulsion, Gel, Suspension
Grade : Topical
Category : Emulsifying Agents, Solubilizers, Surfactant & Foaming Agents, Thickeners and Stabilizers, Topical
Dosage Form : Cream / Lotion / Ointment, Suspension, Solution, Syrup
Grade : Topical, Oral
Category : Rheology Modifiers, Thickeners and Stabilizers, Topical
Dosage Form : Cream / Lotion / Ointment, Gel, Injectable / Parenteral, Tablet
Grade : Topical
Category : Parenteral, Solubilizers, Topical
Dosage Form : Cream / Lotion / Ointment, Emulsion, Injectable / Parenteral, Softgels, Tablet
Grade : Oral, Topical, Parenteral
Category : Film Formers & Plasticizers, Parenteral, Topical
Dosage Form : Emulsion
Grade : Topical
Category : Emulsifying Agents, Surfactant & Foaming Agents, Topical
Dosage Form : Cream / Lotion / Ointment, Suspension, Solution, Syrup
Grade : Topical, Oral
Category : Rheology Modifiers, Thickeners and Stabilizers, Topical
Dosage Form : Cream / Lotion / Ointment, Gel, Injectable / Parenteral, Softgels
Grade : Parenteral, Oral, Topical
Category : Film Formers & Plasticizers, Parenteral, Thickeners and Stabilizers, Topical
Dosage Form : Capsule, Cream / Lotion / Ointment
Grade : Topical and Oral
Category : Film Formers & Plasticizers, Topical
Application : Film Formers & Plasticizers, Topical
Dosage Form : Cream / Lotion / Ointment, Gel, Solution
Grade : Oral, Topical
Category : Emulsifying Agents, Solubilizers, Topical
Application : Emulsifying Agents, Solubilizers, Topical
Excipient Details : Hydrosol 50 is used as a solubilizer and emulsifying agent in oral and topical liquid and semi-solid dosage forms.
Pharmacopoeia Ref : USP/NF
Technical Specs : N/A
Ingredient(s) : Polyoxyl 40 Hydrogenated Castor Oil
Market Place

ABOUT THIS PAGE
We have 1 companies offering Kaolin
Get in contact with the supplier of your choice:


LOOKING FOR A SUPPLIER?
