
Synopsis
Synopsis
0
CEP/COS
0
JDMF
0
EU WC
0
KDMF
0
NDC API
0
VMF
0
Listed Suppliers
0
FDF Dossiers
0
FDA Orange Book

0
Europe

0
Canada

0
Australia

0
South Africa

0
Listed Dossiers
DRUG PRODUCT COMPOSITIONS
0
US Patents
0
US Exclusivities
0
Health Canada Patents
US Medicaid
NA
Annual Reports
NA
Regulatory FDF Prices
NA
0
API
0
FDF
0
Data Compilation #PharmaFlow
0
Stock Recap #PipelineProspector
0
Weekly News Recap #Phispers
0
News #PharmaBuzz
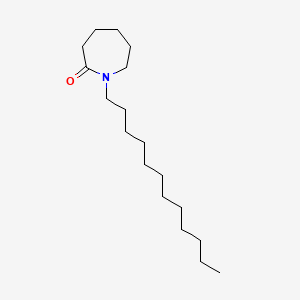

1. 1-dodecylazacycloheptan-2-one
2. 1-dodecylhexahydro-2h-azepin-2-one
3. Azone
1. 59227-89-3
2. N-dodecylcaprolactam
3. 1-dodecylazepan-2-one
4. Azone
5. Tranzone
6. 1-dodecylazacycloheptan-2-one
7. 2h-azepin-2-one, 1-dodecylhexahydro-
8. N-lauryl Caprolactam
9. 1-dodecylhexahydro-2h-azepin-2-one
10. N-0252
11. 1-n-dodecylazacycloheptan-2-one
12. 1f3x9drv9x
13. N-dodecyl-.epsilon.-caprolactam
14. Laurocapramum [latin]
15. Caswell No. 064a
16. Laurocapramum
17. Laurocapram [usan:inn]
18. Usaf Nd-72
19. 1-dodecyl-2-azepanone
20. Einecs 261-668-9
21. Unii-1f3x9drv9x
22. Brn 0167211
23. Trazone
24. Hsdb 7660
25. Laurocapram,(s)
26. N 0252
27. N-n-dodecylcaprolactam
28. Laurocapram [mi]
29. Laurocapram [inn]
30. Laurocapram (usan/inn)
31. Laurocapram [hsdb]
32. Laurocapram [inci]
33. Laurocapram [usan]
34. 2h-azepin-2-one, Hexahydro-n-lauryl-
35. Laurocapram (water Soluble)
36. Dsstox_cid_22086
37. Dsstox_rid_79921
38. Laurocapram [mart.]
39. Dsstox_gsid_42086
40. Schembl23347
41. N-dodecyl-epsilon-caprolactam
42. 4-21-00-03206 (beilstein Handbook Reference)
43. Chembl76591
44. 1-dodecylazacycloheptane-2-one
45. Dtxsid0042086
46. 1-dodecyl-2-azepanone, Aldrichcpr
47. Bcp10858
48. Zinc8214596
49. Tox21_301757
50. Mfcd00190580
51. S4841
52. Stl320722
53. Akos015843226
54. Ccg-267257
55. Cs-w010042
56. Hy-w009326
57. Ks-5054
58. Ncgc00255788-01
59. Ac-11681
60. Cas-59227-89-3
61. Db-053343
62. Ft-0627723
63. L0317
64. D04678
65. 227l893
66. A832216
67. Sr-01000944876
68. Q-201282
69. Sr-01000944876-1
70. Q27252353
71. Laurocapram; Azon; Azone; 1-dodecylazacycloheptan-2-one; N-dodecylcaprolactam; 1-dodecylhexahydro-2h-azepin-2-one
| Molecular Weight | 281.5 g/mol |
|---|---|
| Molecular Formula | C18H35NO |
| XLogP3 | 5.9 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 11 |
| Exact Mass | 281.271864740 g/mol |
| Monoisotopic Mass | 281.271864740 g/mol |
| Topological Polar Surface Area | 20.3 Ų |
| Heavy Atom Count | 20 |
| Formal Charge | 0 |
| Complexity | 240 |
| Isotope Atom Count | 0 |
| Defined Atom Stereocenter Count | 0 |
| Undefined Atom Stereocenter Count | 0 |
| Defined Bond Stereocenter Count | 0 |
| Undefined Bond Stereocenter Count | 0 |
| Covalently Bonded Unit Count | 1 |
1-Dodecylazacycloheptan-2-one (Azone) is a new agent that enhances the percutaneous absorption of a number of different chemicals. This report delineates the enhancement of penetration of clindamycin phosphate, erythromycin base, fusidate sodium, fluorouracil, desonide, amcinonide, and triamcinolone acetonide. For this purpose 1-dodecylazacycloheptan-2-one can be used in concentrations as low as 1%. It is colorless, relatively odorless, nontoxic, and can be applied neat to human skin without any irritation.
PMID:7092271 Stoughton RB; Arch Dermatol 118 (7): 474-7 (1982)
/EXP THER/ To assess the safety and tolerability of a topical gel formulation combining methotrexate and laurocapram and to obtain preliminary information on the therapeutic potential of methotrexate-laurocapram in patients with early-stage mycosis fungoides (stage IA or IB). An open-label, phase 1/2 pilot study /was conducted/. ...Ten patients 18 years or older with histologically confirmed stage IA or IB mycosis fungoides. ... The gel formulation of methotrexate-laurocapram was applied to the total body surface, excluding genital, perianal areas, nipples, face, and skin under the breasts, on an every-other-day basis for 24 consecutive weeks. The safety of methotrexate-laurocapram was assessed in this study by reviewing adverse events and laboratory data. Efficacy outcomes included changes in lesion condition and severity assessments, reduction in area of sample lesions, and the investigator's global evaluation. Adverse events consisted of skin reactions of mild severity. No clinically significant laboratory abnormalities were observed. Based on the investigator's global evaluation at the end of the treatment phase (week 24), 7 (78%) of 9 patients demonstrated a slight-to-moderate response to treatment with methotrexate-laurocapram. Statistical significance (P =.049) was reached for induration and pruritus, a trend (P =.10) was observed for erythema, and no change was found for scaling (P =.37). These findings indicate that the topical administration of methotrexate-laurocapram is safe and in general well tolerated. This treatment may represent a new therapeutic potential for patients with mycosis fungoides.
PMID:12756099 Demierre MF et al; Arch Dermatol 139 (5): 624-8 (2003)
Azone (1-dodecylazacycloheptan-2-one) is an agent that has been shown to enhance percutaneous absorption of drugs. Azone is thought to act by partitioning into skin lipid bilayers and thereby disrupting the structure. An open-label study was done with nine volunteers (two males, seven females; aged 51-76 years) in which Azone cream (1.6%; 100 mg) was topically dosed on a 5 x 10-cm area of the ventral forearm for 21 consecutive days. On days 1, 8, and 15, the Azone cream contained 47 uCi of (14)C Azone. The skin application site was washed with soap and water after each 24-hr dosing. Percutaneous absorption was determined by urinary radioactivity excretion. The (14)C Azone was ring labeled [14C-2-cyclo-heptan]. Radiochemical purity was >98.6% and cold Azone purity was 99%. Percutaneous absorption of the first dose (day 1) was 1.84 +/- 1.56% (SD) of applied dose for 24-hr skin application time. Day 8 percutaneous absorption, after repeated application, increased significantly (p<0.002) to 2.76 +/- 1.91%. Day 15 percutaneous absorption, after continued repeated application, stayed the same at 2.72 +/- 1.21%. In humans, repeated application of Azone results in an initial self-absorption enhancement, probably due to its mechanism of action. However, steady-state percutaneous absorption of Azone is established after this initial change. Thus, Azone can enhance its own absorption as well as that of other compounds. This should be considered relevant for any pharmacological or toxicological evaluation. Washing the skin site of application with soap and water only recovered 1-2% of applied radioactivity. Previous published studies recovered the Azone dose with ethanol washes. Thus, there could potentially be an accumulation of Azone in skin.
PMID:8169776 Wester RC et al; J Pharm Sci 83 (2): 124-5 (1994)
Absorption enhancers are substances used for temporarily increasing a membrane's permeability (e.g., the skin and mucosa), either by interacting with its components (lipids or proteins) or by increasing the membrane/vehicle partition coefficient. ... The enhancing effect of Laurocapram (Azone) is attributed to different mechanisms, such as insertion of its dodecyl group into the intercellular lipidic bilayer, increase of the motion of the alkylic chains of lipids, and fluidization of the hydrophobic regions of the lamellate structure. ...
PMID:16556532 Lopez-Cervantes M et al; Drug Dev Ind Pharm 32 (3): 267-86 (2006)
REF. STANDARDS & IMPURITIES

ANALYTICAL

ABOUT THIS PAGE
38
PharmaCompass offers a list of Laurocapram API manufacturers, exporters & distributors, which can be sorted by GMP, USDMF, JDMF, KDMF, CEP (COS), WC, Price,and more, enabling you to easily find the right Laurocapram manufacturer or Laurocapram supplier for your needs.
Send us enquiries for free, and we will assist you in establishing a direct connection with your preferred Laurocapram manufacturer or Laurocapram supplier.
PharmaCompass also assists you with knowing the Laurocapram API Price utilized in the formulation of products. Laurocapram API Price is not always fixed or binding as the Laurocapram Price is obtained through a variety of data sources. The Laurocapram Price can also vary due to multiple factors, including market conditions, regulatory modifications, or negotiated pricing deals.
A Laurocapram manufacturer is defined as any person or entity involved in the manufacture, preparation, processing, compounding or propagation of Laurocapram, including repackagers and relabelers. The FDA regulates Laurocapram manufacturers to ensure that their products comply with relevant laws and regulations and are safe and effective to use. Laurocapram API Manufacturers are required to adhere to Good Manufacturing Practices (GMP) to ensure that their products are consistently manufactured to meet established quality criteria.
A Laurocapram supplier is an individual or a company that provides Laurocapram active pharmaceutical ingredient (API) or Laurocapram finished formulations upon request. The Laurocapram suppliers may include Laurocapram API manufacturers, exporters, distributors and traders.
click here to find a list of Laurocapram suppliers with USDMF, JDMF, KDMF, CEP, GMP, COA and API Price related information on PharmaCompass.
A Laurocapram DMF (Drug Master File) is a document detailing the whole manufacturing process of Laurocapram active pharmaceutical ingredient (API) in detail. Different forms of Laurocapram DMFs exist exist since differing nations have different regulations, such as Laurocapram USDMF, ASMF (EDMF), JDMF, CDMF, etc.
A Laurocapram DMF submitted to regulatory agencies in the US is known as a USDMF. Laurocapram USDMF includes data on Laurocapram's chemical properties, information on the facilities and procedures used, and details about packaging and storage. The Laurocapram USDMF is kept confidential to protect the manufacturer’s intellectual property.
click here to find a list of Laurocapram suppliers with USDMF on PharmaCompass.
Laurocapram Active pharmaceutical ingredient (API) is produced in GMP-certified manufacturing facility.
GMP stands for Good Manufacturing Practices, which is a system used in the pharmaceutical industry to make sure that goods are regularly produced and monitored in accordance with quality standards. The FDA’s current Good Manufacturing Practices requirements are referred to as cGMP or current GMP which indicates that the company follows the most recent GMP specifications. The World Health Organization (WHO) has its own set of GMP guidelines, called the WHO GMP. Different countries can also set their own guidelines for GMP like China (Chinese GMP) or the EU (EU GMP).
PharmaCompass offers a list of Laurocapram GMP manufacturers, exporters & distributors, which can be sorted by USDMF, JDMF, KDMF, CEP (COS), WC, API price, and more, enabling you to easily find the right Laurocapram GMP manufacturer or Laurocapram GMP API supplier for your needs.
A Laurocapram CoA (Certificate of Analysis) is a formal document that attests to Laurocapram's compliance with Laurocapram specifications and serves as a tool for batch-level quality control.
Laurocapram CoA mostly includes findings from lab analyses of a specific batch. For each Laurocapram CoA document that a company creates, the USFDA specifies specific requirements, such as supplier information, material identification, transportation data, evidence of conformity and signature data.
Laurocapram may be tested according to a variety of international standards, such as European Pharmacopoeia (Laurocapram EP), Laurocapram JP (Japanese Pharmacopeia) and the US Pharmacopoeia (Laurocapram USP).
